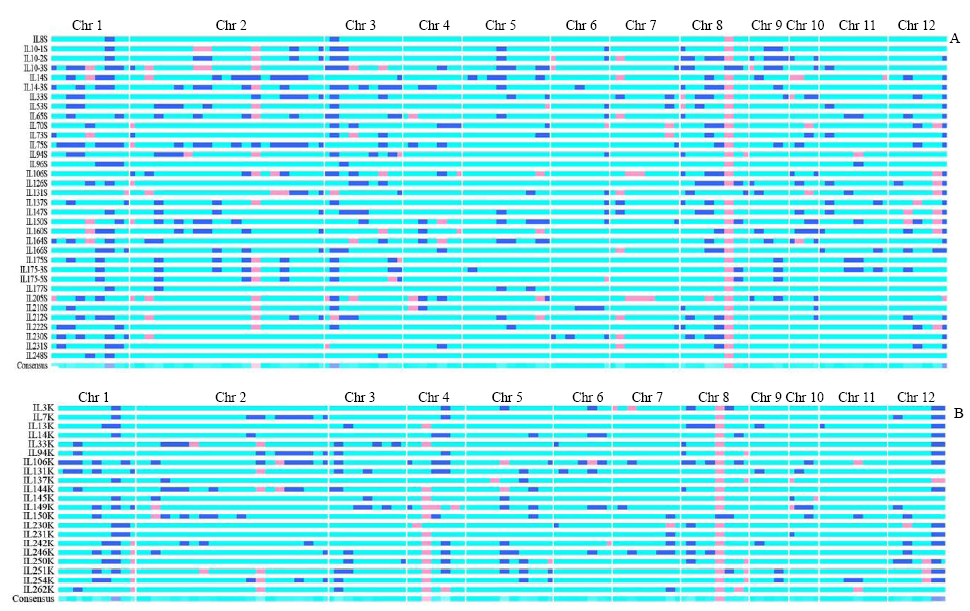
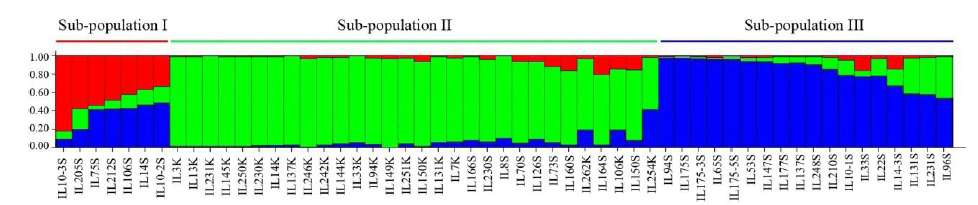
| [1] |
Abdel-Rahman M A, El-Denary M E, Ammar M H, Abdelkhalik A F, Draz A E, Dora S A.2016. QTL validation for grain yield and nitrogen use efficiency under different nitrogen levels in rice.Egypt J Genet Cytol, 44(2): 235-251.
|
| [2] |
All India Coordinated Rice Improvement Project (AICRIP). Annual Progress Report.2009-2010. Hyderabad, India: Directorate of Rice Research: 176.
|
| [3] |
Anandan A, Anumalla M, Pradhan S K, Ali J.2016. Population structure, diversity and trait association analysis in rice (Oryza sativa L.) germplasm for early seedling vigor (ESV) using trait linked SSR markers. PLoS One, 11: e0152406.
|
| [4] |
Andaya V C, Mackill D J.2003. Mapping of QTLs associated with cold tolerance during the vegetative stage in rice.J Exp Bot, 54: 2579-2585.
|
| [5] |
Arbelaez J D, Moreno L T, Singh N, Tung C W, Maron L G, Ospina Y, Martinez C P, Grenier C, Lorieux M, McCouch S.2015. Development and GBS-genotyping of introgression lines (ILs) using two wild species of rice,O. meridionalis and O. rufipogon, in a common recurrent parent, O. sativa cv. Curinga. Mol Breeding, 35: 81.
|
| [6] |
Bai X F, Wu B, Xing Y Z.2012. Yield-related QTLs and their applications in rice genetic improvement.J Integr Plant Biol, 54(5): 300-311.
|
| [7] |
Balakrishnan D, Subrahmanyam D, Badri J, Raju A K, Rao Y V, Beerelli K, Mesapogu S, Surapaneni M, Ponnuswamy R, Padmavathi G, Babu V R, Neelamraju S.2016. Genotype × environment interactions of yield traits in backcross introgression lines derived from Oryza sativa cv. Swarna/Oryza nivara. Front Plant Sci, 7: 1530.
|
| [8] |
Brar D S, Khush G S.2004. Alien introgression in rice.Plant Mol Biol, 35: 35-47.
|
| [9] |
Chapman M A, Abbott R J.2010. Introgression of fitness genes across a ploidy barrier.New Phytol, 186(1): 63-71.
|
| [10] |
Cheema K K, Bains N S, Mangat G S, Das A, Vikal Y, Brar D S, Khush G S, Singh K.2008. Development of high yielding IR64 ×Oryza rufipogon(Griff.) introgression lines and identification of introgressed alien chromosome segments using SSR markers. Euphytica, 160(3): 401-409.
|
| [11] |
Chen J W, Wang L, Pang X F, Pan Q H.2006. Genetic analysis and fine mapping of a rice brown planthopper (Nilaparvata lugens Stål) resistance gene bph19(t). Mol Genet Genom, 275(4): 321-329.
|
| [12] |
Chen S, Wang L, Que Z Q, Pan R Q, Pan Q H.2005. Genetic and physical mapping of Pi37(t), a new gene conferring resistance to rice blast in the famous cultivar St. No. 1. Theor Appl Genet, 11(8): 1563-1570.
|
| [13] |
Derakhshandeh B, Javed M A, Lee C T, Md Afendi S A, Huyop F Z. 2013. Molecular characterization of Malaysian rice germplasm by using microsatellite markers for variety identification. In: International Conference on Sustainable Environment and Agriculture. Singapore: IACSIT Press.
|
| [14] |
Dey S, Badri J, Prakasam V, Bhadana V P, Eswari K B, Laha G S, Priyanka C, Rajkumar A, Ram T.2016. Identification and agro-morphological characterization of rice genotypes resistant to sheath blight.Aust Plant Pathol, 45: 145-153.
|
| [15] |
Evanno G, Regnaut S, Goudet J.2005. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study.Mol Ecol, 14(8): 2611-2620.
|
| [16] |
Fasahat P, Abdullah A, Muhammad K, Karupaiah T, Ratnam W.2012. Red pericarp advanced breeding lines derived from Oryza rufipogon × Oryza sativa: Physicochemical properties, total antioxidant activity, phenolic compounds and vitamin E content. Adv J Food Sci Technol, 4(3): 155-165.
|
| [17] |
Fu X L, Lu Y G, Liu X D, Li J Q.2008. Progress on transferring elite genes from non-AA genome wild rice into Oryza sativa through interspecific hybridization. Rice Sci, 15(2): 79-87.
|
| [18] |
Gaikwad K B, Singh N, Bhatia D, Kaur R, Bains N S, Bharaj T S, Singh K.2014. Yield-enhancing heterotic QTL transferred from wild species to cultivated riceOryza sativa L. PLoS One, 9: e96939.
|
| [19] |
Ganeshan P, Jain A, Parmar B, Rao A R, Sreenu K, Mishra P, Mesapogu S, Subrahmanyam D, Ram T, Sarla N, Rai V.2016. Identification of salt tolerant rice lines among interspecific BILs developed by crossingOryza sativa × O. rufipogon and O. sativa × O. nivara. Aust J Crop Sci, 10(2): 220-228.
|
| [20] |
Guo L B, Ye G Y.2014. Use of major quantitative trait loci to improve grain yield of rice.Rice Sci, 21(2): 65-82.
|
| [21] |
Guo S B, Wei Y, Li X Q, Liu K Q, Huang F K, Chen C H, Gao G Q.2013. Development and identification of introgression lines from cross of Oryza sativa and Oryza minuta. Rice Sci, 20(2): 95-102.
|
| [22] |
Gur A, Zamir D.2004. Unused natural variation can lift yield barriers in plant breeding.PLoS Biol, 2: e245.
|
| [23] |
Han X Y, Yang Y, Wang X M, Zhou J, Zhang W H, Yu C L, Cheng C, Cheng Y, Yan C Q, Chen J P.2014. Quantitative trait loci mapping for bacterial blight resistance in rice using bulked segregant analysis.Int J Mol Sci, 15(7): 11847-11861.
|
| [24] |
Haritha G, Sudhakar T, Chandra D, Ram T, Divya B, Sarla N.2016. Informative ISSR markers help identify genetically distinct accessions of Oryza rufipogon in yield improvement. Rice Sci, 23(5): 225-241.
|
| [25] |
Hasan M M, Rafii M Y, Ismail M R, Mahmood M, Rahim H A, Alam M A, Ashkani S, Malek M A, Latif M A.2015. Marker-assisted backcrossing: A useful method for rice improvement.Biotechnol Biotech Equip, 29(2): 237-254.
|
| [26] |
Jena K K, Khush G S.2000. Exploitation of species in rice improvement: Opportunities, achievements and future challenges. In: Nanda J S. Rice Breeding and Genetic: Research Priorities and Challenges. Enfield: Science Publication: 269-284.
|
| [27] |
Jena K K, Kim S M.2010. Current status of brown planthopper (BPH) resistance and genetics.Rice, 3: 161-171.
|
| [28] |
Jewel Z A, Patwary A K, Maniruzzaman S, Barua R, Begum S N.2011. Physico-chemical and genetic analysis of aromatic rice (Oryza sativa L.) germplasm. Agriculturists, 9: 82-88.
|
| [29] |
Jiang L R, Xu B, Huang R Y, Chen F Y, Dong K M, Huang Y M, Wang H C, Tao Y, Zheng J S, Fang X J.2015. Identification and genetic characterization of a newd10 mutated indica rice germplasm. Plant Gene Trait, 6(6): 1-12.
|
| [30] |
Jiang N, Ferguson A A, Slotkin R K, Lisch D.2011. Pack-mutator-like transposable elements (PackMULEs) induce directional modification of genes through biased insertion and DNA acquisition.Proc Natl Acad Sci USA, 108(4): 1537-1542.
|
| [31] |
Kaladhar K, Swamy B P M, Babu A P, Reddy C S, Sarla N.2008. Mapping quantitative trait loci for yield traits in BC2F2 population derived from Swarna ×O. nivara cross. Rice Genet Newsl, 24: 34-36.
|
| [32] |
Koseki M, Kitazawa N, Yonebayashi S, Maehara Y, Wang Z X, Minobe Y.2010. Identification and fine mapping of a major quantitative trait locus originating from wild rice, controlling cold tolerance at the seedling stage.Mol Genet Genom, 284(1): 45-54.
|
| [33] |
Kubo T, Yoshimura A, Kurata N.2016. Pollen killer gene S35 function requires interaction with an activator that maps close to S24, another pollen killer gene in rice. G3: Genes Genom Genet, 6(5): 1459-1468.
|
| [34] |
Kumar B, Gomez S M, Boopathi N M, Kumar S S, Kumaresan D, Biji K R, Babu B K, Prasad N S R, Shanmugasundaram P, Babu R C.2005. Identification of microsatellite markers associated with drought tolerance in rice (Oryza sativa L.) using bulked line analysis. Trop Agric Res, 17: 39-47.
|
| [35] |
Kwak K J, Kim J Y, Kim Y O, Kang H.2007. Characterization of transgenic Arabidopsis plants over expressing high mobility group B proteins under high salinity, drought or cold stress. Plant Cell Physiol, 48(2): 221-231.
|
| [36] |
Lakshmi V J, Swamy B P M, Kaladhar K, Sarla N.2010. BPH resistance in introgression lines of Swarna/Oryza nivara and KMR3/O. rufipogon. DRR News Lett, 8: 4.
|
| [37] |
Lang N T, Buu B C.2008. Development of PCR-based markers for aroma (fgr) gene in rice(Oryza sativa L.). Omonrice, 16: 16-23.
|
| [38] |
Lang N T, Buu B C, Ismail A.2008. Molecular mapping and marker-assisted selection for salt tolerance in rice (Oryza sativa L.). Omonrice, 16: 50-56.
|
| [39] |
Lang N T, Buu B C.2010. Initial marker-assisted selection in rice breeding at cuu long delta rice research institute.Omonrice, 17: 8-21.
|
| [40] |
Li J, Xu P, Neng D X, Zhou J W, Hu F Y, Wan J M, Tao D Y.2008. Identification of four genes for stable hybrid sterility.Euphytica, 164: 699-708.
|
| [41] |
Li S C, Wang S Q, Deng Q M, Zheng A P, Zhu J, Liu H N, Wang L X, Gao F Y, Zou T, Huang B, Cao X M, Xu L Z, Yu C, Ai P, Li P.2012. Identification of genome-wide variations among three elite restorer lines for hybrid rice.PLoS One, 7: e30952.
|
| [42] |
Li S Q, Yang G H, Li S B, Li Y S, Chen Z Y, Zhu Y G.2005. Distribution of fertility-restorer genes for wild-abortive and Honglian CMS lines of rice in the AA genome species of genus Oryza. Ann Bot, 96(3): 461-466.
|
| [43] |
Madurangi S A P, Ratnasekera D, Senanayake S G J N, Samarasinghe W L G, Hemachandra P V.2013. Antixenosis and antibiosis effects of Oryza nivara accessions harbouring bph2 gene on brown planthopper [Nilaparvata lugens(Stal)]. J Nat Sci Found Sri Lanka, 41(2): 147-154.
|
| [44] |
Mahender A, Anandan A, Pradhan S K.2015. Early seedling vigour, an imperative trait for direct-seeded rice: An overview on physio-morphological parameters and molecular markers.Planta, 241(5): 1027-1050.
|
| [45] |
Nair S, Prasada Rao U, Bennett J, Mohan M.1995. Detection of a highly heterozygous locus in recombinant inbred lines of rice and its possible involvement in heterosis.Theor Appl Genet, 91: 978-986.
|
| [46] |
Ndjiondjop M N, Manneh B, Cissoko M, Drame N K, Kakai R G, Bocco R, Baimey H, Wopereis M.2010. Drought resistance in an interspecific backcross population of rice (Oryza spp.) derived from the cross WAB56-104(O. sativa) × CG14, 179(4): 364-373.
|
| [47] |
Nguyen B D.2001. Molecular mapping of aluminum tolerance in rice. Texas: Texas Technology University.
|
| [48] |
Pritchard J K, Stephens M, Donnelly P.2000. Inference of population structure using multi locus genotype data.Genetics, 155(2): 945-959.
|
| [49] |
Rai V, Sreenu K, Pushpalatha B, Babu, Brajendra A P, Sandhya G, Sarla N.2010. Swarna/Oryza nivara and KMR3/O. rufipogon introgression lines tolerant to drought and salinity. DRR News Lett, 8: 4.
|
| [50] |
Rangel P N, Brondani R P V, Rangel P H N, Brondani C.2008. Agronomic and molecular characterization of introgression lines from the interspecific cross Oryza sativa BG90-2 × Oryza glumaepatula RS-16. Genet Mol Res, 7(1): 184-195.
|
| [51] |
Rangel P N, Vianello R P, Melo A T O, Rangel P H N, Mendonça J A, Brondani C.2013. Yield QTL analysis of Oryza sativa × O. glumaepatula introgression lines. Pesq Agropec Bras, 48(3): 280-286.
|
| [52] |
Russell P J, Hertz P E, McMillan B.2013. Biology: The Dynamics of Science. Canada: Yolanda Cossio.
|
| [53] |
Sang X C, Li Y F, Luo Z K, Ren D Y, Fang L K, Wang N, Zhao F M, Ling Y H, Yang Z L, Liu Y S, He G H.2012. CHIMERIC FLORAL ORGANS1, encoding a monocot-specific MADS box protein, regulates floral organ identity in rice. Plant Physiol, 160(2): 788-807.
|
| [54] |
Sarao P S, Sahi G K, Neelam K, Mangat G S, Patra B C, Singh K.2016. Donors for resistance to brown planthopper Nilaparvata lugens(Stål) from wild rice species. Rice Sci, 23(4): 219-224.
|
| [55] |
Sarla N, Bobba S, Siddiq E A.2003. ISSR and SSR markers based on AG and GA repeats delineate geographically diverse Oryza nivara accessions and reveal rare alleles. Curr Sci, 84: 683-690.
|
| [56] |
Sarla N, Swamy B P M, Sudhakar T, Babu A P, Kaladhar K, Reddy C S, Reddy G A, Ramesha M S, Rani N S, Viraktamath B C.2009. High yielding rice lines from elite × wild crosses.DRR News Lett, 7: 9.
|
| [57] |
Sharma S, Upadhyaya H D, Varshney R K, Gowda C L L.2013. Pre-breeding for diversification of primary gene pool and genetic enhancement of grain legumes.Front Plant Sci, 4: 309.
|
| [58] |
Surapaneni M, Balakrishnan D, Mesapogu S, Addanki K R, Yadavalli V R, Tripura Venkata V G N, Neelamraju S.2017. Identification of major effect QTLs for agronomic traits and CSSLs in rice from Swarna/ Oryza nivara derived backcross inbred lines. Front Plant Sci, 8: 1027.
|
| [59] |
Swamy B P M, Sarla N.2008. Yield-enhancing quantitative trait loci (QTLs) from wild species.Biotechnol Adv, 26(1): 106-120.
|
| [60] |
Swamy B P M.2009. Genome wide mapping of quantitative trait loci (QTLs) for yield and grain quality traits in O. sativa cv Swarna × O. nivara backcross population. Hyderabad: Osmania University.
|
| [61] |
Swamy B P M, Kaladhar K, Ramesha M S, Viraktamath B C, Sarla N.2011. Molecular mapping of QTLs for yield and yield-related traits in Oryza sativa cv Swarna × O. nivara (IRGC81848) backcross population. Rice Sci, 18(3): 178-186.
|
| [62] |
Swamy B P M, Kaladhar K, Shobharani N, Prasad G S V, Viraktamath B C, Reddy G A, Sarla N.2012. QTL analysis for grain quality traits in 2 BC2F2 populations derived from crosses between Oryza sativa cv Swarna and 2 accessions of O. nivara. J Hered, 103(3): 442-452.
|
| [63] |
Swamy B P M, Kaladhar K, Reddy G A, Viraktamath B C, Sarla N.2014. Mapping and introgression of QTL for yield and related traits in two backcross populations derived fromOryza sativa cv. Swarna and two accessions of O. nivara. J Genet, 93(3): 643-654.
|
| [64] |
Thalapati S, Guttikonda H, Nannapaneni N D, Adari P B, Reddy C S, Swamy B P M, Batchu A K, Basava R K, Viraktamath B C, Neelamraju S.2015. Heterosis and combining ability in rice as influenced by introgressions from wild species Oryza rufipogon including qyld2.1 sub-QTL into the restorer line KMR3. Euphytica, 202(1): 81-95.
|
| [65] |
Thomson M J, Tai T H, McClung A M, Lai X H, Hinga M E, Lobos K B, Xu Y, Martinez C P, McCouch S R.2003. Mapping quantitative trait loci for yield, yield components and morphological traits in an advanced backcross population between Oryza rufipogon and the Oryza sativa cultivar Jefferson. Theor Appl Genet, 107(3): 479-493.
|
| [66] |
Tian F, Li D J, Fu Q, Zhu Z F, Fu Y C, Wang X K, Sun C Q.2006. Construction of introgression lines carrying wild rice (Oryza rufipogon Griff.) segments in cultivated rice(Oryza sativa L.) background and characterization of introgressed segments associated with yield-related traits. Theor Appl Genet, 112(3): 570-580.
|
| [67] |
van Berloo R.2008. GGT 2.0: Versatile software for validation and analysis of genetic data.J Hered, 99(2): 232-236.
|
| [68] |
Verma S K, Saxena R R, Saxena R R, Xalxo M S, Verulkar S B.2014. QTL for grain yield under water stress and non-stress conditions over years in rice (Oryza sativa L.). AJCS, 8(6): 916-926.
|
| [69] |
Wang Y M, Dong Z Y, Zhang Z J, Lin X Y, Shen Y, Zhou D W, Liu B.2005. Extensive de novo genomic variation in rice induced by introgression from wild rice(Zizania latifolia Griseb.). Genetics, 170(4): 1945-1956.
|
| [70] |
Weng X Y, Wang L, Wang J, Hu Y, Du H, Xu C G, Xing Y Z, Li X H, Xiao J H, Zhang Q F.2014. Grain number, plant height, and heading date 7 is a central regulator of growth, development, and stress response.Plant Physiol, 164(2): 735-747.
|
| [71] |
Xiao J, Li J, Grandillo S, Ahn S N, Yuan L, Tanksley S D, McCouch S R.1998. Identification of trait-improving quantitative trait loci alleles from a wild rice relative,Oryza rufipogon. Genetics, 150(2): 899-909.
|
| [72] |
Xu B.2007. Genetic analysis and gene mapping research of dwarf tillering mutant in indica rice. Fujian, China: Xiamen University.
|
| [73] |
Yadaw R B, Dixit S, Raman A, Mishra K K, Vikram P, Swamy B P M, Sta Cruz M T, Maturan P T, Pandey M, Kumar A.2013. A QTL for high grain yield under lowland drought in the background of popular rice variety Sabitri from Nepal.Field Crops Res, 144: 281-287.
|
| [74] |
Yang C W, Zhang T Y, Wang H, Zhao N, Liu B.2012. Heritable alteration in salt-tolerance in rice induced by introgression from wild rice (Zizania latifolia). Rice, 5: 1-2.
|
| [75] |
Yao M Z, Wang J F, Chen H Y, Zhai H Q, Zhang H S.2005. Inheritance and QTL mapping of salt tolerance in rice.Rice Sci, 12(1): 25-32.
|
| [76] |
Ye H Y, Du H, Tang N, Li X H, Xiong L Z.2009. Identification and expression profiling analysis of TIFY family genes involved in stress and phytohormone responses in rice.Plant Mol Biol, 71(3): 291-305.
|
| [77] |
Zhang Z H, Qu X S, Wan S, Chen L H, Zhu Y G.2005. Comparison of QTL controlling seedling vigour under different temperature conditions using recombinant inbred lines in rice (Oryza sativa). Ann Bot, 95(3): 423-429.
|
| [78] |
Zheng K L, Subudhi P K, Domingo J, Magpantay G, Huang N.1995. Rapid DNA isolation for marker assisted selection in rice breeding.Rice Genet Newsl, 12: 255-258.
|