
Rice Science ›› 2018, Vol. 25 ›› Issue (5): 250-260.DOI: 10.1016/j.rsci.2018.08.001
收稿日期:2018-03-31
接受日期:2018-06-28
出版日期:2018-09-28
发布日期:2018-06-11
. [J]. Rice Science, 2018, 25(5): 250-260.
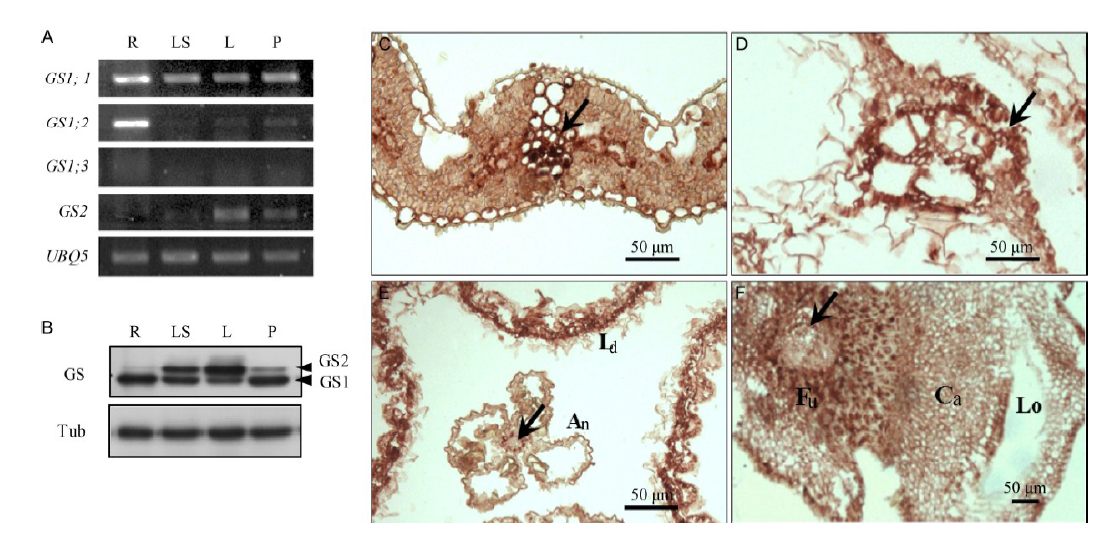
Fig. 1. Expression level of glutamine synthetase (GS) genes in various organs of rice plants. A, GS mRNA expression. RNA was extracted from the root (R), leaf sheath (LS), and leaf (L) of 4-week-old seedling, while panicle (P) was collected at the pre-flowering stage. B, GS protein accumulation. C, Immunolocalization of GS protein in flag leaf. D, Immunolocalization of GS protein in leaf sheath. E, Immunolocalization of GS protein in anther. F, Immunolocalization of GS protein in carpel.Arrows indicate vascular bundles. An, Anther; Ca, Carpel; Fu, Funiculus; Ld, Lodicule; Lo, Locule.
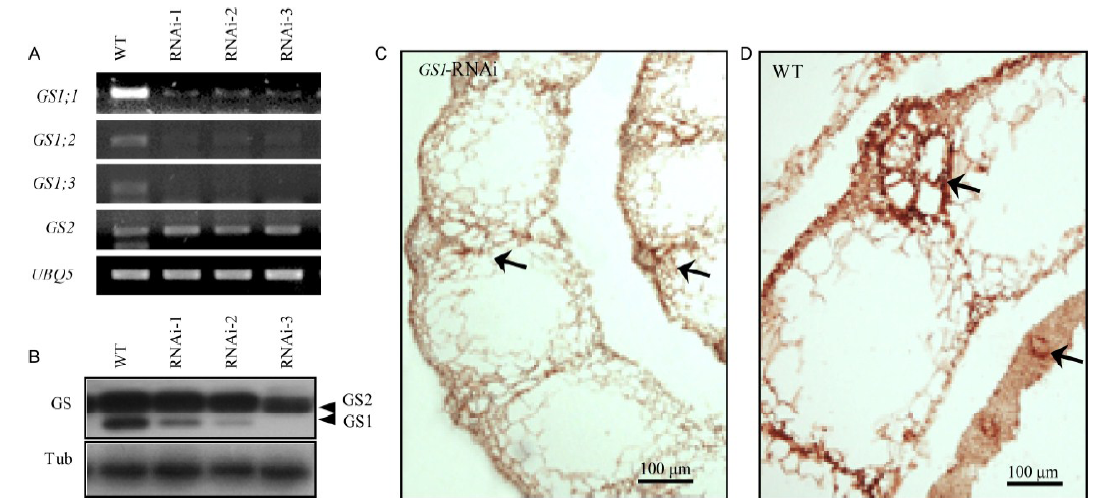
Fig. 2. Glutamine synthetase (GS) expression in wild type (WT) and GS1 RNAi transgenic (GS1-RNAi) rice plants. A, GS mRNA expression. reverse-transcription PCR analysis was performed using the leaves of 4-week-old seedlings. B, GS protein accumulation in the leaves of transgenic plants. C, Immunolocalization of GS protein in the leaf sheath of transgenic rice. D, Immunolocalization of GS protein in the leaf sheath of the WT. Arrows indicate vascular bundles.
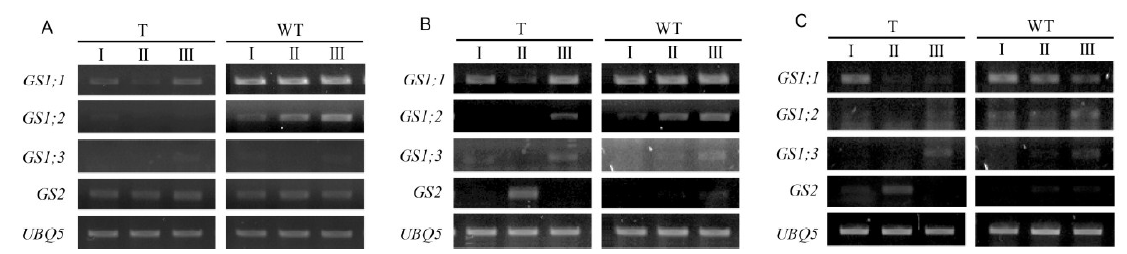
Fig. 3. Expression of glutamine synthetase (GS) mRNAs in flag leaf (A), leaf sheath (B) and panicle (C) during reproductive stages from pre- to post-flowering.WT, Wild type; T, Transgenic rice; I, Pre-flowering stage (7 d before flowering); II, Flowering stage; III, Post-flowering stage (7 d after flowering).
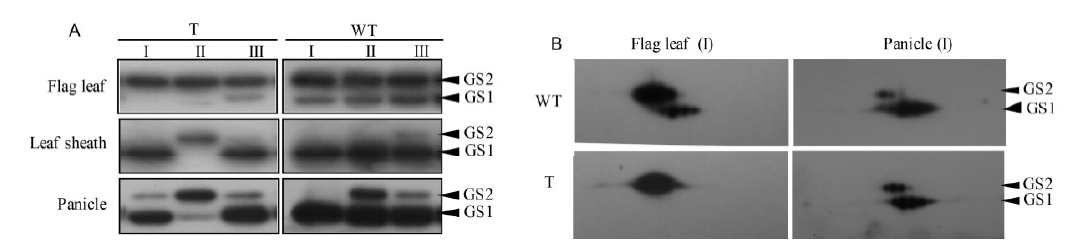
Fig. 4. Accumulation of GS protein in flag leaf, leaf sheath and panicle from pre- to post-flowering stage. A, GS1 and GS2 protein analysis in the RNAi transgenic rice. B, Immunoblot of 2D electrophoresis profile. Total protein (40 μg) was separated by two-dimensional gel electrophoresis. WT, Wild type; T, GS1;1 RNAi transgenic rice; I, Pre-flowering stage (7 d before flowering); II, Flowering stage; III, Post-flowering stage (7 d after flowering).
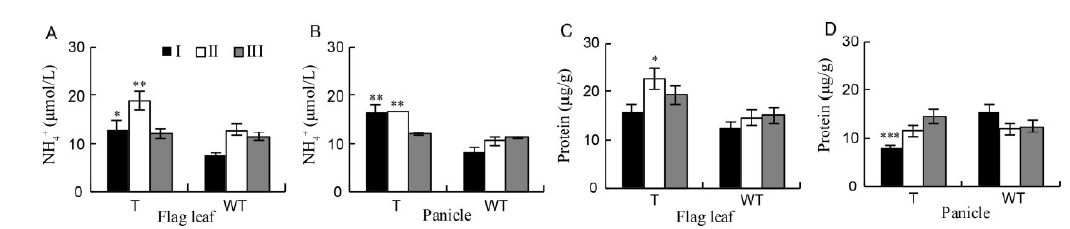
Fig. 5. Ammonium (NH4+) contents in the flag leaves and panicles between GS1 RNAi transgenic and wild-type rice during reproductive stages.A, Ammonium content in leaves; B, Ammonium content in panicles; C, Total soluble protein in leaves; D, Total soluble protein in panicles.WT, Wild type; T, Transgenic rice; I, Pre-flowering stage (7 d before flowering); II, Flowering stage; III, Post-flowering stage (7 d after flowering). Data are mean ± SE from three independent experiments using three randomly selected plants. Asterisks represent significant differences (two-way ANOVA and Bonferroni post-test, *, P < 0.05; **, P < 0.01; ***, P < 0.001).
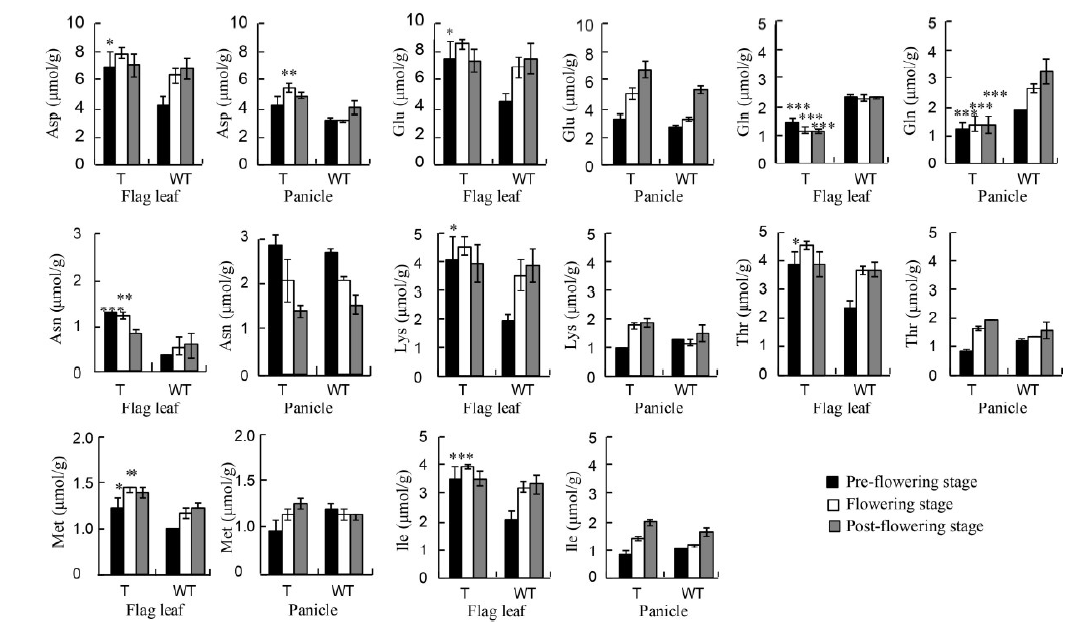
Fig. 6. Amino acid concentrations in flag leaves and panicles of GS1 RNAi transgenic and wild-type rice during the reproductive stages. T, Transgenic rice; WT, Wild type; Asp, Aspartate; Glu, Glutamic acid; Gln, Glutamine; Asn, Asparagine; Lys, Lycine; Thr, Threonine; Met, Methionine; Ile, Isoleucine.Data are mean ± SE of measurements from three independent experiments using three randomly selected plants. Asterisks represent significant differences (two-way ANOVA and Bonferroni post-test, *, P < 0.05; **, P < 0.01; ***, P < 0.001).
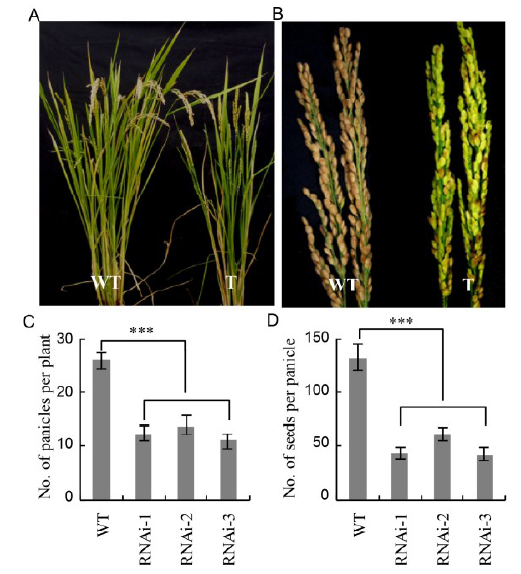
Fig. 7. Phenotype of GS1 RNAi transgenic (T) and wild-type (WT) rice at 16th week after transplanting. A, Panicles; B, Ears; C, Number of panicles per plant; D, Number of seeds per panicle. Data are mean ± SE of measurements from 12 individual plants in three replicates. Asterisk indicates that the value significantly differed from the wild type (one-way ANOVA; ***, P < 0.001).
| 1 | Azevedo R A, Lancien M, Lea P J.2006. The aspartic acid metabolic pathway, an exciting and essential pathway in plants.Amino Acids, 30(2): 143-162. |
| 2 | Bernard S M, Habash D Z.2009. The importance of cytosolic glutamine synthetase in nitrogen assimilation and recycling.New Phytol, 182(3): 608-620. |
| 3 | Blackwell R D, Murray A J S, Lea P J.1987. Inhibition of photosynthesis in barley with decreased levels of chloroplastic glutamine synthetase activity.J Exp Bot, 38(11): 1799-1809. |
| 4 | Brugière N, Dubois F, Masclaux C, Sangwan R S, Hirel B.2000. Immunolocalization of glutamine synthetase in senescing tobacco (Nicotiana tabacum L.) leaves suggests that ammonia assimilation is progressively shifted to the mesophyll cytosol.Planta, 211(4): 519-527. |
| 5 | Cai H M, Zhou Y, Xiao J H, Li X H, Zhang Q F, Lian X M.2009. Overexpressed glutamine synthetase gene modifies nitrogen metabolism and abiotic stress responses in rice.Plant Cell Rep, 28(3): 527-537. |
| 6 | Cantón F R, Suárez M F, Cánovas F M.2005. Molecular aspects of nitrogen mobilization and recycling in trees.Photosynth Res, 83(2): 265-278. |
| 7 | Fuentes S I, Allen D J, Ortiz-Lopez A, Hernández G.2001. Over-expression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations.J Exp Bot, 52: 1071-1081. |
| 8 | Funayama K, Kojima S, Tabuchi-Kobayashi M, Sawa Y, Nakayama Y, Hayakawa T, Yamaya T.2013. Cytosolic glutamine synthetase1;2 is responsible for the primary assimilation of ammonium in rice roots.Plant Cell Physiol, 54(6): 934-943. |
| 9 | Gravois K A, Helms R S.1992. Path analysis of rice yield and yield components as affected by seeding rate.Agron J, 84(1): 1-4. |
| 10 | Guan M, de Bang T C, Pedersen C, Schjoerring J K.2016. Cytosolic glutamine synthetase Gln1;2 is the main isozyme contributing to GS1 activity and can be up-regulated to relieve ammonium toxicity.Plant Physiol, 171(3): 1921. |
| 11 | Habash D Z, Massiah A J, Rong H L, Wallsgrove R M, Leigh R A.2006. The role of cytosolic glutamine synthetase in wheat.Ann Appl Biol, 138(1): 83-89. |
| 12 | Hayashi H, Chino M.1990. Chemical composition of phloem sap from the uppermost internode of the rice plant.Plant Cell Physiol, 31(2): 247-251. |
| 13 | Hirel B, Bertin P, Quilleré I, Bourdoncle W, Attagnant C, Dellay C, Gouy A, Cadiou S, Retailliau C, Falque M, Gallais A.2001. Towards a better understanding of the genetic and physiological basis for nitrogen use efficiency in maize.Plant Physiol, 125(3): 1258-1270. |
| 14 | Ishiyama K, Inoue E, Tabuchi M, Yamaya T, Takahashi H.2004. Biochemical background and compartmentalized functions of cytosolic glutamine synthetase for active ammonium assimilation in rice roots.Plant Cell Physiol, 45(11): 1640-1647. |
| 15 | Jing Z P, Gallardo F, Pascual M B, Sampalo R, Romero J, de Navarra A T, Cánovas F M.2004. Improved growth in a field trial of transgenic hybrid poplar overexpressing glutamine synthetase.New Phytol, 164(1): 137-145. |
| 16 | Kamachi K, Yamaya T, Mae T, Ojima K.1991. A role for glutamine synthetase in the remobilization of leaf nitrogen during natural senescence in rice leaves.Plant Physiol, 96(2): 411-417. |
| 17 | Kamachi K, Yamaya T, Hayakawa T, Mae T, Ojima K.1992. Vascular bundle-specific localization of cytosolic glutamine synthetase in rice leaves.Plant Physiol, 99(4): 1481-1486. |
| 18 | Konishi N, Ishiyama K, Beier M P, Inoue E, Kanno K, Yamaya T, Takahashi H, Kojima S.2017. Contributions of two cytosolic glutamine synthetase isozymes to ammonium assimilation in Arabidopsis roots.J Exp Bot, 68(3): 613-625. |
| 19 | Lea P J, Miflin B J.2003. Glutamate synthase and the synthesis of glutamate in plants.Plant Physiol Bioch, 41(6): 555-564. |
| 20 | Lea P J, Sodek L, Parry M A J, Shewry P R, Halford N G.2006. Asparagine in plants.Ann Appl Biol, 150(1): 1-26. |
| 21 | Lothier J, Gaufichon L, Sormani R, Lemaître T, Azzopardi M, Morin H, Chardon F, Reisdorf-Cren M, Avice J C, Masclaux- Daubresse C.2011. The cytosolic glutamine synthetase GLN1;2 plays a role in the control of plant growth and ammonium homeostasis in Arabidopsis rosettes when nitrate supply is not limiting.J Exp Bot, 62(4): 1375-1390. |
| 22 | Mae T, Ohira K.1981. The remobilization of nitrogen related to leaf growth and senescence in rice plants (Oryza sativa L.).Plant Cell Physiol, 22(6): 1067-1074. |
| 23 | Martin A, Lee J, Kichey T, Gerentes D, Zivy M, Tatout C, Dubois F, Balliau T, Valot B, Davanture M, Tercé-Laforgue T, Quilleré I, Coque M, Gallais A, Gonzalez-Moro M B, Bethencourt L, Habash D Z, Lea P J, Charcosset A, Perez P, Murigneux A, Sakakibara H, Edwards K J, Hirel B.2006. Two cytosolic glutamine synthetase isoforms of maize are specifically involved in the control of grain production.Plant Cell, 18(11): 3252-3274. |
| 24 | Masclaux-Daubresse C, Reisdorf-Cren M, Pageau K, Lelandais M, Grandjean O, Kronenberger J, Valadier M H, Feraud M, Jouglet T, Suzuki A.2006. Glutamine synthetase-glutamate synthase pathway and glutamate dehydrogenase play distinct roles in the sink-source nitrogen cycle in tobacco.Plant Physiol, 140(2): 444-456. |
| 25 | Miki D, Shimamoto D.2004. Simple RNAi vectors for stable and transient suppression of gene function in rice.Plant Cell Physiol, 45(4): 490-495. |
| 26 | Miller B C, Hill J E, Roberts S R.1991. Plant population effects on growth and yield in water-seeded rice.Agron J, 83: 291-297. |
| 27 | Obara M, Sato T, Sasaki S, Kashiba K, Nagano A, Nakamura I, Ebitani T, Yano M, Yamaya T.2004. Identification and characterization of a QTL on chromosome 2 for cytosolic glutamine synthetase content and panicle number in rice.Theor Appl Genet, 110(1): 1-11. |
| 28 | Oliveira I C, Brears T, Knight T J, Clark A, Coruzzi G M.2002. Overexpression of cytosolic glutamine synthetase: Relation to nitrogen, light, and photorespiration.Plant Physiol, 129(3): 1170-1180. |
| 29 | Park S, Lee K, Kang K, Kim Y S, Lee S, Kweon S, Back K.2010. Tryptophan boost caused by senescence occurred independently of cytoplasmic glutamine synthetase.Biosci Biotechnol Bioc, 74(11): 2352-2354. |
| 30 | Postles J, Curtis T Y, Powers S J, Elmore J S, Mottram D S, Halford N G.2016. Changes in free amino acid concentration in rye grain in response to nitrogen and sulfur availability, and expression analysis of genes involved in asparagine metabolism.Front Plant Sci, 7: 917. |
| 31 | Rastogi R, Chourey P S, Muhitch M J.1998. The maize glutamine synthetase GS1 gene is preferentially expressed in kernel pedicels and is developmentally-regulated.Plant Cell Physiol, 39(4): 443-446. |
| 32 | Sakurai N, Hayakawa T, Nakamura T, Yamaya T.1996. Changes in the cellular localization of cytosolic glutamine synthetase protein in vascular bundles of rice leaves at various stages of development.Planta, 200(3): 306-311. |
| 33 | Sakurai N, Katayama Y, Yamaya T.2008. Overlapping expression of cytosolic glutamine synthetase and phenylalanine ammonia- lyase in immature leaf blades of rice.Physiol Plant, 113(3): 400-408. |
| 34 | Swarbreck S M, Defoin-Platel M, Hindle M, Saqi M, Habash D Z.2011. New perspectives on glutamine synthetase in grasses.J Exp Bot, 62(4): 1511-1522. |
| 35 | Tabuchi M, Sugiyama K, Ishiyama K, Inoue E, Sato T, Takahashi H, Yamaya T.2005. Severe reduction in growth rate and grain filling of rice mutants lacking OsGS1;1, a cytosolic glutamine synthetase 1;1.Plant J, 42(5): 641-651. |
| 36 | Tabuchi M, Abiko T, Yamaya T.2007. Assimilation of ammonium ions and reutilization of nitrogen in rice (Oryza sativa L.).J Exp Bot, 58(9): 2319-2327. |
| 37 | Tobin A K, Yamaya T.2001. Cellular compartmentation of ammonium assimilation in rice and barley.J Exp Bot, 52: 591-604. |
| 38 | Wallsgrove R M, Turner J C, Hall N P, Kendall A C, Bright S W J.1987. Barley mutants lacking chloroplast glutamine synthetase: Biochemical and genetic analysis.Plant Physiol, 83(1): 155-158. |
| 39 | Xu Q, Chen W F, Xu Z J.2015. Relationship between grain yield and quality in rice germplasms grown across different growing areas.Breeding Sci, 65(3): 226-232. |
| 40 | Yamaya T, Hayakawa T, Tanasawa K, Kamachi K, Mae T, Ojima K.1992. Tissue distribution of glutamate synthase and glutamine synthetase in rice leaves.Plant Physiol, 100(3): 1427-1432. |
| 41 | Yamaya T, Kusano M.2014. Evidence supporting distinct functions of three cytosolic glutamine synthetases and two NADH-glutamate synthases in rice.J Exp Bot, 65(19): 5519-5525. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||