
Rice Science ›› 2025, Vol. 32 ›› Issue (2): 131-136.DOI: 10.1016/j.rsci.2025.01.001
• • 下一篇
收稿日期:2024-09-10
接受日期:2024-12-02
出版日期:2025-03-28
发布日期:2025-04-14
. [J]. Rice Science, 2025, 32(2): 131-136.
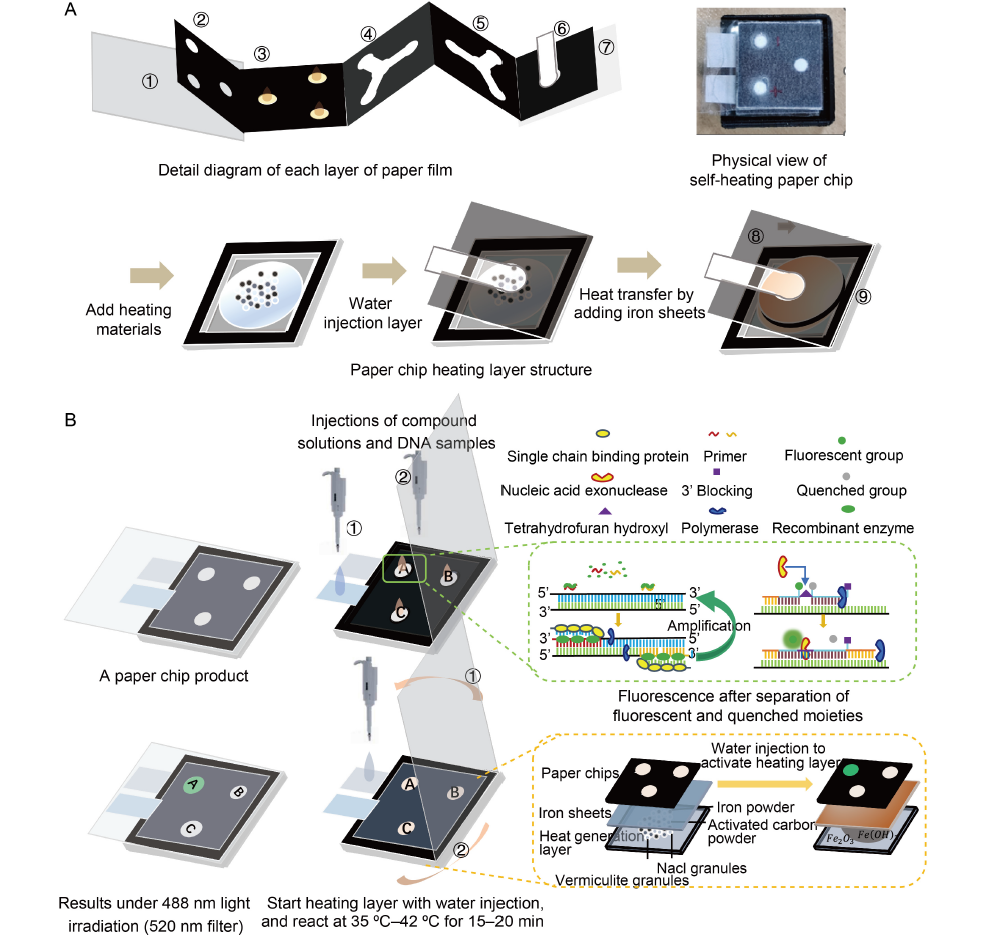
Fig. 1. Schematic diagram of overall structure and reaction of paper-based recombinant polymerase amplification (RPA). A, Fabrication of the layers of paper chips. B, Operation of RPA paper chip. The black area represents hydrophobic zones and the white area represents hydrophilic zones.
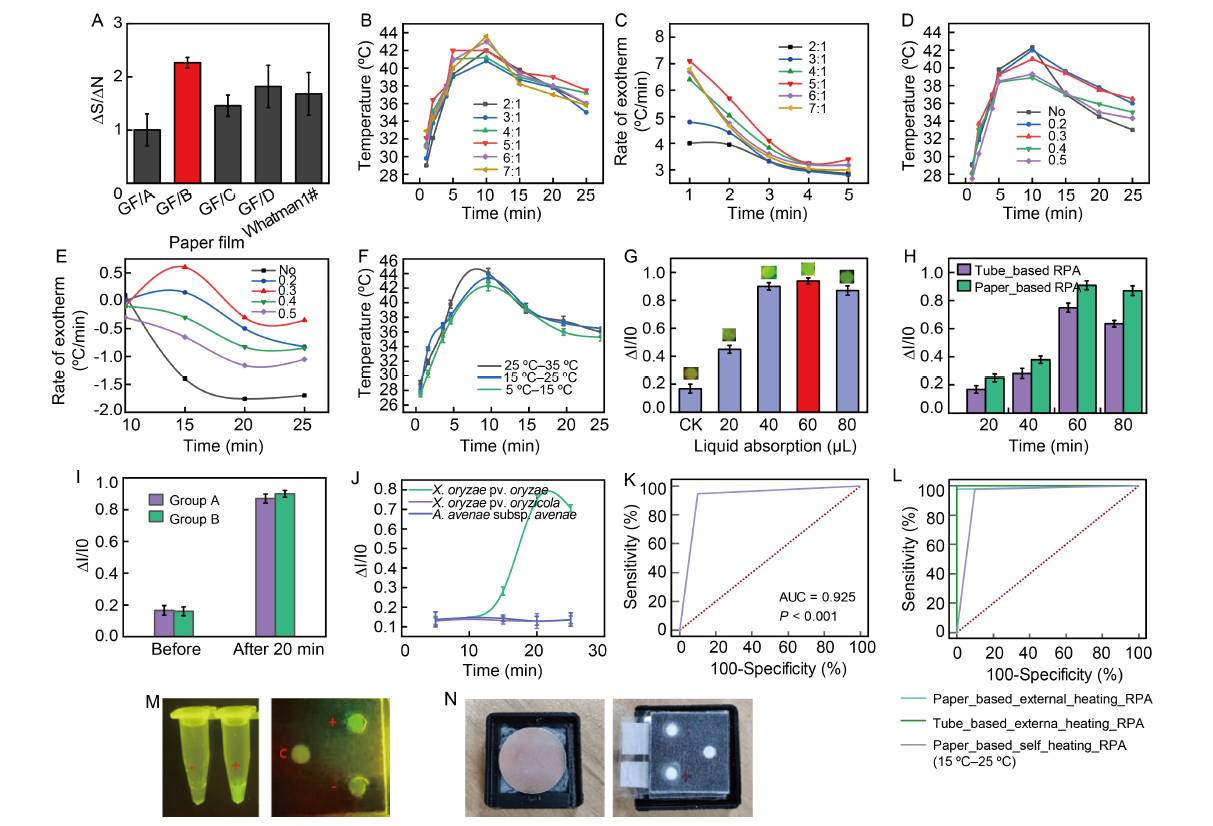
Fig. 2. Diagram of experimental results. A, Comparison of fluorescence intensity of recombinant polymerase amplification (RPA) in different paper films. ∆S represents the change in fluorescence intensity of the reaction pad before and after the RPA reaction, and ∆N is the change in fluorescence intensity of the control pad before and after the RPA reaction without sample DNA. B, Effect of the ratio of iron powder to activated carbon on temperature and time. C, Effect of the ratio of iron powder to activated carbon on the exothermic rate during the first 5 min. D, Effect of the ratio of iron powder to vermiculite on temperature and time. E, Effect of vermiculite insulation on the rate of heat release between 10‒25 min. F, Variation in heating layer temperature profile at different ambient temperatures. G, Effect of complex solution volume on the activation of pre-deposited reactants and assay outcomes. H, Comparison of paper-based RPA and tube-based RPA outcomes. I, Comparison of RPA results from self-heating paper chips (Group B) with those from ordinary paper chips using external heating equipment (Group A). J, Specific detection of different pathogens (Xanthomonas oryzae pv. Oryzae, X. oryzae pv. oryzicol, and Acidovorax avenae subsp. Avenae) using a self-heating paper chip device. I0 represents fluorescence signal intensity in the paper-based detection region when there is no response, and ∆I represents the difference in fluorescence signal intensity between the reacted and non-reacted paper-based detection regions. K, Receiver operating characteristic (ROC) analysis of samples detected by paper-based self-heating RPA chip. L, Comparison of reliability of this chip method with RPA conventional tube-based solutions and standard paper-based chips using an externally heated platform, as evaluated by ROC curves. M, Physical appearance of paper-based and tube-based RPA results. ‘c’ refers to control group, ‘+’ for positive, and ‘-’ for negative. N, Physical view of paper chip heating layer and overall device. Fluorescence photometric values of the detection area on the paper and test tube substrate were measured under 495 nm wavelength irradiation and 520 ± 10 nm wavelength acquisition (at the end of the assay, images of all the detection areas were captured with a smartphone and converted from colour intensity to optical density using ImageJ software). Data were expressed as Mean ± SD of three replicates.
| [1] | Aghababaie M, Foroushani E S, Changani Z, et al. 2023. Recent advances in the development of enzymatic paper-based microfluidic biosensors. Biosens Bioelectron, 226: 115131. |
| [2] | Ahn H, Batule B S, Seok Y, et al. 2018. Single-step recombinase polymerase amplification assay based on a paper chip for simultaneous detection of multiple foodborne pathogens. Anal Chem, 90(17): 10211-10216. |
| [3] | Banerjee A, Bag M K, Chandra A K, et al. 2023a. Development and application of recombinase polymerase amplification for rapid detection of rice false smut pathogen (Ustilaginoidea virens). Crop Prot, 167: 106204. |
| [4] | Banerjee A, Bharti S, Kumar J, et al. 2023b. Recombinase polymerase amplification based rapid detection of aroma gene in rice. Rice Sci, 30(2): 96-99. |
| [5] | Bao M D, Chen Q, Xu Z H, et al. 2021. Challenges and opportunities for clustered regularly interspaced short palindromic repeats based molecular biosensing. ACS Sens, 6(7): 2497-2522. |
| [6] | Eady M, Park B, Hinton A. 2020. Rapid identification of Campylobacter strains cultured under aerobic incubation using hyperspectral microscope imaging. J Food Prot, 83(3): 405-411. |
| [7] | Erkocyigit B A, Ozufuklar O, Yardim A, et al. 2023. Biomarker detection in early diagnosis of cancer: Recent achievements in point-of-care devices based on paper microfluidics. Biosensors, 13(3): 387. |
| [8] | Kaymaz S V, Akar U, Elitas M. 2021. Nucleic acid extraction methods and technologies for on-site plant-pathogen diagnostics. ACS Agric Sci Technol, 1(6): 581-585. |
| [9] | Kim H J, Kim Y, Park S J, et al. 2018. Development of colorimetric paper sensor for pesticide detection using competitive-inhibiting reaction. BioChip J, 12(4): 326-331. |
| [10] | Lee H, Yi S Y, Kwon J S, et al. 2021. Rapid and highly sensitive pathogen detection by real-time DNA monitoring using a nanogap impedimetric sensor with recombinase polymerase amplification. Biosens Bioelectron, 179: 113042. |
| [11] | Lee S H, Park S M, Kim B N, et al. 2019. Emerging ultrafast nucleic acid amplification technologies for next-generation molecular diagnostics. Biosens Bioelectron, 141: 111448. |
| [12] | Liu H X, Zhou X M, Liu W P, et al. 2016. Paper-based bipolar electrode electrochemiluminescence switch for label-free and sensitive genetic detection of pathogenic bacteria. Anal Chem, 88(20): 10191-10197. |
| [13] | Magro L, Jacquelin B, Escadafal C, et al. 2017. Paper-based RNA detection and multiplexed analysis for Ebola virus diagnostics. Sci Rep, 7(1): 1347. |
| [14] | Rabello L G,da Conceição Ribeiro R C. 2021. A novel vermiculite/ vegetable polyurethane resin-composite for thermal insulation eco-brick production. Compos Part B Eng, 221: 109035. |
| [15] | Rohrman B A, Richards-Kortum R R. 2012. A paper and plastic device for performing recombinase polymerase amplification of HIV DNA. Lab Chip, 12(17): 3082-3088. |
| [16] | Roy S, Mohd-Naim N F, Safavieh M, et al. 2017. Colorimetric nucleic acid detection on paper microchip using loop mediated isothermal amplification and crystal violet dye. ACS Sens, 2(11): 1713-1720. |
| [17] | Seok Y, Joung H A, Byun J Y, et al. 2017. A paper-based device for performing loop-mediated isothermal amplification with real- time simultaneous detection of multiple DNA targets. Theranostics, 7(8): 2220-2230. |
| [18] | Shasmita, Samal P, Mohapatra P K, et al. 2021. Improved photosystem II and defense enzymes activity in rice (Oryza sativa) by biopriming against Xanthomonas oryzae pv. oryzae. Funct Plant Biol, 48(3): 298-311. |
| [19] | Shruthi U, Nagaveni V, Raghavendra B K. 2019. A review on machine learning classification techniques for plant disease detection. In: 2019 5th International Conference on Advanced Computing & Communication Systems (ICACCS). March 15-16, 2019, Coimbatore, India: IEEE: 281-284. |
| [20] | Somvanshi S B, Ulloa A M, Zhao M, et al. 2022. Microfluidic paper-based aptasensor devices for multiplexed detection of pathogenic bacteria. Biosens Bioelectron, 207: 114214. |
| [21] | Tang R H, Yang H, Gong Y, et al. 2017. A fully disposable and integrated paper-based device for nucleic acid extraction, amplification and detection. Lab Chip, 17(7): 1270-1279. |
| [22] | Thepbandit W, Papathoti N K, Daddam J R, et al. 2021. Identification of salicylic acid mechanism against leaf blight disease in Oryza sativa by SR-FTIR microspectroscopic and docking studies. Pathogens, 10(6): 652. |
| [23] | Toth A, Bobok E. 2017. Basic equations of fluid mechanics and thermodynamics. In: Flow and Heat Transfer in Geothermal Systems. Amsterdam, the Netherlands: Elsevier: 21-55. |
| [24] | Wang Y Q, Ke Y Q, Liu W J, et al. 2020. A one-pot toolbox based on Cas12a/crRNA enables rapid foodborne pathogen detection at attomolar level. ACS Sens, 5(5): 1427-1435. |
| [25] | Weng S Z, Hu X J, Wang J H, et al. 2021. Advanced application of Raman spectroscopy and surface-enhanced Raman spectroscopy in plant disease diagnostics: A review. J Agric Food Chem, 69(10): 2950-2964. |
| [26] | Yang N, Wang P, Xue C Y, et al. 2018. A portable detection method for organophosphorus and carbamates pesticide residues based on multilayer paper chip. J Food Process Eng, 41(8): e12867. |
| [27] | Zhang J N, Yang Y, Feng X P, et al. 2020. Identification of bacterial blight resistant rice seeds using terahertz imaging and hyperspectral imaging combined with convolutional neural network. Front Plant Sci, 11: 821. |
| [28] | Zhu Z B, Li R, Zhang H W, et al. 2022. PAM-free loop-mediated isothermal amplification coupled with CRISPR/Cas12a cleavage (Cas-PfLAMP) for rapid detection of rice pathogens. Biosens Bioelectron, 204: 114076. |
| [29] | Zhuang J W, Zhao Z Y, Lian K, et al. 2022. SERS-based CRISPR/ Cas assay on microfluidic paper analytical devices for supersensitive detection of pathogenic bacteria in foods. Biosens Bioelectron, 207: 114167. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||