
Rice Science ›› 2026, Vol. 33 ›› Issue (2): 260-276.DOI: 10.1016/j.rsci.2026.02.002
• • 上一篇
收稿日期:2025-09-19
接受日期:2025-12-12
出版日期:2026-03-28
发布日期:2026-04-01
. [J]. Rice Science, 2026, 33(2): 260-276.
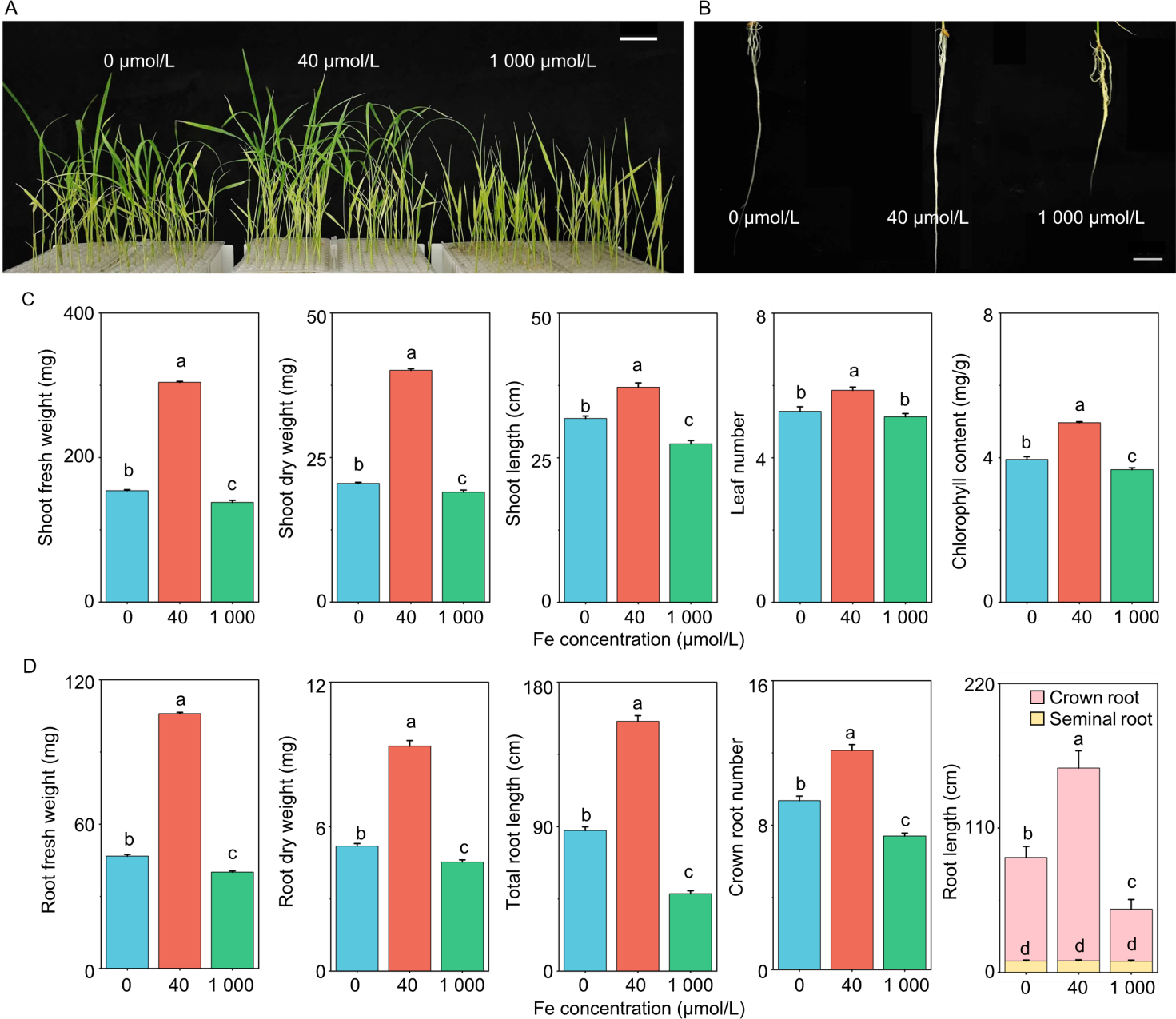
Fig. 1. Performance of shoot and root of three-week-old rice seedlings under iron (Fe) deficiency (0 μmol/L), control (40 μmol/L), and excess (1 000 μmol/L) conditions. A and B, Shoot (A) and root (B) phenotype of rice seedlings after three weeks under three different Fe conditions. Scale bars in A and B are 4 cm and 2 cm, respectively. C, Fresh weight, dry weight, and shoot length, as well as leaf number, chlorophyll content of the first leaf. D, Fresh weight of root, dry weight of root, total root length, number of crown roots, and lengths of seminal and crown roots. In C and D, data are mean ± SD (n = 3). Different lowercase letters above bars indicate significant differences at the 0.05 level according to Student’s t-test.
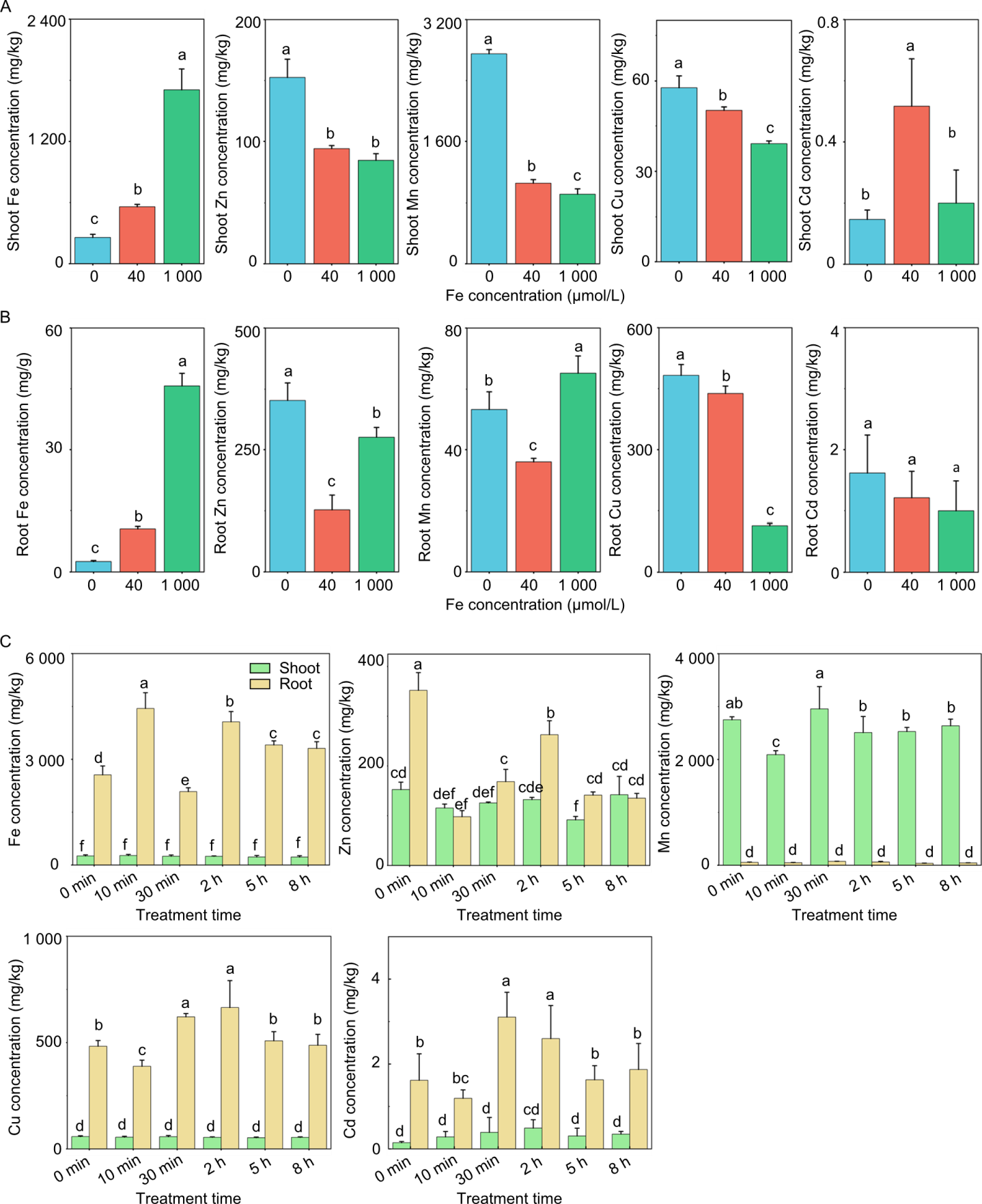
Fig. 2. Effects of iron (Fe) stress and Fe supplementation on metal ion concentrations in rice roots and shoots. A and B, Concentrations of Fe, zinc (Zn), manganese (Mn), copper (Cu), and cadmium (Cd) in shoots and roots of three-week-old rice seedlings under Fe deficiency (0 μmol/L), control (40 μmol/L), and excess (1 000 μmol/L) conditions. C, Dynamic changes in shoot and root ion concentrations in Fe-deficient rice after supplementing with 40 μmol/L Fe. Data are mean ± SD (n = 3). Different lowercase letters above bars indicate significant differences at the 0.05 level according to Student’s t-test (A and B) and one-way ANOVA (C), respectively.
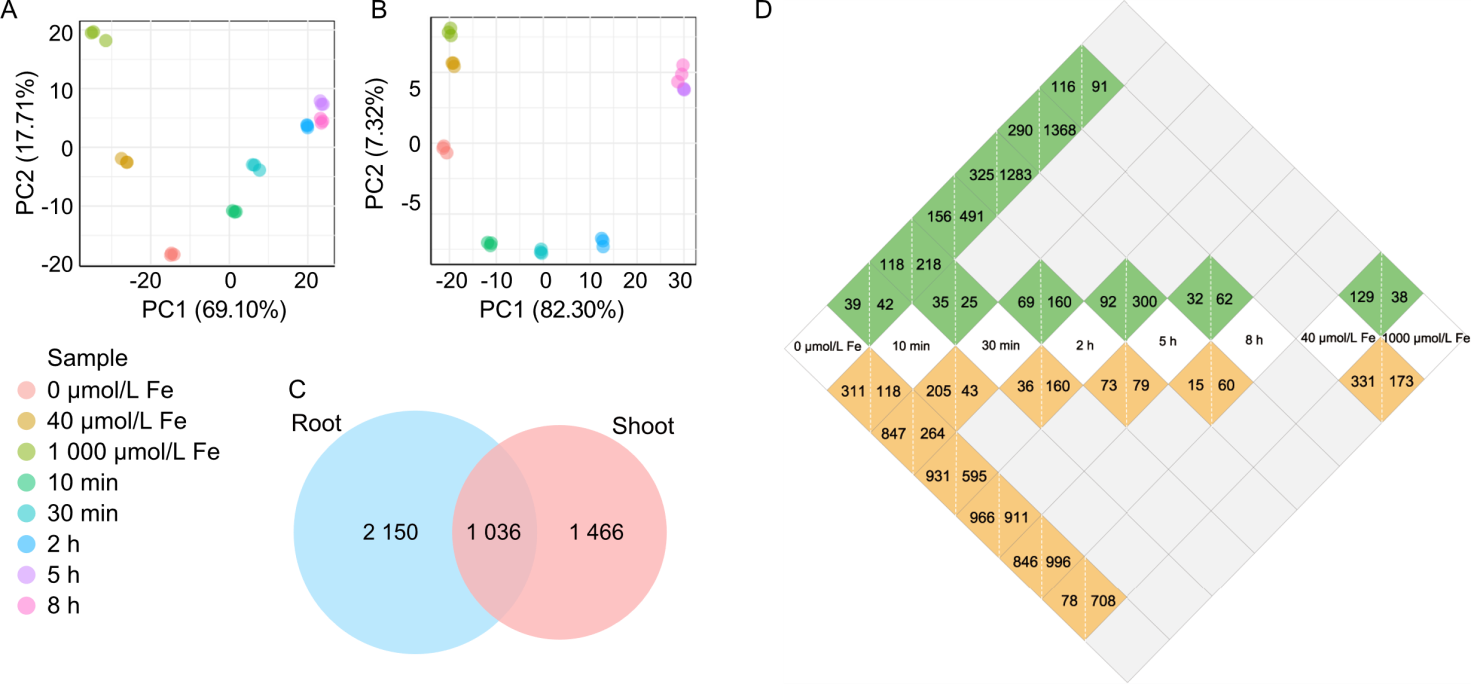
Fig. 3. RNA sequencing analysis of steady-state response of rice to iron (Fe) deficiency and excess, and dynamic response of Fe-supplemented rice under Fe deficiency. A and B, Principal component analysis (PCA) of RNA sequencing data from root (A) and shoot (B) samples under eight growth conditions: static treatment groups [control (40 μmol/L), deficiency (0 μmol/L), excess (1 000 μmol/L)] and dynamic Fe supplementation time points (10 min, 30 min, 2 h, 5 h, 8 h) in Fe-deficient plants. C, Venn diagram showing total number of unique differentially expressed genes (DEGs) identified in both roots and shoots across all comparisons. Each treatment includes three biological replicates (15 plants per replicate).
| NCBI_Locus | Gene | Tissue | Clustera |
|---|---|---|---|
| LOC4342862 | Nramp1 | Root, shoot | 8, 6 |
| LOC4330161 | YSL2 | Root, shoot | 8, 6 |
| LOC4336546 | YSL16 | Root | 4 |
| LOC9268232 | YSL15 | Root | 5 |
| LOC4346978 | VIT2 | Root, shoot | 8, 6 |
| LOC4336074 | VIT1 | Root | 4 |
| LOC4342431 | EIN2 | Root | 4 |
| LOC4324610 | COPT1 | Root | 5 |
| LOC4332069 | FRDL1 | Root, shoot | 7, 6 |
| LOC4326394 | ZIP1 | Root | 8 |
| LOC4333667 | IRT2 | Root | 4 |
| LOC4333669 | IRT1 | Root, shoot | 4, 6 |
| LOC4343412 | ABCG43 | Root | 4 |
| LOC4332125 | MTP4 | Shoot | 6 |
Table 1. Metal homeostasis-related genes involved in all differentially expressed genes (DEGs) in roots and shoots
| NCBI_Locus | Gene | Tissue | Clustera |
|---|---|---|---|
| LOC4342862 | Nramp1 | Root, shoot | 8, 6 |
| LOC4330161 | YSL2 | Root, shoot | 8, 6 |
| LOC4336546 | YSL16 | Root | 4 |
| LOC9268232 | YSL15 | Root | 5 |
| LOC4346978 | VIT2 | Root, shoot | 8, 6 |
| LOC4336074 | VIT1 | Root | 4 |
| LOC4342431 | EIN2 | Root | 4 |
| LOC4324610 | COPT1 | Root | 5 |
| LOC4332069 | FRDL1 | Root, shoot | 7, 6 |
| LOC4326394 | ZIP1 | Root | 8 |
| LOC4333667 | IRT2 | Root | 4 |
| LOC4333669 | IRT1 | Root, shoot | 4, 6 |
| LOC4343412 | ABCG43 | Root | 4 |
| LOC4332125 | MTP4 | Shoot | 6 |
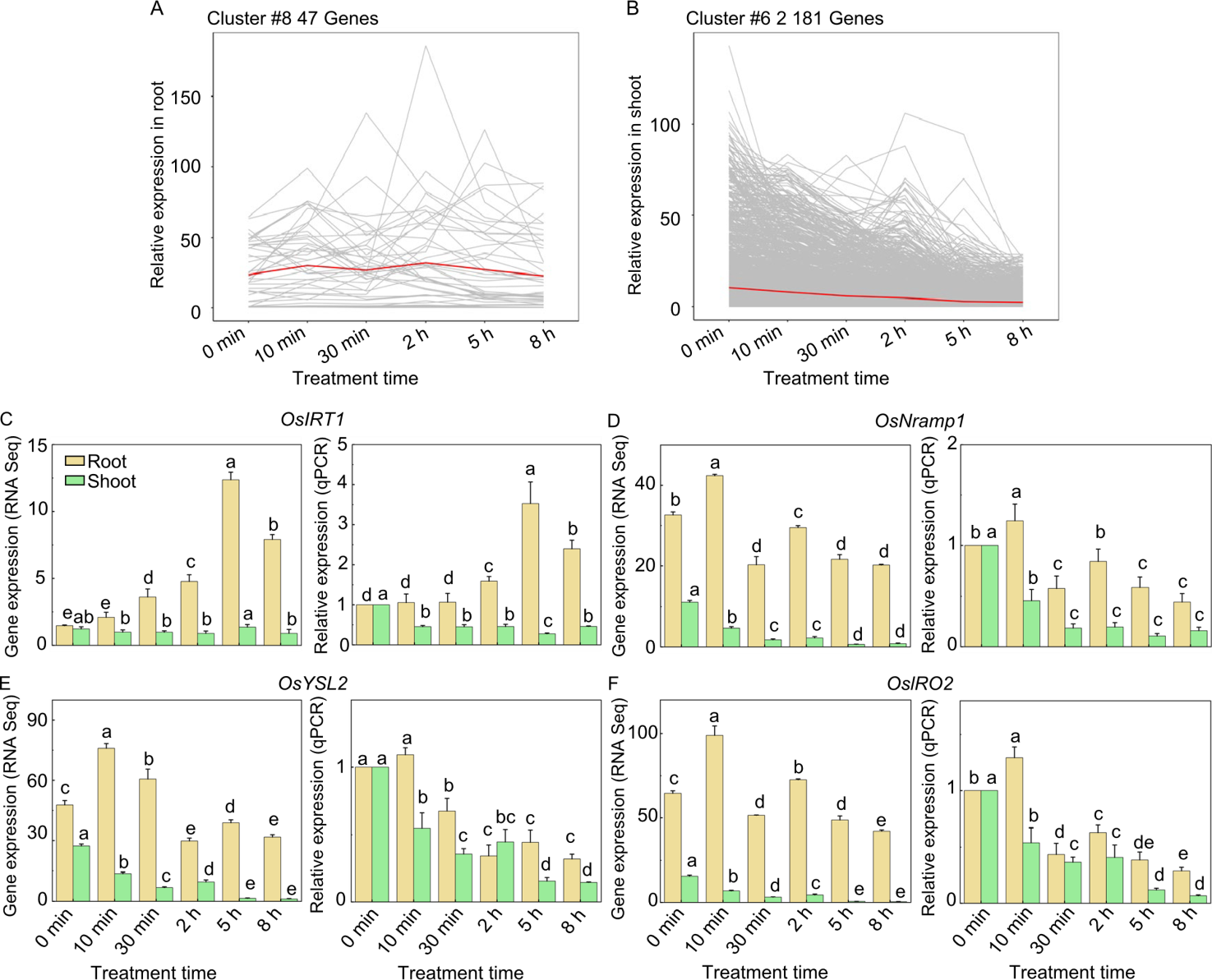
Fig. 4. Cluster analysis of gene expression and expression of iron transport genes in rice following iron deficiency and supplementation. A and B, Relative expression of cluster #8 in roots (A) and cluster #6 in shoots (B). Pearson correlation is used as the distance metric in k-means clustering. The number of differentially expressed genes (DEGs) included in each cluster is noted in the upper left corner of the graph. Complete clustering data for DEGs in roots and shoots are shown in Fig. S1, respectively. Each grey line represents the expression of one gene across three biological replicates, with the red line indicating the average expression of all genes within the cluster. C-F, Transcript levels of RNA sequencing (RNA-seq, left) and qRT-PCR (right) of OsIRT1, OsNramp1, OsYSL2, and OsIRO2 in roots and shoots of iron-deficient rice seedlings after iron supplementation, respectively. The internal gene is the house-keeping gene EP. Samples collected at specified time points (0 min, 10 min, 30 min, 2 h, 5 h, 8 h after iron supplementation). Expression levels in qRT-PCR are normalized, setting the condition with 0 μmol/L Fe at 0 min as 1. Different lowercase letters above bars indicate significant differences at the 0.05 level between root and shoot samples by Student’s t-test.
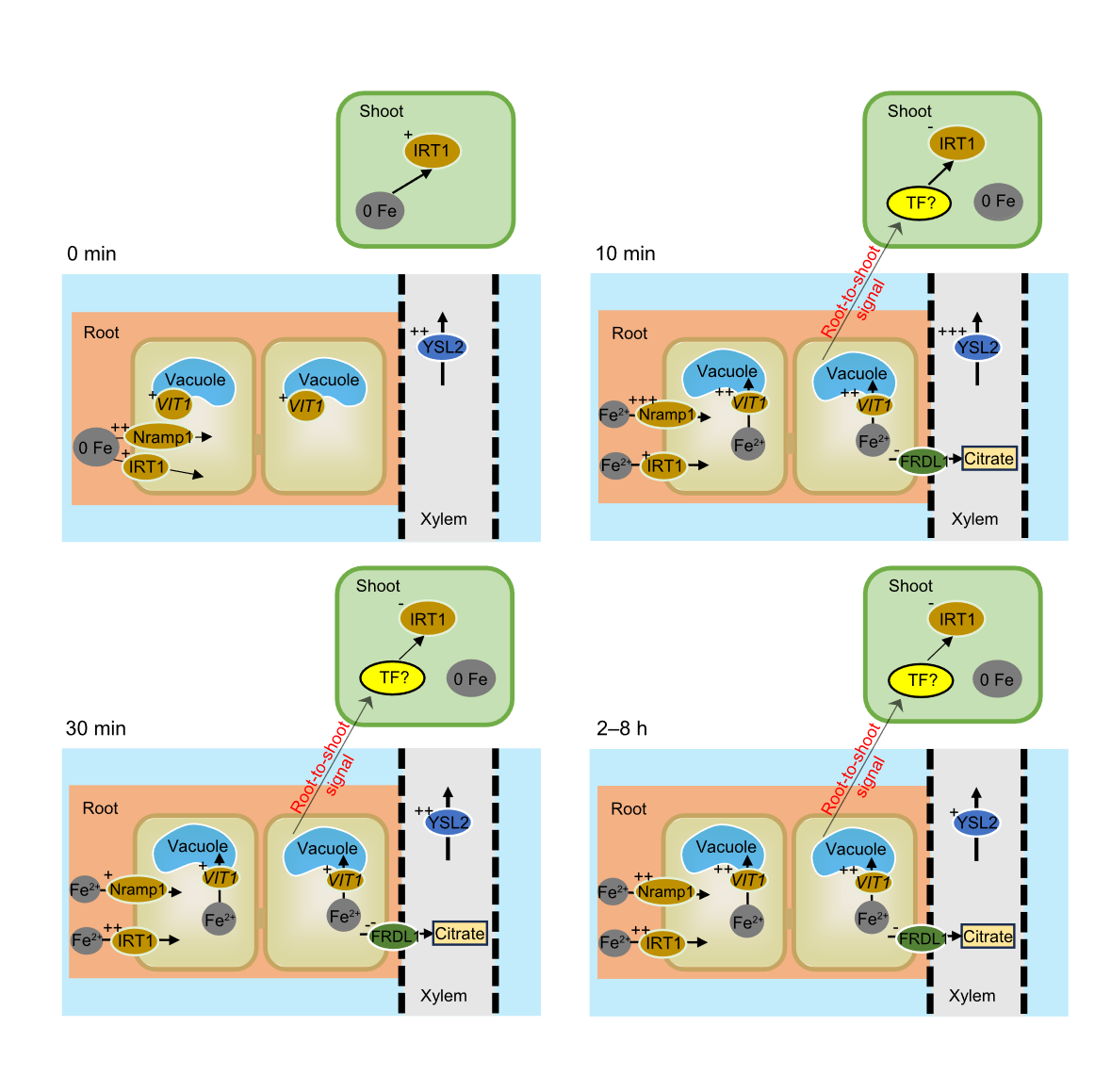
Fig. 5. Patterns of regulation of short-term iron (Fe) uptake by rice roots and shoots after Fe supplementation in Fe-deficient rice. ‘-’ indicates downregulation, ‘+’ indicates upregulation, and the higher number of ‘+’ indicates higher expression. ‘0 Fe’ indicates Fe deficiency treatment (0 μmol/L). FRDL, Ferric reductase defective like; IRT, Iron-regulated transporter; Nramp, Natural resistance-associated macrophage protein; TF, Transcription factors; VIT, Vacuolar iron transporter; YSL, Yellow stripe-like transporter.
| [1] | Amini S, Arsova B, Gobert S, et al. 2021. Transcriptional regulation of ZIP genes is independent of local zinc status in Brachypodium shoots upon zinc deficiency and resupply. Plant Cell Environ, 44(10): 3376-3397. |
| [2] | Aoyama T, Kobayashi T, Takahashi M, et al. 2009. OsYSL18 is a rice iron(III)-deoxymugineic acid transporter specifically expressed in reproductive organs and phloem of lamina joints. Plant Mol Biol, 70(6): 681-692. |
| [3] | Aung M S, Masuda H, Kobayashi T, et al. 2018. Physiological and transcriptomic analysis of responses to different levels of iron excess stress in various rice tissues. Soil Sci Plant Nutr, 64(3): 370-385. |
| [4] | Barberon M, Dubeaux G, Kolb C, et al. 2014. Polarization of IRON-REGULATED TRANSPORTER 1 (IRT1) to the plant-soil interface plays crucial role in metal homeostasis. Proc Natl Acad Sci USA, 111(22): 8293-8298. |
| [5] | Bashir K, Nozoye T, Nagasaka S, et al. 2017. Paralogs and mutants show that one DMA synthase functions in iron homeostasis in rice. J Exp Bot, 68(7): 1785-1795. |
| [6] | Boonyaves K, Gruissem W, Bhullar N K. 2016. NOD promoter-controlled AtIRT1 expression functions synergistically with NAS and FERRITIN genes to increase iron in rice grains. Plant Mol Biol, 90(3): 207-215. |
| [7] | Cai Y R, Li Y, Liang G. 2021. FIT and bHLH Ib transcription factors modulate iron and copper crosstalk in Arabidopsis. Plant Cell Environ, 44(5): 1679-1691. |
| [8] | Cailliatte R, Schikora A, Briat J F, et al. 2010. High-affinity manganese uptake by the metal transporter NRAMP1 is essential for Arabidopsis growth in low manganese conditions. Plant Cell, 22(3): 904-917. |
| [9] | Cao L D, Gao Y, Yu J L, et al. 2021. Streptomyces hygroscopicus OsiSh-2-induced mitigation of Fe deficiency in rice plants. Plant Physiol Biochem, 158: 275-283. |
| [10] | Conte S S, Walker E L. 2011. Transporters contributing to iron trafficking in plants. Mol Plant, 4(3): 464-476. |
| [11] | Curie C, Panaviene Z, Loulergue C, et al. 2001. Maize yellow stripe1 encodes a membrane protein directly involved in Fe(III) uptake. Nature, 409: 346-349. |
| [12] | Dehnad D, Ghorani B, Emadzadeh B, et al. 2024. Recent advances in iron encapsulation and its application in food fortification. Crit Rev Food Sci Nutr, 64: 12685-12701. |
| [13] | DeLoose M, Clúa J, Cho H, et al. 2024. Recent advances in unraveling the mystery of combined nutrient stress in plants. Plant J, 117(6): 1764-1780. |
| [14] | DiDonato R J Jr, Roberts L A, Sanderson T, et al. 2004. Arabidopsis Yellow Stripe-Like2 (YSL2): A metal-regulated gene encoding a plasma membrane transporter of nicotianamine-metal complexes. Plant J, 39(3): 403-414. |
| [15] | dos Santos L R, de Sousa Paula L, Pereira Y C, et al. 2021. Brassinosteroids-mediated amelioration of iron deficiency in soybean plants: Beneficial effects on the nutritional status, photosynthetic pigments and chlorophyll fluorescence. J Plant Growth Regul, 40(5): 1803-1823. |
| [16] | Fang X Z, Xu X L, Ye Z Q, et al. 2024. Excessive iron deposition in root apoplast is involved in growth arrest of roots in response to low pH. J Exp Bot, 75(10): 3188-3200. |
| [17] | Forde B G. 2014. Nitrogen signalling pathways shaping root system architecture: An update. Curr Opin Plant Biol, 21: 30-36. |
| [18] | Garnica M, Bacaicoa E, Mora V, et al. 2018. Shoot iron status and auxin are involved in iron deficiency-induced phytosiderophores release in wheat. BMC Plant Biol, 18(1): 105. |
| [19] | Inoue H, Kobayashi T, Nozoye T, et al. 2009. Rice OsYSL15 is an iron-regulated iron(III)-deoxymugineic acid transporter expressed in the roots and is essential for iron uptake in early growth of the seedlings. J Biol Chem, 284(6): 3470-3479. |
| [20] | Ishimaru Y, Masuda H, Bashir K, et al. 2010. Rice metal-nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J, 62(3): 379-390. |
| [21] | Judesse Soviguidi D R, Ping H Q, Pan B Z, et al. 2025. Oryza sativa iron regulated transporter 1 (OsIRT1) and OsIRT2 are involved in ferrous iron uptake in rice. Plant Physiol Biochem, 226: 110059. |
| [22] | Kavitha P G, Kuruvilla S, Mathew M K. 2015. Functional characterization of a transition metal ion transporter, OsZIP 6 from rice (Oryza sativa L.). Plant Physiol Biochem, 97: 165-174. |
| [23] | Kawakami Y, Bhullar N K. 2021. Delineating the future of iron biofortification studies in rice: Challenges and future perspectives. J Exp Bot, 72(6): 2099-2113. |
| [24] | Kirk G J D, Manwaring H R, Ueda Y, et al. 2022. Below-ground plant-soil interactions affecting adaptations of rice to iron toxicity. Plant Cell Environ, 45(3): 705-718. |
| [25] | Kobayashi T. 2019. Understanding the complexity of iron sensing and signaling cascades in plants. Plant Cell Physiol, 60(7): 1440-1446. |
| [26] | Kobayashi T, Ogo Y, Itai R N, et al. 2007. The transcription factor IDEF1 regulates the response to and tolerance of iron deficiency in plants. Proc Natl Acad Sci USA, 104: 19150-19155. |
| [27] | Kobayashi T, Nakanishi Itai R, Nishizawa N K. 2014. Iron deficiency responses in rice roots. Rice, 7(1): 27. |
| [28] | Kobayashi T, Ozu A, Kobayashi S, et al. 2019. OsbHLH058 and OsbHLH059 transcription factors positively regulate iron deficiency responses in rice. Plant Mol Biol, 101(4/5): 471-486. |
| [29] | Koike S, Inoue H, Mizuno D, et al. 2004. OsYSL2 is a rice metal-nicotianamine transporter that is regulated by iron and expressed in the phloem. Plant J, 39(3): 415-424. |
| [30] | Lee S, Chiecko J C, Kim S A, et al. 2009. Disruption of OsYSL15 leads to iron inefficiency in rice plants. Plant Physiol, 150(2): 786-800. |
| [31] | Lee S, Jeong H J, Kim S A, et al. 2010. OsZIP5 is a plasma membrane zinc transporter in rice. Plant Mol Biol, 73(4/5): 507-517. |
| [32] | Li X L, Zhang H M, Ai Q, et al. 2016. Two bHLH transcription factors, bHLH34 and bHLH104, regulate iron homeostasis in Arabidopsis thaliana. Plant Physiol, 170(4): 2478-2493. |
| [33] | Li Y Y, Zhao J T, Zhang B W, et al. 2016. The influence of iron plaque on the absorption, translocation and transformation of mercury in rice (Oryza sativa L.) seedlings exposed to different mercury species. Plant Soil, 398: 87-97. |
| [34] | Liang G, Zhang H M, Li X L, et al. 2017. bHLH transcription factor bHLH115 regulates iron homeostasis in Arabidopsis thaliana. J Exp Bot, 68(7): 1743-1755. |
| [35] | Liang G, Zhang H M, Li Y, et al. 2020. Oryza sativa FER-LIKE FE DEFICIENCY-INDUCED TRANSCRIPTION FACTOR (OsFIT/ OsbHLH156) interacts with OsIRO2 to regulate iron homeostasis. J Integr Plant Biol, 62(5): 668-689. |
| [36] | Liu Z J, Giehl R F H, Hartmann A, et al. 2020. Seminal and nodal roots of barley differ in anatomy, proteome and nitrate uptake capacity. Plant Cell Physiol, 61(7): 1297-1308. |
| [37] | Mohammed K F A, Kaul T, Agrawal P K, et al. 2023. Function identification and characterization of Oryza sativa ZRT and IRT-like proteins computationally for nutrition and biofortification in rice. J Biomol Struct Dyn, 41(15): 7490-7510. |
| [38] | Nishiyama R, Kato M, Nagata S, et al. 2012. Identification of Zn-nicotianamine and Fe-2’-deoxymugineic acid in the phloem sap from rice plants (Oryza sativa L.). Plant Cell Physiol, 53(2): 381-390. |
| [39] | Nozoye T, Nagasaka S, Bashir K, et al. 2014. Nicotianamine synthase 2 localizes to the vesicles of iron-deficient rice roots, and its mutation in the YXXϕ or LL motif causes the disruption of vesicle formation or movement in rice. Plant J, 77(2): 246-260. |
| [40] | Ogo Y, Itai R N, Nakanishi H, et al. 2006. Isolation and characterization of IRO2, a novel iron-regulated bHLH transcription factor in graminaceous plants. J Exp Bot, 57(11): 2867-2878. |
| [41] | Ogo Y, Itai R N, Nakanishi H, et al. 2007. The rice bHLH protein OsIRO2 is an essential regulator of the genes involved in Fe uptake under Fe-deficient conditions. Plant J, 51(3): 366-377. |
| [42] | Ogo Y, Kobayashi T, Nakanishi Itai R, et al. 2008. A novel NAC transcription factor, IDEF2 that recognizes the iron deficiency-responsive element 2 regulates the genes involved in iron homeostasis in plants. J Biol Chem, 283(19): 13407-13417. |
| [43] | Pedas P, Ytting C K, Fuglsang A T, et al. 2008. Manganese efficiency in barley: Identification and characterization of the metal ion transporter HvIRT1. Plant Physiol, 148(1): 455-466. |
| [44] | Santi S, Schmidt W. 2009. Dissecting iron deficiency-induced proton extrusion in Arabidopsis roots. New Phytol, 183(4): 1072-1084. |
| [45] | Santiago-Rivas S, Moreda-Piñeiro A, Bermejo-Barrera A, et al. 2007. Fractionation metallothionein-like proteins in mussels with on line metal detection by high performance liquid chromatography-inductively coupled plasma-optical emission spectrometry. Talanta, 71(4): 1580-1586. |
| [46] | Schaaf G, Schikora A, Häberle J, et al. 2005. A putative function for the Arabidopsis Fe-phytosiderophore transporter homolog AtYSL2 in Fe and Zn homeostasis. Plant Cell Physiol, 46(5): 762-774. |
| [47] | Selby-Pham J, Lutz A, Moreno-Moyano L T, et al. 2017. Diurnal changes in transcript and metabolite levels during the iron deficiency response of rice. Rice, 10(1): 14. |
| [48] | Senoura T, Sakashita E, Kobayashi T, et al. 2017. The iron-chelate transporter OsYSL9 plays a role in iron distribution in developing rice grains. Plant Mol Biol, 95(4/5): 375-387. |
| [49] | Sharma R, Yeh K C. 2020. The dual benefit of a dominant mutation in Arabidopsis IRON DEFICIENCY TOLERANT1 for iron biofortification and heavy metal phytoremediation. Plant Biotechnol J, 18(5): 1200-1210. |
| [50] | Sheraz S, Wan Y F, Venter E, et al. 2021. Subcellular dynamics studies of iron reveal how tissue-specific distribution patterns are established in developing wheat grains. New Phytol, 231(4): 1644-1657. |
| [51] | Sinclair S A, Senger T, Talke I N, et al. 2018. Systemic upregulation of MTP2- and HMA2-mediated Zn partitioning to the shoot supplements local Zn deficiency responses. Plant Cell, 30(10): 2463-2479. |
| [52] | Subedi S R, Sandhu N, Singh V K, et al. 2019. Genome-wide association study reveals significant genomic regions for improving yield, adaptability of rice under dry direct seeded cultivation condition. BMC Genomics, 20(1): 471. |
| [53] | Tiong J, McDonald G K, Genc Y, et al. 2014. HvZIP7 mediates zinc accumulation in barley (Hordeum vulgare) at moderately high zinc supply. New Phytol, 201(1): 131-143. |
| [54] | Vert G, Grotz N, Dédaldéchamp F, et al. 2002. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell, 14(6): 1223-1233. |
| [55] | Wang B L, Li G, Zhang W H. 2015. Brassinosteroids are involved in Fe homeostasis in rice (Oryza sativa L.). J Exp Bot, 66(9): 2749-2761. |
| [56] | Wang N, Cui Y, Liu Y, et al. 2013. Requirement and functional redundancy of Ib subgroup bHLH proteins for iron deficiency responses and uptake in Arabidopsis thaliana. Mol Plant, 6(2): 503-513. |
| [57] | Wang Q, Zhu Y C, Zou X, et al. 2020. Nitrogen deficiency-induced decrease in cytokinins content promotes rice seminal root growth by promoting root meristem cell proliferation and cell elongation. Cells, 9(4): 916. |
| [58] | Wang S D, Li L, Ying Y H, et al. 2020. A transcription factor OsbHLH156 regulates Strategy II iron acquisition through localising IRO2 to the nucleus in rice. New Phytol, 225(3): 1247-1260. |
| [59] | Wang W J, Ye J, Ma Y R, et al. 2020. OsIRO3 plays an essential role in iron deficiency responses and regulates iron homeostasis in rice. Plants, 9(9): 1095. |
| [60] | Yamagata A, Murata Y, Namba K, et al. 2022. Uptake mechanism of iron-phytosiderophore from the soil based on the structure of yellow stripe transporter. Nat Commun, 13(1): 7180. |
| [61] | Yuan Y X, Wu H L, Wang N, et al. 2008. FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res, 18(3): 385-397. |
| [62] | Zeng H Q, Wu H C, Yan F, et al. 2021. Molecular regulation of zinc deficiency responses in plants. J Plant Physiol, 261: 153419. |
| [63] | Zhang C, Shinwari K I, Luo L, et al. 2018. OsYSL13 is involved in iron distribution in rice. Int J Mol Sci, 19(11): 3537. |
| [64] | Zhang H M, Li Y, Yao X N, et al. 2017. POSITIVE REGULATOR OF IRON HOMEOSTASIS1, OsPRI1, facilitates iron homeostasis. Plant Physiol, 175(1): 543-554. |
| [65] | Zhang H M, Li Y, Pu M N, et al. 2020. Oryza sativa POSITIVE REGULATOR OF IRON DEFICIENCY RESPONSE 2 (OsPRI2) and OsPRI3 are involved in the maintenance of Fe homeostasis. Plant Cell Environ, 43(1): 261-274. |
| [66] | Zhang J, Liu B, Li M S, et al. 2015. The bHLH transcription factor bHLH104 interacts with IAA-LEUCINE RESISTANT3 and modulates iron homeostasis in Arabidopsis. Plant Cell, 27(3): 787-805. |
| [67] | Zhang J L, Zhu Y C, Yu L J, et al. 2022. Research advances in cadmium uptake, transport and resistance in rice (Oryza sativa L.). Cells, 11(3): 569. |
| [68] | Zhu J M, Li J, Hu X Y, et al. 2024. Role of transcription factor complex OsbHLH156-OsIRO2 in regulating manganese, copper, and zinc transporters in rice. J Exp Bot, 75(3): 1112-1127. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||