
Rice Science ›› 2021, Vol. 28 ›› Issue (6): 547-556.DOI: 10.1016/j.rsci.2021.09.003
收稿日期:2020-07-01
接受日期:2020-10-12
出版日期:2021-11-28
发布日期:2021-11-28
. [J]. Rice Science, 2021, 28(6): 547-556.
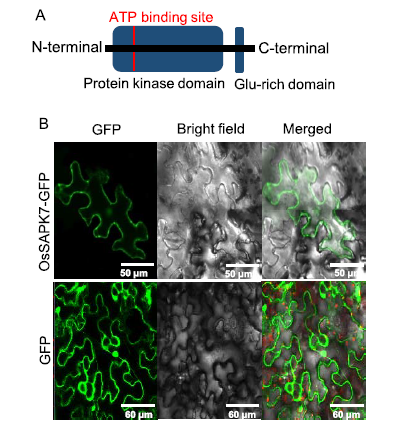
Fig. 1. Domain structure and subcellular localization of OsSAPK7.A, Schematic representation of the domain structure of OsSAPK7. The highly conserved kinase region at the N-terminal and the Glu-rich motif at the C-terminal are shown.B, Green fluorescence protein (GFP) signals arising from the OsSAPK7- GFP fusion proteins expressed in tobacco leaves were detected using a confocal microscope. Red signals represent chloroplast auto-fluorescence.The only GFP signal expressed by pCAMBIA-1300 in tobacco leaves served as a negative control.
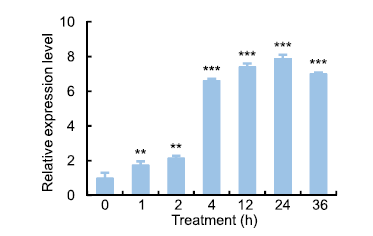
Fig. 2. Expression of OsSAPK7 in rice under salt-stress conditions.After 20-day-old rice seedlings were subjected to 100 mmol/L NaCl treatment, OsSAPK7 expression levels were assessed by qRT-PCR. Means and standard errors were obtained from three biological replicates. ** and *** indicate statistically significant differences at the 0.01 and 0.001 levels, respectively.
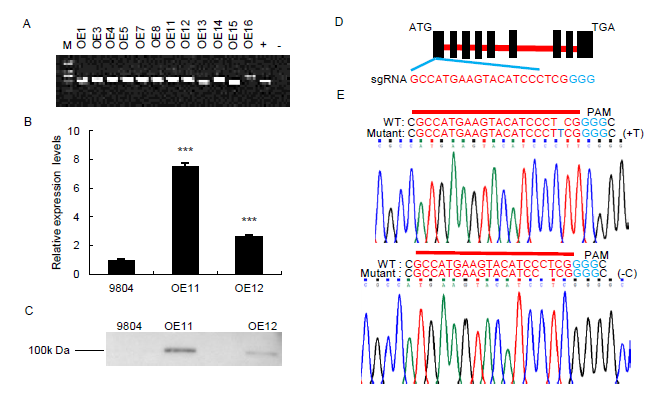
Fig. S1. Graphic representation of vector constructs and expression analysis of OsSAPK7 in transgenic plants.A, Positive OsSAPK7-overexpression transgenic plants was verified by PCR method. M, marker; +, positive; -, negative.B, Expression of OsSAPK7 in wild type 9804 and OsSAPK7-overexpression transgenic plants as determined by qRT-PCR. Actin gene was the control. Values are Mean ± SE (n = 3). ***, P < 0.001 according to the Student’s t-test.C, Protein expression of OsSAPK7 in 9804 and OsSAPK7-overexpressing transgenic plants as assessed by western blotting.D, A schematic description of the WT OsSAPK7 gene. Red letters represent the target sequence.E, Sanger sequencing results of mutations in OsSAPK7 knockouts. Above is the editing sequence of the target site, and below is the sequencing chromatogram.
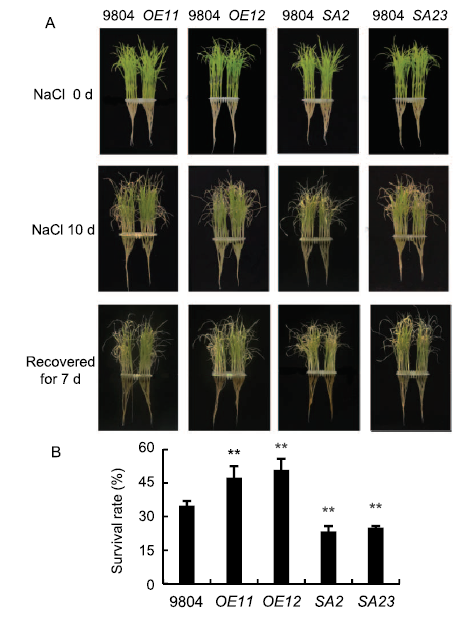
Fig. 3. Phenotypic reactions of OsSAPK7-overexpression, OsSAPK7 knockout and wild type (WT) 9804 lines under salt-stress conditions.A, Phenotypic comparison of seedings grown under salt stress at the seedling stage. OsSAPK7-overexpression lines (OE11 and OE12), OsSAPK7 knockout lines (SA2 and SA23) and 9804 plants under normal conditions for 20-day-old seedlings were transferred to Hoagland's nutrient solution supplemented with 100 mmol/L NaCl for 10 d and then recovered for 7 d.B, Survival rates of OE11, OE12, SA2, SA23 and 9804 plants after recovered for 7 d. Values are Mean ± SE (n = 3). **, P < 0.05 according to the Student's t-test.
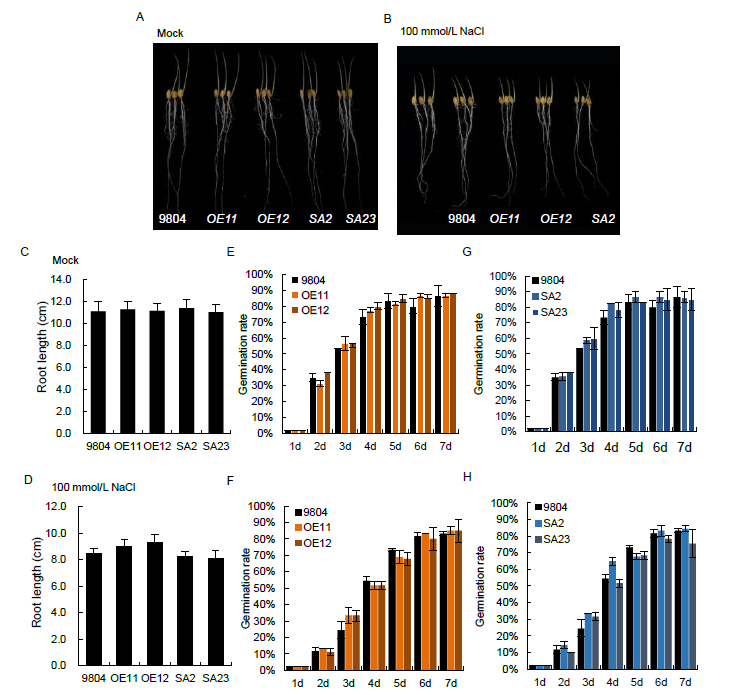
Fig. S2. Root growth and germination rates of OsSAPK7 transgenic lines and wild type 9804 under salt-stress conditions.A and B, Phenotype of OsSAPK7-overexpression (OE11 and OE12), OsSAPK7 knockout (SA2 and SA23) and wild type 9804 plants grown in distilled water (A) or in distilled water supplemented with 100 mmol/L NaCl solution (B).C and D, Root lengths of OsSAPK7-overexpression, OsSAPK7 knockout and 9804 plants grown in distilled water (C) or in distilled water supplemented with 100 mmol/L NaCl solution (D).E to H, Seed germination rates of OsSAPK7-overexpression, OsSAPK7 knockout and wild type 9804 plants grown in distilled water or in distilled water supplemented with 100 mmol/L NaCl solution.Values are Mean ± SE (n = 3). *, P < 0.05 according to the Student’s t-test.
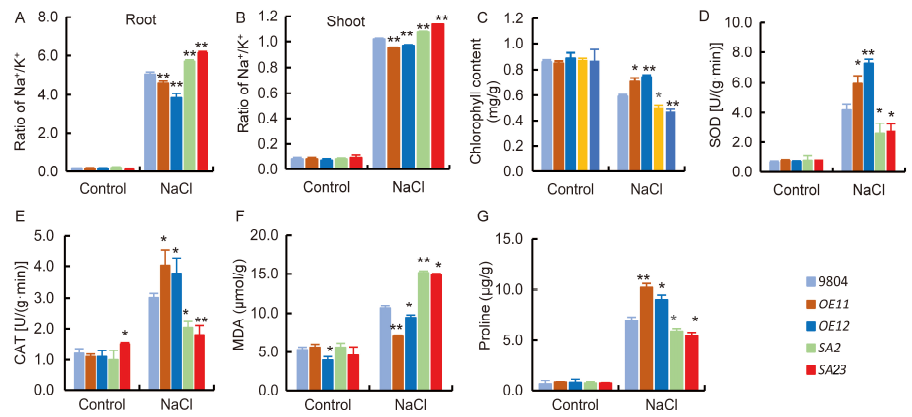
Fig. 4. Responses of 20-day-old transgenic lines and wild type 9804 at 6 d after exposure to 100 mmol/L NaCl treatment.A, Na+/K+ ratio in roots. B, Na+/K+ ratio in shoots. C, Chlorophyll content in the leaves under control and salt-stress conditions for 6 d. D, Superoxide dismutase (SOD) activity in leaves. E, Catalase (CAT) activity in leaves. F, Malomdiadehyde (MDA) content of leaves. G, Proline content of leaves.OE11 and OE12 are the OsSAPK7-overexpression lines. SA2 and SA23 are the OsSAPK7 knockout lines. Values are Mean ± SE (n = 3). * and ** indicate significant differences at the 0.05 and 0.01 levels according to the Student's t-test compared with 9804, respectively.
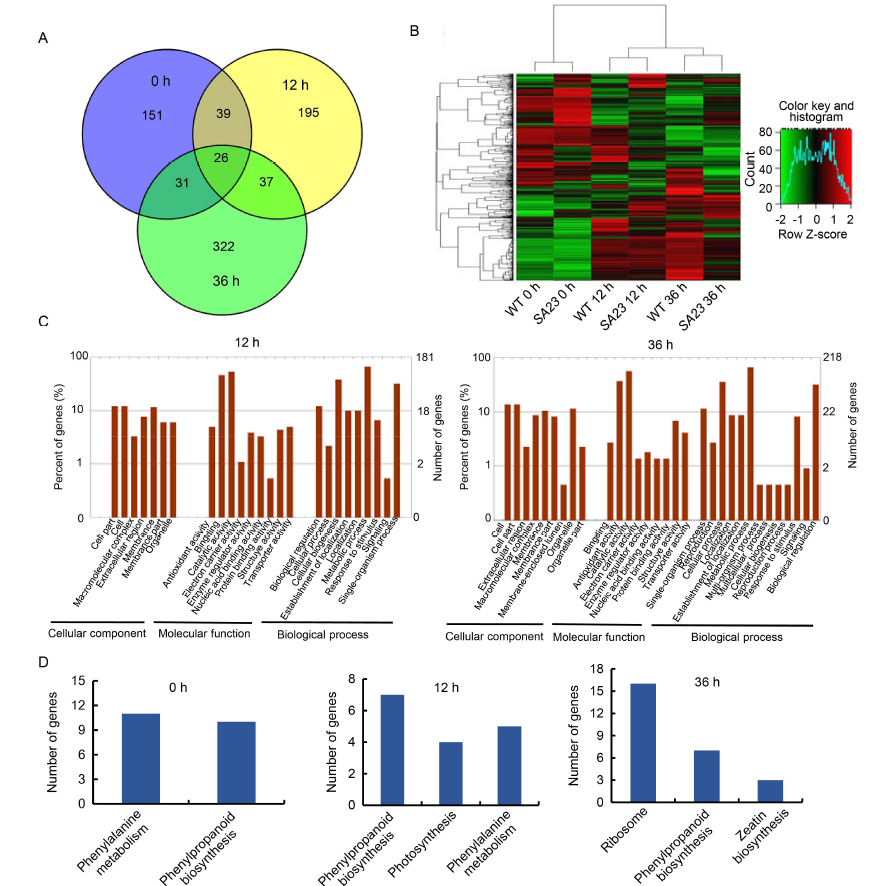
Fig. 5. Transcriptome profiling analysis of OsSAPK7 knockout line SA23 and wild type (WT, 9804) under salt stress conditions.A, Venn diagram of differentially expressed genes (DEGs) in WT vs SA23 at 0, 12 and 36 h under salt-stress conditions.B, Hierarchical clustering of DEGs between WT and SA23 at 12 and 36 h under salt stress. The color scale represents log2 of the fragment per kilo base of exon model per million mapped fragments (FPKM).C, Gene Ontology analysis of DEGs in WT vs SA23 at 12 and 36 h under salt-stress conditions.D, Kyoto Encyclopedia of Genes and Genomes pathway analysis of DEGs in WT vs SA23.
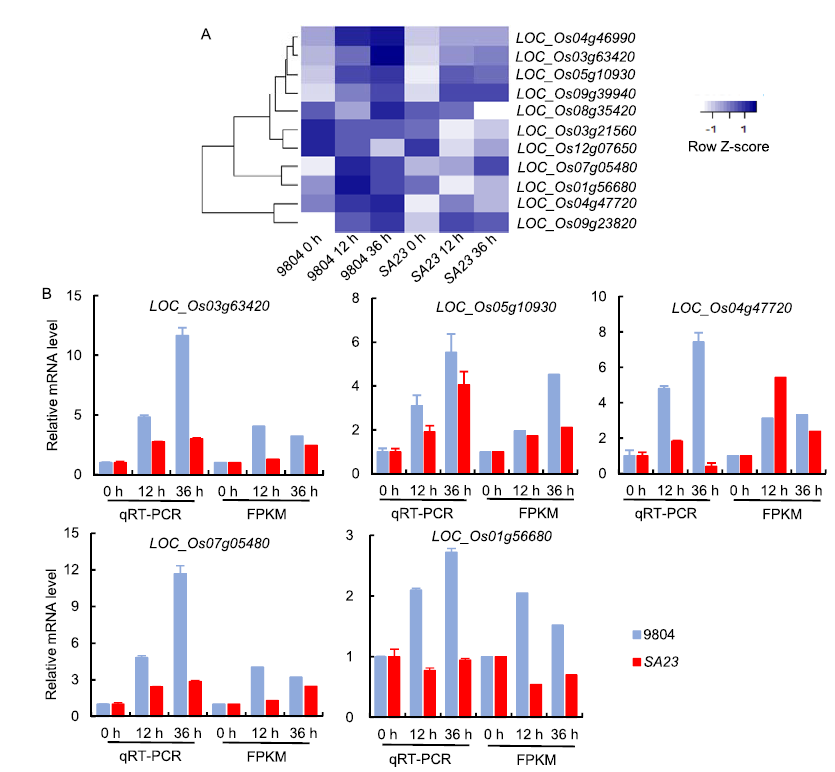
Fig. 6. Expression patterns of differentially expressed genes (DEGs) in OsSAPK7 knockout line SA23 compared with wild type 9804.A, Hierarchical clustering of DEGs associated with electron carrier activity, photosynthesis and zeatin synthesis pathway regulated by OsSAPK7 after salt-stress treatment. Color scale represents the log2 of the fragment per kilo base of exon model per million mapped fragments (FPKM).B, Expression patterns of key genes in 9804 and SA23 assessed by qRT-PCR and FPKM. Values are Mean ± SE (n = 3).
| [1] | Anderberg R J, Walker-Simmons M K. 1992. Isolation of a wheat cDNA clone for an abscisic acid-inducible transcript with homology to protein kinases. Proc Natl Acad Sci USA, 89(21): 10183-10187. |
| [2] | Ashraf M A, Ashraf M, Ali Q. 2010. Response of two genetically diverse wheat cultivars to salt stress at different growth stages: Leaf lipid peroxidation and phenolic contents. Pak J Bot, 42(1): 559-565. |
| [3] | Bhatt T, Sharma A, Puri S, Minhas A P. 2020. Salt tolerance mechanisms and approaches: Future scope of halotolerant genes and rice landraces. Rice Sci, 27(5): 368-383. |
| [4] | Boudsocq M, Barbier-Brygoo H, Lauriere C. 2004. Identification of nine sucrose nonfermenting 1-related protein kinases 2 activated by hyperosmotic and saline stresses in Arabidopsis thaliana. J Biol Chem, 279: 41758-41766. |
| [5] | Civello P M, Martinez G A, Chaves A R, Anon M C. 1995. Peroxidase from strawberry fruit (Fragaria ananassa Duch): Partial purification and determination of some properties. J Agric Food Chem, 43(10): 2596-2601. |
| [6] | Curtis M D, Grossniklaus U. 2003. A gateway cloning vector set for high-throughput functional analysis of genes in planta. Plant Physiol, 133(2): 462-469. |
| [7] | Dey A, Samanta M K, Gayen S, Maiti M K. 2016. The sucrose non-fermenting 1-related kinase 2 gene SAPK9 improves drought tolerance and grain yield in rice by modulating cellular osmotic potential, stomatal closure and stress-responsive gene expression. BMC Plant Biol, 16: 158. |
| [8] | Diédhiou C J, Popova O V, Dietz K J, Golldack D. 2008. The SNF1-type serine-threonine protein kinase SAPK4 regulates stress-responsive gene expression in rice. BMC Plant Biol, 8: 49. |
| [9] | Garg R, Jhanwar S, Tyagi A K, Jain M. 2010. Genome-wide survey and expression analysis suggest diverse roles of glutaredoxin gene family members during development and sesponse to various stimuli in rice. DNA Res, 17(6): 353-367. |
| [10] | Horie T, Karahara I, Katsuhara M. 2015. Salinity tolerance mechanisms in glycophytes: An overview with the central focus on rice plants. Rice, 5(1): 11. |
| [11] | Kim B. 2017. Western blot techniques. In: Espina V. Molecular Profiling: Methods and Protocols, Methods in Molecular Biology. Springer Science and Business Media LLC: 1606: 133-139. |
| [12] | Kobayashi Y, Yamamoto S, Minami H, Kagaya Y, Hattori T. 2004. Differential activation of the rice sucrose nonfermenting1-related protein kinase2 family by hyperosmotic stress and abscisic acid. Plant Cell, 16(5): 1163-1677. |
| [13] | Kulik A, Wawer I, Krzywińska E, Bucholc M, Dobrowolska G. 2011. SnRK2 protein kinases: Key regulators of plant response to abiotic stresses. OMICS: J Interg Biol, 15(12): 859-872. |
| [14] | Lee H J, Park Y J, Seo P J, Kim J H, Sim H J, Kim S G, Park C M. 2015. Systemic immunity requires SnRK28-mediated nuclear import of NPR1 in Arabidopsis. Plant Cell, 27(12): 3425-3438. |
| [15] | Li B, Huang J, Wang L, Li J, Liang Y Y, Chen J. 2020. A review on how plant hormones and environment factors are involved in rice root hair development. Chin J Rice Sci, 34(4): 287-299. (in Chinese with English abstract) |
| [16] | Lou D J, Wang H P, Yu D Q. 2018. The sucrose non-fermenting-1- related protein kinases SAPK1 and SAPK2 function collaboratively as positive regulators of salt stress tolerance in rice. BMC Plant Biol, 18: 203-219. |
| [17] | Ma X L, Zhang Q Y, Zhu Q L, Liu W, Chen Y, Qiu R, Wang B, Yang Z F, Li H Y, Lin Y R, Xie Y Y, Shen R X, Chen S F, Wang Z, Chen Y L, Guo J X, Chen L T, Zhao X C, Dong Z C, Liu Y G. 2005. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot. Mol Plant, 8(8): 1274-1284. |
| [18] | Munns R, Tester M. 2008. Mechanisms of salinity tolerance. Annu Rev Plant Biol, 59: 651-681. |
| [19] | Passricha N, Saifi S K, Kharb P, Tuteja N. 2019. Marker-free transgenic rice plant overexpressing pea LecRLK imparts salinity tolerance by inhibiting sodium accumulation. Plant Mol Biol, 99(3): 265-281. |
| [20] | Rodo A P, Brugière N, Vankova R, Malbeck J, Olson J M, Haines S C, Martin R C, Habben J E, Mok D W S, Mok M C. 2008. Over-expression of a zeatin O-glucosylation gene in maize leads to growth retardation and tasselseed formation. J Exp Bot, 59(10): 2673-2686. |
| [21] | Schmidt R, Mieulet D, Hubberten H M, Obata T, Hoefgen R, Fernie A R, Fisahn J, San B S, Guiderdoni E, Schippers J H M, Mueller-Roeber B. 2013. Salt-responsive ERF1 regulates reactive oxygen species-dependent signaling during the initial response to salt stress in rice. Plant Cell, 25(6): 2115-2131. |
| [22] | Tezvergil-Mutluay A, Agee K A, Hoshika T, Carrilho M, Breschi L, Tjäderhane L, Nishitani Y, Carvalho R M, Looney S, Tay F R, Pashley D H. 2010. The requirement of zinc and calcium ions for functional MMP activity in demineralized dentin matrices. Plant Soil, 26(11): 1059-1067. |
| [23] | Trapnell C, Williams B A, Pertea G, Mortazavi A, Kwan G, van Baren M J, Salzberg S L, Word B J, Pachter L. 2010. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol, 28(5): 511-515. |
| [24] | Vives-Peris V, Gómez-Cadenas A, Pérez-Clemente R M. 2017. Citrus plants exude proline and phytohormones under abiotic stress conditions. Plant Cell Rep, 36(6): 1971-1984. |
| [25] | Wang Y Q, Song F H, Zhu J W, Zhang S S, Yang Y D, Chen T T, Tang B X, Dong L L, Ding N, Zhang Q, Bai Z X, Dong X N, Chen H X, Sun M Y, Zhai S, Sun Y B, Yu L, Lan L, Xiao J F, Fang X D, Lei H X, Zhang Z, Zhao W M. 2017. GSA: Genome sequence archive. Genom Proteom Bioinf, 15(1): 14-18. |
| [26] | Xu J, Duan X G, Yang J, Beeching J R, Zhang P. 2013. Reactive oxygen species scavenging by overproduction of superoxide dismutase and catalase delays postharvest physiological deterioration of cassava storage roots. Plant Physiol, 161(3): 1517-1528. |
| [27] | Xu M R, Huang L Y, Zhang F, Zhu L H, Zhou Y L, Li Z K. 2013. Genome-wide phylogenetic analysis of stress-activated protein kinase genes in rice (OsSAPKs) and expression profiling in response to Xanthomonas oryzae pv oryzicola infection. Plant Mol Biol Rep, 31(4): 877-885. |
| [28] | Ying S, Zhang D F, Li H Y, Liu Y H, Shi Y S, Song Y C, Wang T Y, Li Y. 2011. Cloning and characterization of a maize SnRK2 protein kinase gene confers enhanced salt tolerance in transgenic Arabidopsis. Plant Cell Rep, 30(9): 1683-1699. |
| [29] | Yoshida S, Forno D A, Cock J H, Gomez K A. 1976. Laboratory Manual for Physiological Studies of Rice. Manila, the Philippines: International Rice Research Institute: 61-64. |
| [30] | Zhang M, Smith J A, Harberd N P, Jiang C F. 2016. The regulatory roles of ethylene and reactive oxygen species (ROS) in plant salt stress responses. Plant Mol Biol, 91(6): 651-659. |
| [31] | Zheng Z F, Xu X P, Crosley R A, Greenwalt S A, Sun Y J, Blakeslee B, Wang L Z, Ni W T, Sopko M S, Yao C L, Yau K, Burton S, Zhuang M B, McCaskill D G, Gachotte D, Thompson M, Greene T W. 2010. The protein kinase SnRK2. 6 mediates the regulation of sucrose metabolism and plant growth in Arabidopsis. Plant Physiol, 153(1): 99-113. |
| [32] | Zhou Y B, Liu C, Tang D Y, Yan L, Wang D, Yang Y Z, Gui J S, Zhao X Y, Li L G, Tang X D, Yu F, Li J L, Liu L L, Zhu Y H, Lin J Z, Liu X M. 2018. The receptor-like cytoplasmic kinase STRK1 phosphorylates and activates CatC, thereby regulating H2O2 homeostasis and improving salt tolerance in rice. Plant Cell, 30(5): 1100-1118. |
| [33] | Zhu J K. 2003. Regulation of ion homeostasis under salt stress. Curr Opin Plant Biol, 6(5): 441-445. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||