
Rice Science ›› 2021, Vol. 28 ›› Issue (5): 493-500.DOI: 10.1016/j.rsci.2021.07.009
收稿日期:2020-08-14
接受日期:2021-01-08
出版日期:2021-09-28
发布日期:2021-09-28
. [J]. Rice Science, 2021, 28(5): 493-500.
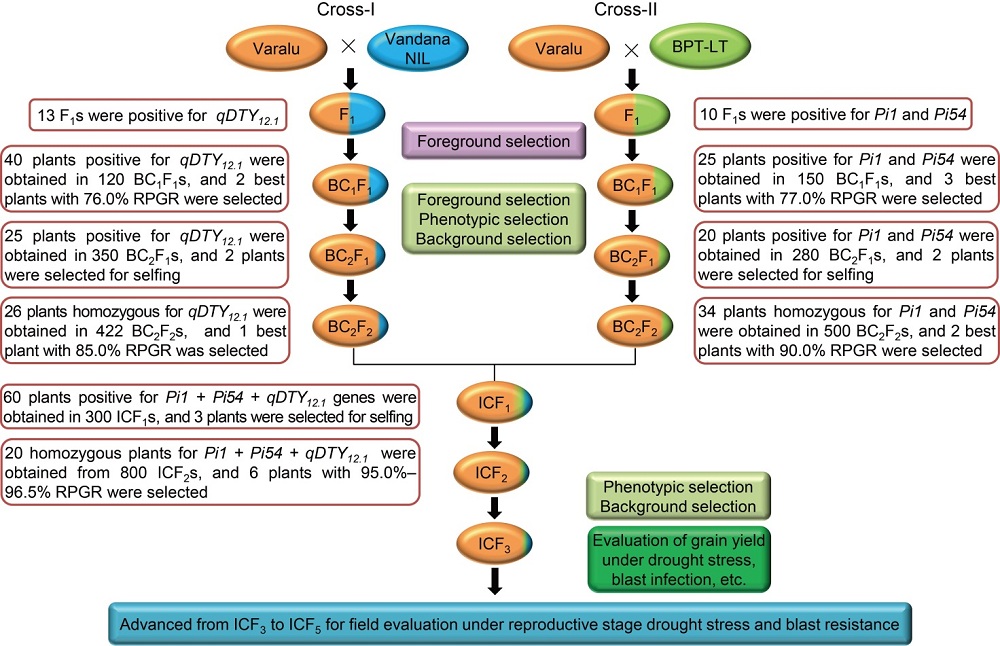
Fig. 1. Schematic representation of marker-assisted gene pyramiding followed for introgression of qDTY12.1 for improvement of grain yield under reproductive stage drought stress and blast resistance genes (Pi54 and Pi1) in Varalu variety.NIL, Near-isogenic line; BPT-LT, Elite rice line with the genetic background of Samba Mahsuri containing two blast resistance genes Pi54 and Pi1; RPGR, Recurrent parent genome recovery; IC, Intercross.
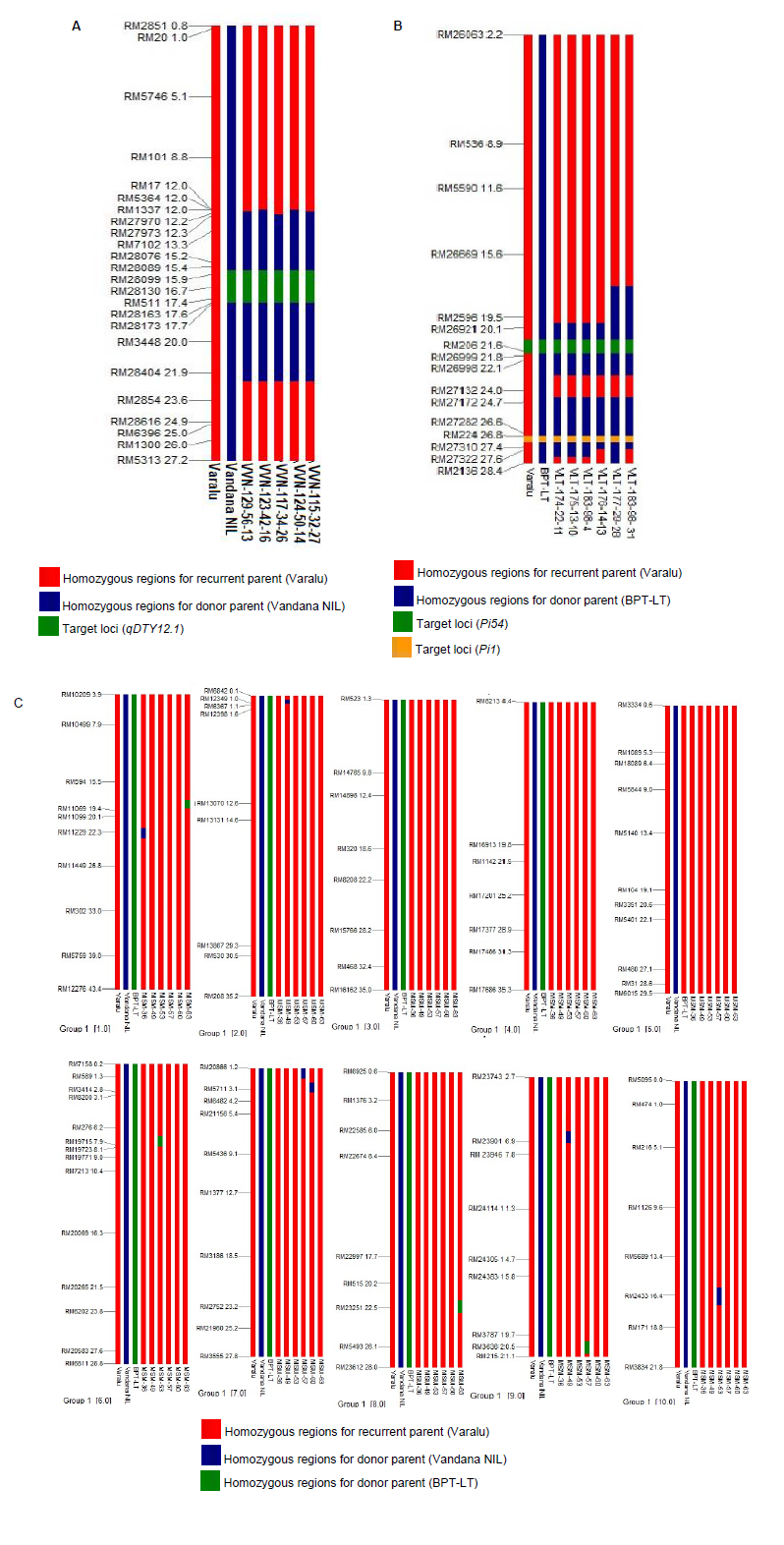
Fig. S1. Graphical representation of BC2F2 selected lines.A, Graphical representation of BC2F2 selected lines of cross-I (Varalu × Vandana NIL) for the donor genome introgression associated with the qDTY12.1 on target chromosome 12. Red colour indicates homozygous regions for recurrent parent Varalu, blue colour indicates the donor parents (BPT-LT) and green colour indicates qDTY12.1. =B, Graphical representation of BC2F2 selected lines of cross-II (Varalu × BPT-LT) for the donor genome introgression associated with the blast resistance genes, Pi54 and Pi1 on target chromosome 11. Red colour indicates homozygous regions for recurrent parent Varalu, blue colour indicates the donor parents (BPT-LT), green colour indicates Pi54 gene and black colour indicates Pi1 gene. C, Graphical representation of pyramided lines for the donor genome introgression on non-target chromosomes 1?10. Red colour indicates homozygous regions for recurrent parent Varalu and the green and blue colours indicate the donor parents (BPT-LT and Vandana NIL) genome region, respectively.
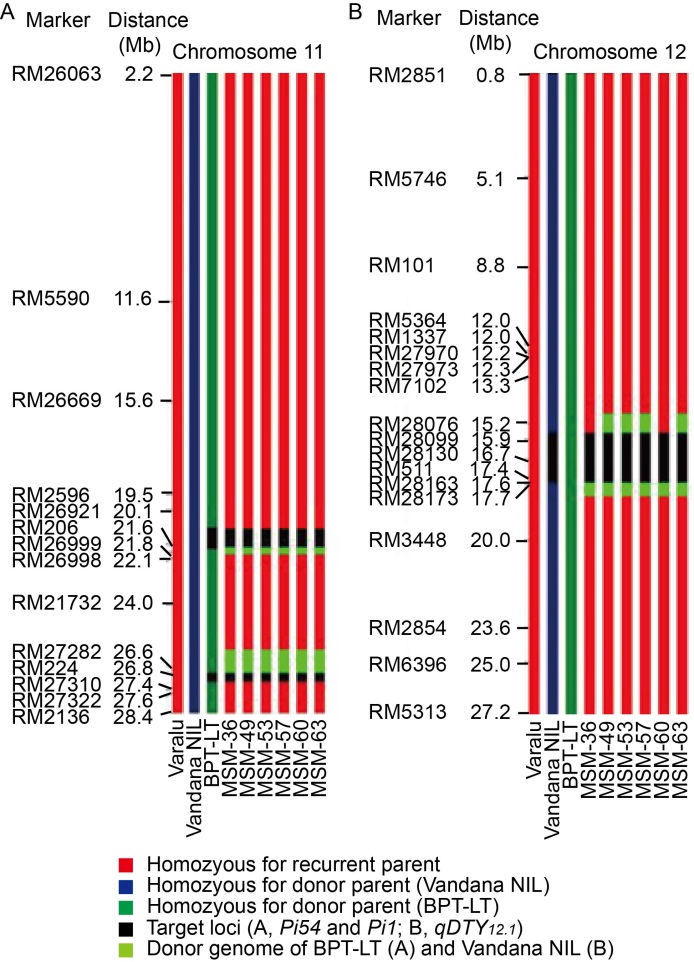
Fig. 2. Graphical representation of selected pyramided lines of Varalu for donor genome introgression associated with blast resistance genes Pi54 and Pi1 on chromosome 11 and qDTY12.1 on chromosome 12.A, At Pi54 locus, a donor segment introgression was limited to only about 0.3 Mb at the proximal end; at Pi1 locus, about 0.2 Mb donor genome was observed at the proximal end. B, At qDTY12.1 locus, a donor genomic region about 0.7 and 0.1 Mb at the proximal and distal ends, respectively, was observed in four lines on chromosome 12. Two lines (MSM-36 and MSM-60) showed limited donor segments in comparison with other lines.NIL, Near-isogenic line; BPT-LT, Elite rice line with the genetic background of Samba Mahsuri containing two blast resistance genes Pi54 and Pi1.

Fig. S2. Phenotypes of improved lines.A, Representation of single plants of improved lines (Pi54 + Pi1 + qDTY12.1) and six selected improved lines of Varalu, showing superior performance in comparison with the recurrent parent under drought conditions. B, Panicles of the recurrent parent and selected improved lines of Varalu. C, Grain type of selected lines showing similarity with the recurrent parent.

Fig. S3. Blast phenotypic screening of selected BC2F2 lines of cross-II (Varalu × BPT-LT) and their parents. P1, VLT-174-22-11; P2, VLT-175-13-10; P3, VLT-176-14-13; P4, VLT-183-98-4; P5, VLT-177-29-28; P6, VLT-183-98-31 and donor parent BPT-LT showed resistance whereas recurrent parent, Varalu showed susceptible reaction for blast.

Fig. 3. Phenotypic screening of pyramided lines (Pi54 + Pi1 + qDTY12.1) against blast disease. A, All the intercross derived lines and donor parent BPT-LT (an elite rice line with the genetic background of Samba Mahsuri containing two blast resistance genes Pi54 and Pi1) were highly resistant whereas the recurrent parent Varalu showed susceptible against blast. B, Lesions were observed on the leaf surface of the recurrent parent Varalu while the pyramided lines and donor parent showed no lesion on the leaf surface.
| Parameter | Drought condition | Control condition | |||
|---|---|---|---|---|---|
| Varalu | MSM-36 (IET26753) | Varalu | MSM-36 (IET26753) | ||
| Yield advantage of MSM-36 over Varalu (%) | 18.80 | 13.80 | |||
| Grain yield (kg/hm2) | 4293 | 5098 | 4747 | 5404 | |
| Blast severity index | 6.2 | 2.95 | |||
| Days to 50% flowering (d) | 91 | 93 | 95 | 96 | |
| No. of panicles per m2 | 389 | 391 | 323 | 343 | |
| Plant height (cm) | 82 | 85 | 78 | 76 | |
| Varalu | MSM-36 (IET26753) | ||||
| Hulling rate (%) | 77.9 | 78.7 | |||
| Milling rate (%) | 67.7 | 68.2 | |||
| Head rice recovery (%) | 60.0 | 58.6 | |||
| Grain length (mm) | 6.13 | 6.17 | |||
| Grain width (mm) | 2.04 | 2.04 | |||
| Ratio of grain length to width | 3.04 | 3.02 | |||
| Grain type | LS | LS | |||
| Grain chalkiness | Very occasionally | Very occasionally | |||
| Alkali spreading value | 7 | 7 | |||
| Amylose content (%) | 24.11 | 26.22 | |||
| Gel consistency | 22 | 25 | |||
Table S2 Performance of best pyramided lines under drought and controlled conditions in national trials (Zone VII).
| Parameter | Drought condition | Control condition | |||
|---|---|---|---|---|---|
| Varalu | MSM-36 (IET26753) | Varalu | MSM-36 (IET26753) | ||
| Yield advantage of MSM-36 over Varalu (%) | 18.80 | 13.80 | |||
| Grain yield (kg/hm2) | 4293 | 5098 | 4747 | 5404 | |
| Blast severity index | 6.2 | 2.95 | |||
| Days to 50% flowering (d) | 91 | 93 | 95 | 96 | |
| No. of panicles per m2 | 389 | 391 | 323 | 343 | |
| Plant height (cm) | 82 | 85 | 78 | 76 | |
| Varalu | MSM-36 (IET26753) | ||||
| Hulling rate (%) | 77.9 | 78.7 | |||
| Milling rate (%) | 67.7 | 68.2 | |||
| Head rice recovery (%) | 60.0 | 58.6 | |||
| Grain length (mm) | 6.13 | 6.17 | |||
| Grain width (mm) | 2.04 | 2.04 | |||
| Ratio of grain length to width | 3.04 | 3.02 | |||
| Grain type | LS | LS | |||
| Grain chalkiness | Very occasionally | Very occasionally | |||
| Alkali spreading value | 7 | 7 | |||
| Amylose content (%) | 24.11 | 26.22 | |||
| Gel consistency | 22 | 25 | |||
| Variety/Line | Hulling rate (%) | Milling rate (%) | HRR (%) | GL (mm) | GW (mm) | GL/GW | Grain type | Grain chalkiness | ASV | AC (%) | GC (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Varalu | 77.9 | 68.9 | 57.3 | 6.13 | 2.04 | 3.04 | LS | VOC | 7 | 25.51 | 28 |
| Vandana NIL | 66.7 | 59.3 | 52.4 | 5.90 | 2.20 | 2.68 | MB | VOC | 3 | 23.12 | 63 |
| BPT-LT | 79.6 | 71.2 | 68.5 | 5.40 | 2.10 | 2.57 | MB | VOC | 4 | 27.61 | 61 |
| MSM-36 | 78.9 | 69.5 | 59.8 | 6.12 | 2.05 | 3.00 | LS | VOC | 7 | 26.79 | 27 |
| MSM-49 | 77.9 | 69.8 | 58.2 | 6.04 | 2.01 | 3.00 | LS | VOC | 4 | 25.34 | 28 |
| MSM-53 | 77.5 | 68.5 | 58.3 | 6.10 | 2.02 | 3.02 | LS | VOC | 6 | 26.38 | 26 |
| MSM-57 | 78.7 | 68.4 | 59.2 | 6.12 | 2.02 | 3.04 | LS | VOC | 7 | 26.18 | 28 |
| MSM-60 | 78.2 | 69.4 | 58.7 | 6.17 | 2.03 | 3.04 | LS | VOC | 7 | 25.88 | 29 |
| MSM-63 | 78.4 | 68.6 | 59.7 | 6.11 | 2.03 | 3.01 | LS | VOC | 6 | 26.58 | 29 |
Table 2 Grain and cooking quality of selected pyramided lines of Varalu.
| Variety/Line | Hulling rate (%) | Milling rate (%) | HRR (%) | GL (mm) | GW (mm) | GL/GW | Grain type | Grain chalkiness | ASV | AC (%) | GC (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Varalu | 77.9 | 68.9 | 57.3 | 6.13 | 2.04 | 3.04 | LS | VOC | 7 | 25.51 | 28 |
| Vandana NIL | 66.7 | 59.3 | 52.4 | 5.90 | 2.20 | 2.68 | MB | VOC | 3 | 23.12 | 63 |
| BPT-LT | 79.6 | 71.2 | 68.5 | 5.40 | 2.10 | 2.57 | MB | VOC | 4 | 27.61 | 61 |
| MSM-36 | 78.9 | 69.5 | 59.8 | 6.12 | 2.05 | 3.00 | LS | VOC | 7 | 26.79 | 27 |
| MSM-49 | 77.9 | 69.8 | 58.2 | 6.04 | 2.01 | 3.00 | LS | VOC | 4 | 25.34 | 28 |
| MSM-53 | 77.5 | 68.5 | 58.3 | 6.10 | 2.02 | 3.02 | LS | VOC | 6 | 26.38 | 26 |
| MSM-57 | 78.7 | 68.4 | 59.2 | 6.12 | 2.02 | 3.04 | LS | VOC | 7 | 26.18 | 28 |
| MSM-60 | 78.2 | 69.4 | 58.7 | 6.17 | 2.03 | 3.04 | LS | VOC | 7 | 25.88 | 29 |
| MSM-63 | 78.4 | 68.6 | 59.7 | 6.11 | 2.03 | 3.01 | LS | VOC | 6 | 26.58 | 29 |
| Gene | Marker | Chr. | Position (Mb) | Primer | Anneling temperature (ºC) | Expected product size (bp) | Reference | |
|---|---|---|---|---|---|---|---|---|
| DP | RP | |||||||
| Pi54 | RM206 | 11 | 22.01 | F: 5′CCCATGCGTTTAACTATTCT3′ R: 5′CGTTCCATCGATCCGTATGG3′ | 55 | 147 | 165 | Patroti et al, 2019 |
| Pi1 | RM224 | 11 | 27.67 | F: 5′CTCGATCGATCTTCACGAGG3′ R:5′TGCTATAAAAGGCATTCGGG3′ | 55 | 157 | 175 | Patroti et al, 2019 |
| qDTY12.1 | RM28099 | 12 | 15.84 | F: 5′TGTGCGGATGCGGGTAAGTCC3′ R: 5′CCACCTGTCAACCACCGAAACC3′ | 55 | 120 | 130 | Dixit et al, 2014 |
| RM28130 | 12 | 16.7 | F: 5′CAGCAGACGTTCCGGTTCTACTCG3′ R: 5′AGGACGGTGGTGGTGATCTGG3′ | 55 | 175 | 155 | Dixit et al, 2014 | |
| RM511 | 12 | 17.39 | F: 5′CTTCGATCCGGTGACGAC3′ R: 5′AACGAAAGCGAAGCTGTCTC3′ | 55 | 130 | 145 | Bernier et al, 2007 | |
| RM28163 | 12 | 17.53 | F: 5′GTCCATGCCCAAGACACAAC3′ R: 5′GTTACATCATGGGTGACCCC3′ | 55 | 167 | 175 | Dixit et al, 2014 | |
Table S1 Details of SSR markers used for foreground selection.
| Gene | Marker | Chr. | Position (Mb) | Primer | Anneling temperature (ºC) | Expected product size (bp) | Reference | |
|---|---|---|---|---|---|---|---|---|
| DP | RP | |||||||
| Pi54 | RM206 | 11 | 22.01 | F: 5′CCCATGCGTTTAACTATTCT3′ R: 5′CGTTCCATCGATCCGTATGG3′ | 55 | 147 | 165 | Patroti et al, 2019 |
| Pi1 | RM224 | 11 | 27.67 | F: 5′CTCGATCGATCTTCACGAGG3′ R:5′TGCTATAAAAGGCATTCGGG3′ | 55 | 157 | 175 | Patroti et al, 2019 |
| qDTY12.1 | RM28099 | 12 | 15.84 | F: 5′TGTGCGGATGCGGGTAAGTCC3′ R: 5′CCACCTGTCAACCACCGAAACC3′ | 55 | 120 | 130 | Dixit et al, 2014 |
| RM28130 | 12 | 16.7 | F: 5′CAGCAGACGTTCCGGTTCTACTCG3′ R: 5′AGGACGGTGGTGGTGATCTGG3′ | 55 | 175 | 155 | Dixit et al, 2014 | |
| RM511 | 12 | 17.39 | F: 5′CTTCGATCCGGTGACGAC3′ R: 5′AACGAAAGCGAAGCTGTCTC3′ | 55 | 130 | 145 | Bernier et al, 2007 | |
| RM28163 | 12 | 17.53 | F: 5′GTCCATGCCCAAGACACAAC3′ R: 5′GTTACATCATGGGTGACCCC3′ | 55 | 167 | 175 | Dixit et al, 2014 | |
| [1] | Anyaoha C O, Fofana M, Gracen V, Tongoona P, Mande S. 2019. Introgression of two drought QTLs into FUNAABOR-2 early generation backcross progenies under drought stress at reproductive stage. Rice Sci, 26(1): 32‒41. |
| [2] | Asibi A E, Chai Q, Coulter J A. 2019. Rice blast: A disease with implications for global food security. Agronomy, 9(8): 451. |
| [3] | Bernier J, Kumar A, Venuprasad R, Spaner D, Atlin G N. 2007. A large-effect QTL for grain yield under reproductive-stage drought stress in upland rice. Crop Sci, 47(2): 507‒516. |
| [4] | Bernier J, Serraj R, Kumar A, Venuprasad R, Impa S, Gowda V R, Oane R, Spaner D, Atlin G. 2009a. The large-effect drought- resistance QTL qtl12.1 increases water uptake in upland rice. Field Crops Res, 110: 139‒146. |
| [5] | Bernier J, Kumar A, Venuprasad R, Spaner D, Verlukar S, Mandal N P, Sinha P K, Peeraju P, Dongre P R, Mahto R N, Atlin G. 2009b. Characterization of the effect of a QTL for drought resistance in rice, qtl12.1 over a range of environments in the Philippines and eastern India. Euphytica, 166(2): 207‒217. |
| [6] | Cagampang G B, Perez C M, Juliano B O. 1973. A gel consistency test for eating quality of rice. J Sci Food Agric, 24(12): 1589‒1594. |
| [7] | Chukwu S C, Rafii Y M, Ramlee S I, Ismail S I, Oladosu Y, Okporie E, Onyishi G, Utobo E, Ekwu L, Swaray S, Jalloh M. 2019. Marker-assisted selection and gene pyramiding for resistance to bacterial leaf blight disease of rice (Oryza sativa L.). Biotechnol Biotechnol Equip, 33(1): 440-455. |
| [8] | Dean R, van Kan J A L, Pretorius Z A, Hammond-Kosack K E, Di Pietro A, Spanu P D, Rudd J J, Dickman M, Kahmann R, Ellis J, Foster G D. 2012. The Top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol, 13(4): 414-430. |
| [9] | Devi S J S R, Kuldeep S, Umakanth B, Vishalakshi B, Renuka P, Sudhakar K V, Prasad M S, Viraktamath B C, Ravindrababu V, Madhav M S. 2015. Development and identification of novel rice blast resistant sources and their characterization using molecular markers. Rice Sci, 22(6): 300-308. |
| [10] | Devi S J S R, Singh K, Umakanth B, Vishalakshi B, Rao K V S, Suneel B, Sharma S K, Kadambari G K M, Prasad M S, Senguttvel P, Divya P S, Madhav M S. 2020. Identification and characterization of a large effect QTL fromOryza glumaepatula revealed Pi68(t) as putative candidate gene for rice blast resistance. Rice, 13: 17. |
| [11] | Dixit S, Swamy B M, Vikram P, Ahmed H U, Cruz M S, Amante M, Atri D, Leung H, Kumar A. 2012. Fine mapping of QTLs for rice grain yield under drought reveals sub-QTLs conferring a response to variable drought severities. Theor Appl Genet, 125(1): 155-169. |
| [12] | Dixit S, Huang B E, Sta Cruz M T, Maturan P T, Ontoy J C E, Kumar A. 2014. QTLs for tolerance of drought and breeding for tolerance of abiotic and biotic stress: An integrated approach. PLoS One, 9(10): e109574. |
| [13] | Dixit S, Singh U M, Singh A K, Alam S, Venkateshwarlu C, Nachimuthu V V, Yadav S, Abbai R, Selvaraj R, Devi M N, Ramayya P J, Badri J, Ram T, Lakshmi J, Lakshmidevi G, Vidhya L R K J, Padmakumari A P, Laha G S, Prasad M S, Seetalam M, Singh V K, Kumar A. 2020. Marker assisted forward breeding to combine multiple biotic-abiotic stress resistance/tolerance in rice. Rice, 13: 29. |
| [14] | IRRI. 2002. Standard Evaluation System for Rice. Los Banos, Manila, the Philippine: International Rice Research Institute. |
| [15] | IRRI. 2004. Standard Evaluation System for Rice. Los Banos, Manila, the Philippine: International Rice Research Institute. |
| [16] | Jamaloddin M, Durga Rani C V, Swathi G, Anuradha C, Vanisri S, Rajan C P D, Krishnam Raju S, Bhuvaneshwari V, Jagadeeswar R, Laha G S, Prasad M S, Satyanarayana P V, Cheralu C, Rajani G, Ramprasad E, Sravanthi P, Arun Prem Kumar N, Aruna Kumari K, Yamini K N, Mahesh D, Sanjeev Rao D, Sundaram R M, Sheshu Madhav M. 2020. Marker assisted gene pyramiding (MAGP) for bacterial blight and blast resistance into mega rice variety ‘Tellahamsa’. PLoS One, 15(6): e0234088. |
| [17] | Juliano B O. 1971. A simplified assay for milled rice amylose. Cereal Sci Today, 16(11): 334-360. |
| [18] | Juliano B O, Perez C M, Kaushik R, Khush G S. 1990. Some grain properties of IR36-based starch mutants. Starch, 42(7): 256-260. |
| [19] | Khan G H, Shikari A B, Vaishnavi R, Najeeb S, Padder B A, Bhat Z A, Parray G A, Bhat A M, Kumar R, Singh N K. 2018. Marker-assisted introgression of three dominant blast resistance genes into an aromatic rice cultivar Mushk Budji. Sci Rep, 8(1): 4091. |
| [20] | Kumar A, Dixit S, Ram T, Yadaw R B, Mishra K K, Mandal N P. 2014. Breeding high-yielding drought-tolerant rice: Genetic variations and conventional and molecular approaches. J Exp Bot, 65(21): 6265-6278. |
| [21] | Kumar R, Venuprasad R, Atlin G N. 2007. Genetic analysis of rainfed lowland rice drought tolerance under naturally occurring stress in eastern India: Heritability and QTL effects. Field Crops Res, 103(1): 42-52. |
| [22] | Mackill D J, Bonman J M. 1992. Inheritance of blast resistance in near isogenic lines of rice. Phytopathology, 82(7): 746-749. |
| [23] | Mishra K K, Vikram P, Yadaw R B, Swamy B P M, Dixit S, Staruz M T G, Marker S, Kumar A. 2013. qDTY12.1: A locus with a consistent effect on grain yield under drought in rice. BMC Genet, 14: 12. |
| [24] | Murray H G, Thompson W F. 1980. Rapid isolation of high molecular weight DNA. Nucl Acids Res, 8(19): 4321-4325. |
| [25] | Muthu V, Abbai R, Nallathambi J, Rahman H, Ramasamy S, Kambale R, Thulasinathan T, Ayyenar B, Muthurajan R. 2020. Pyramiding QTLs controlling tolerance against drought, salinity, and submergence in rice through marker assisted breeding. PLoS One, 15(1): e0227421. |
| [26] | Patroti P, Vishalakshi B, Umakanth B, Suresh J, Senguttuvel P, Madhav M S. 2019. Marker-assisted pyramiding of major blast resistance genes in Swarna-Sub1, an elite rice variety (Oryza sativa L.). Euphytica, 215(11): 179. |
| [27] | Serraj R, McNally K L, Slamet-Loedin I, Kohli A, Haefele S M, Atlin G, Kumar A. 2011. Drought resistance improvement in rice: An integrated genetic and resource management strategy. Plant Prod Sci, 14(1): 1-14. |
| [28] | Sharma T R, Madhav M S, Singh B K, Shanker P, Jana T K, Dalal V, Pandit A, Singh A, Gaikwad K, Upreti H C, Singh N K. 2005. High-resolution mapping, cloning and molecular characterization of thePi-kh gene of rice, which confers resistance to Magnaporthe grisea. Mol Genet Genomics, 274(6): 569-578. |
| [29] | Swathi G, Durga Rani C V, Jamaloddin M, Sheshu Madhav M, Vanisree S, Anuradha C, Ranjit Kumar N, Aruna Prem Kumar N, Aruna Kumari K, Bhogadhi S C, Ramprasad E, Sravanthi P, Krishinam Raju S, Bhuvaneswari V, Rajan C P D, Jagadeeswar R. 2019. Marker-assisted introgression of the major bacterial blight resistance genes,Xa21 and xa13, and blast resistance gene, Pi54, into the popular rice variety, JGL1798. Mol Breeding, 39: 58. |
| [30] | Venuprasad R, Lafitte H R, Atlin G N. 2007. Response to direct selection for grain yield under drought stress in rice. Crop Sci, 47: 285-293. |
| [31] | Vikram P, Swamy B P M, Dixit S, Ahmed H U, Sta Cruz M T, Singh A K, Kumar A. 2011. qDTY1.1, a major QTL for rice grain yield under reproductive-stage drought stress with a consistent effect in multiple elite genetic backgrounds. BMC Genet, 12: 89. |
| [32] | Wassmann R, Jagadish S V K, Sumfleth K, Pathak H, Howell G, Ismail A, Serraj R, Redona E, Singh R K, Heuer S. 2009. Regional vulnerability of climate change impacts on Asian rice production and scope for adaptation. Adv Agron, 102: 91‒133. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||