
Rice Science ›› 2024, Vol. 31 ›› Issue (6): 712-724.DOI: 10.1016/j.rsci.2024.08.001
收稿日期:2024-04-25
接受日期:2024-08-05
出版日期:2024-11-28
发布日期:2024-12-10
. [J]. Rice Science, 2024, 31(6): 712-724.
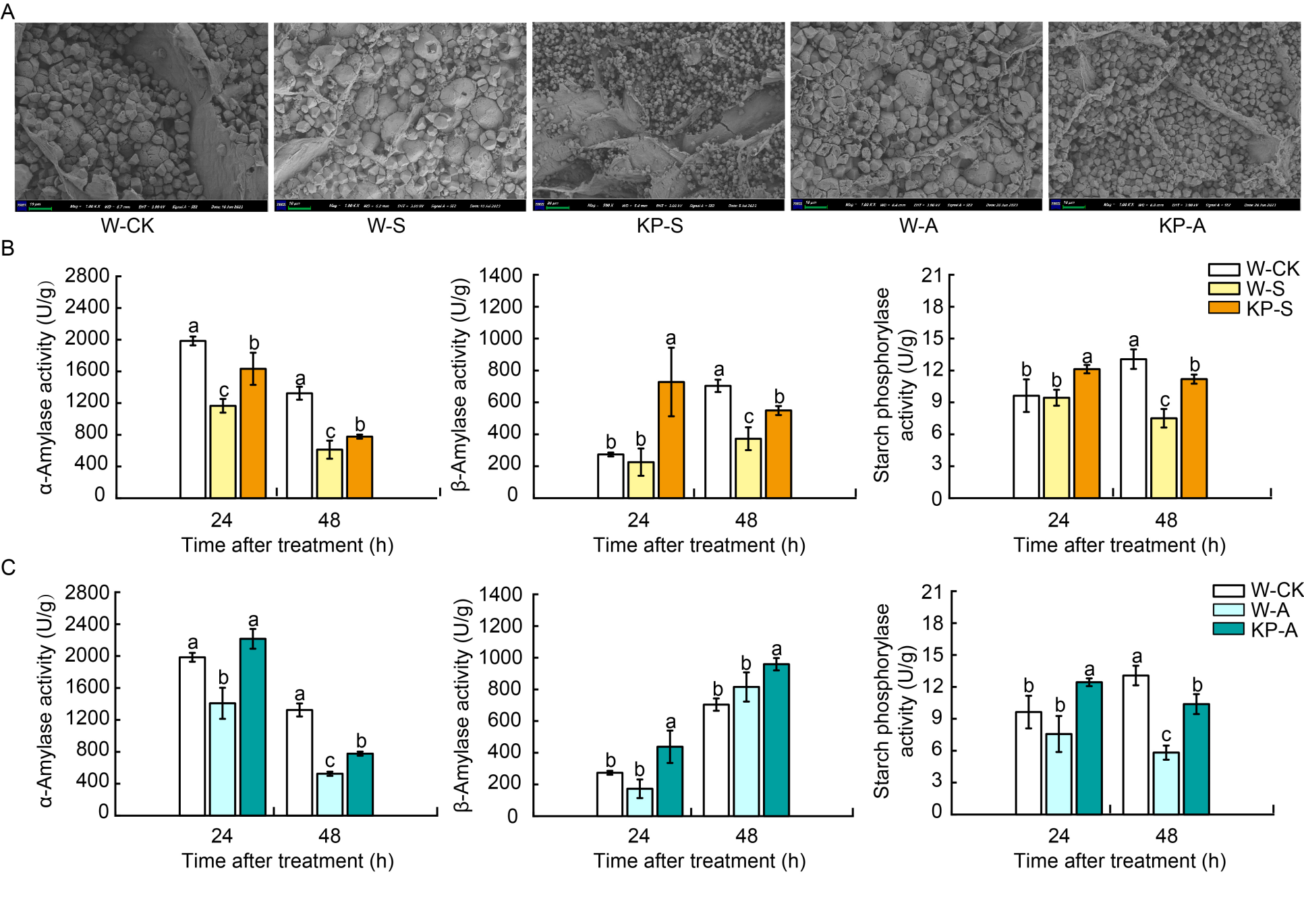
Fig. 1. Mixed-oligosaccharide (KP) priming increases starch and sucrose metabolism during seedling establishment under salt and alkaline stress. A, Visualization of starch granules from hypocotyl regions of W-CK, W-S, W-A, KP-S, and KP-A after 48 h germination. Scale bars, 10 µm. B and C, Amylase and starch phosphorylase activities in W- and KP-primed seeds under salt stress (B) and alkaline stress (C) after 24 and 48 h imbibition. W-CK, Distilled water priming + distilled water culture; W-S, Distilled water priming + 50 mmol/L NaCl culture; KP-S, KP-priming + 50 mmol/L NaCl culture; W-A, Distilled water priming + 15 mmol/L Na2CO3 culture; KP-A, KP-priming + 15 mmol/L Na2CO3 culture. Data are Mean ± SE of three replicates. The lowercase letters above the bars represent significant differences between groups according to Duncan’s multiple range test at P < 0.05.
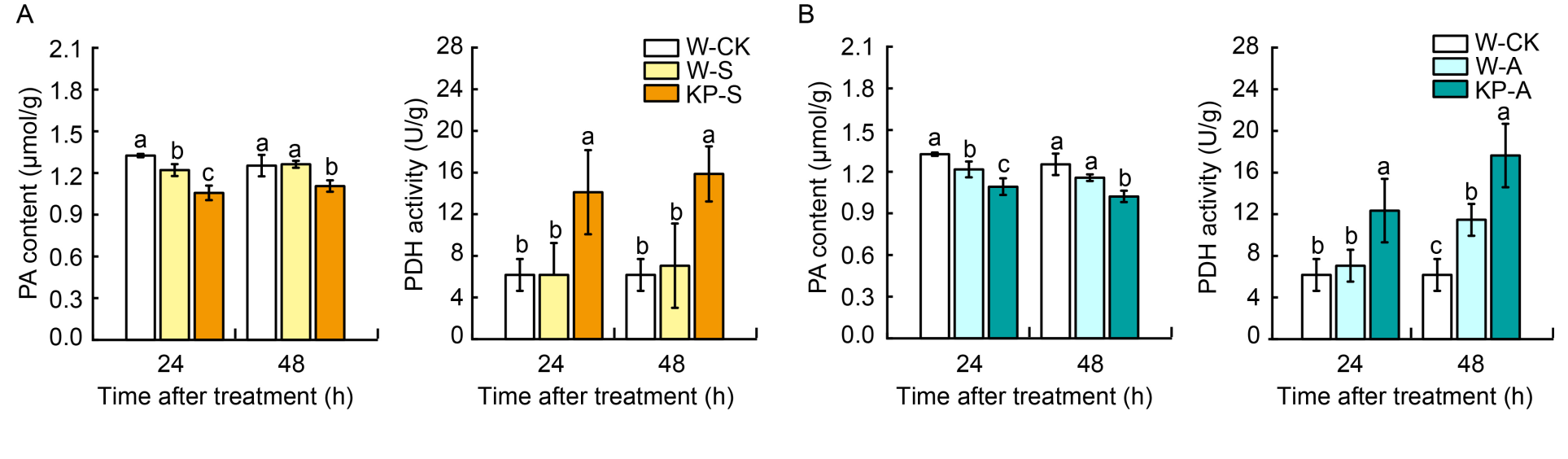
Fig. 2. Mixed-oligosaccharide (KP) priming decreases pyruvate (PA) content and increase pyruvate dehydrogenase (PDH) activity during seedling establishment under salt and alkaline stress. A and B, PA content and PDH activity in W- and KP-primed seeds under salt stress (A) and alkaline stress (B) after 24 and 48 h imbibition. W-CK, Distilled water priming + distilled water culture; W-S, Distilled water priming + 50 mmol/L NaCl culture; KP-S, KP-priming + 50 mmol/L NaCl culture; W-A, Distilled water priming + 15 mmol/L Na2CO3 culture; KP-A, KP-priming + 15 mmol/L Na2CO3 culture. Data are Mean ± SE of three replicates. The lowercase letters on vertical bars represent significant differences between groups according to Duncan’s multiple range test at P < 0.05.
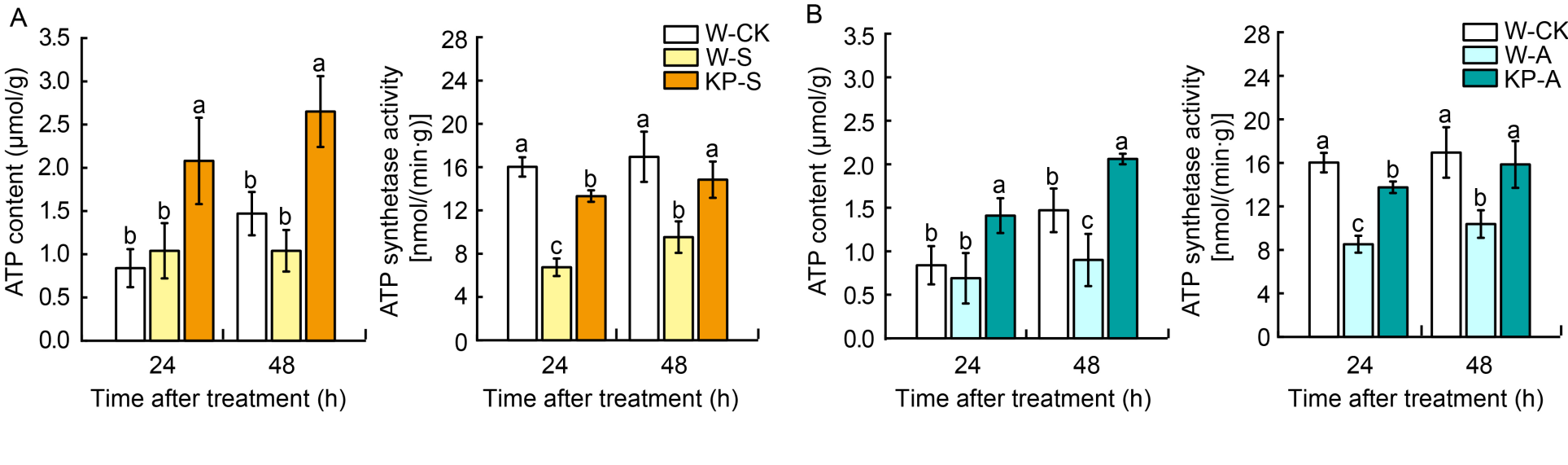
Fig. 3. Comparing ATP content and ATP synthase activity in water (W) and mixed-oligosaccharide (KP) priming under salt and alkaline stress. A and B, ATP content and ATP synthase activity in W- and KP-primed seeds under salt stress (A) and alkaline stress (B) after 24 and 48 h imbibition. W-CK, Distilled water priming + distilled water culture; W-S, Distilled water priming + 50 mmol/L NaCl culture; KP-S, KP-priming + 50 mmol/L NaCl culture; W-A, Distilled water priming + 15 mmol/L Na2CO3 culture; KP-A, KP-priming + 15 mmol/L Na2CO3 culture. Data are Mean ± SE of three replicates. The lowercase letters on vertical bars represent significant differences between groups according to Duncan’s multiple range test at P < 0.05.
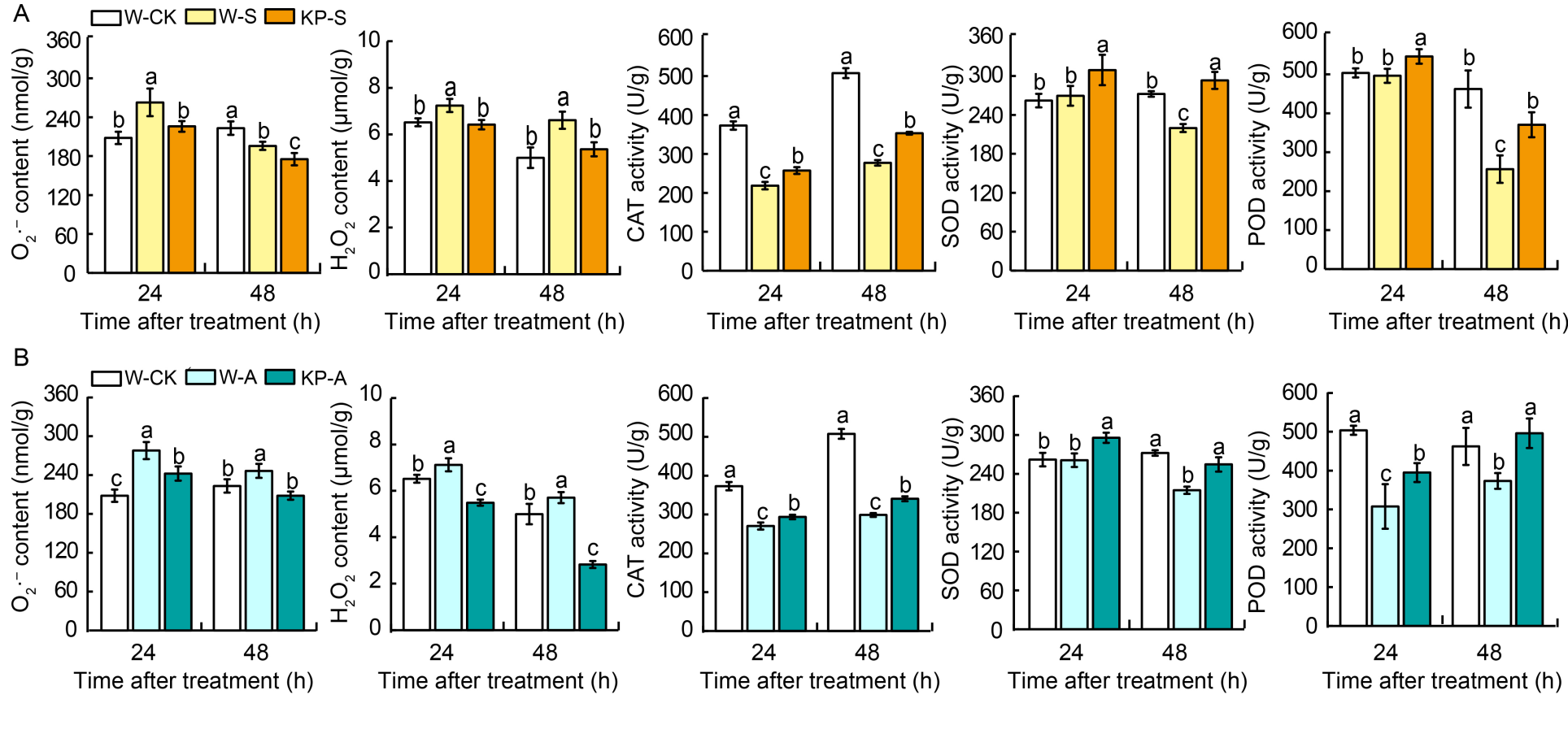
Fig. 4. Mixed-oligosaccharide (KP) priming enhances detoxification of reactive oxygen species during seedling establishment under salt stress. A and B, Contents of O2·̄, H2O2, and the activities of superoxide dismutase (SOD), peroxidases (POD), catalase (CAT) in W- and KP-primed seeds under salt stress (A) and alkaline stress (B) after 24 and 48 h imbibition. W-CK, Distilled water priming + distilled water culture; W-S, Distilled water priming + 50 mmol/L NaCl culture; KP-S, KP-priming + 50 mmol/L NaCl culture; W-A, Distilled water priming + 15 mmol/L Na2CO3 culture; KP-A, KP-priming + 15 mmol/L Na2CO3 culture. Data are Mean ± SE of three replicates. The lowercase letters above the represent significant differences between groups according to Duncan’s multiple range test at P < 0.05.
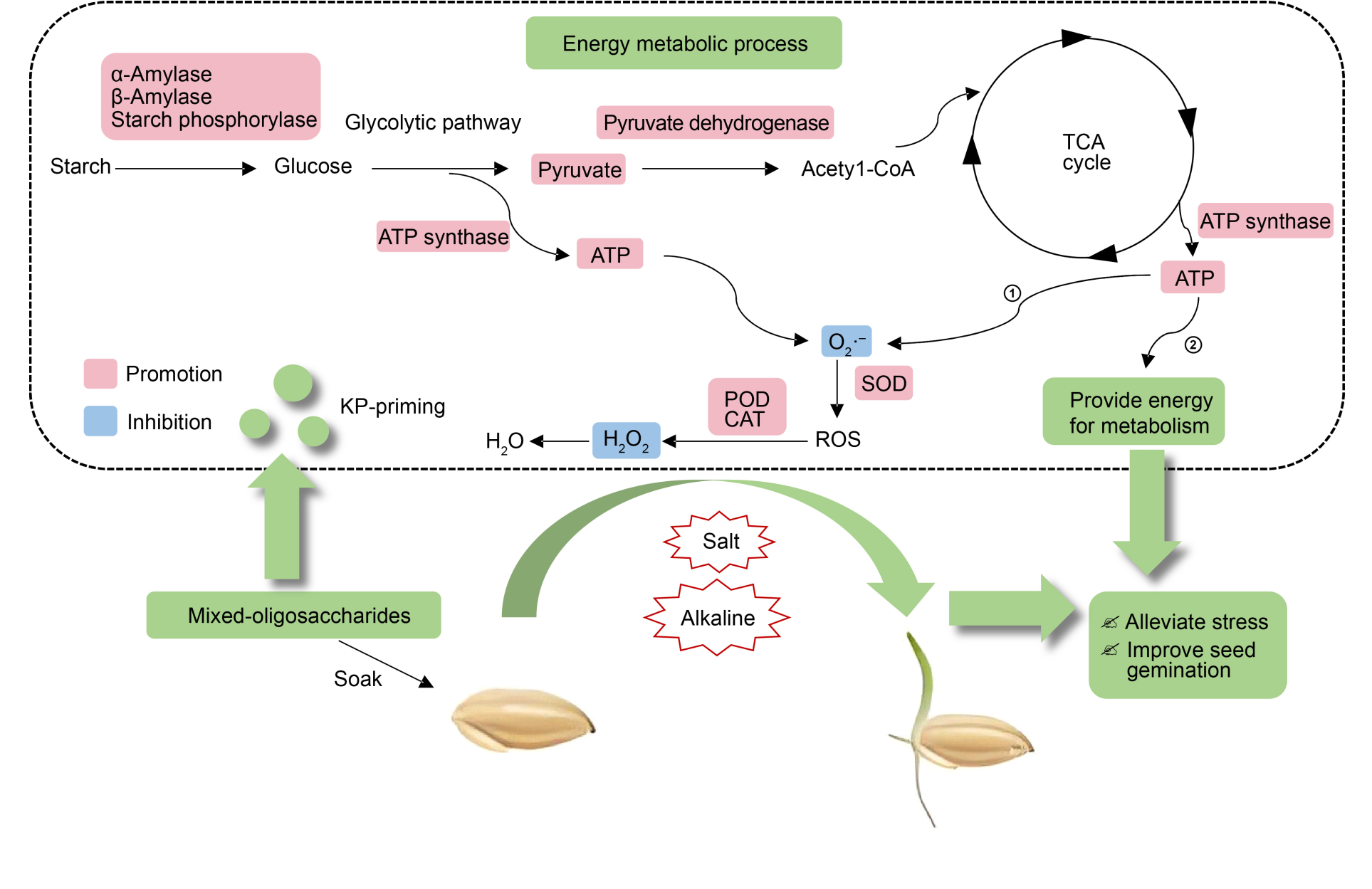
Fig. 5. Schematic diagram of alleviating stress and improving seed gemination of rice by which pre-soaking mixed-oligosaccharides (KP) influences energy metabolic process. TCA, Tricarboxylic acid cycle; SOD, Superoxide dismutase; POD, Peroxidases; CAT, Catalase; ROS, Reactive oxygen species.
| [1] | Acharya P, Jayaprakasha G K, Crosby K M, Jifon J L, Patil B S. 2020. Nanoparticle-mediated seed priming improves germination, growth, yield, and quality of watermelons (Citrullus lanatus) at multi-locations in texas. Sci Rep, 10(1): 5037. |
| [2] | Al-Quraan N A, AL-Ajlouni Z I, Obedat D I. 2019. The GABA shunt pathway in germinating seeds of wheat (Triticum aestivum L.) and barley (Hordeum vulgare L.) under salt stress. Seed Sci Res, 29(4): 250-260. |
| [3] | An Y, Gao Y, Tong S Z, Liu B. 2021. Morphological and physiological traits related to the response and adaption of Bolboschoenus planiculmis seedlings grown under salt-alkaline stress conditions. Front Plant Sci, 12: 567782. |
| [4] | Atwell B J, Greenway H, Colmer T D. 2015. Efficient use of energy in anoxia-tolerant plants with focus on germinating rice seedlings. New Phytol, 206(1): 36-56. |
| [5] | Bandehagh A, Taylor N L. 2020. Can alternative metabolic pathways and shunts overcome salinity induced inhibition of central carbon metabolism in crops? Front Plant Sci, 11: 1072. |
| [6] | Che-Othman M H, Jacoby R P, Millar A H, Taylor N L. 2020. Wheat mitochondrial respiration shifts from the tricarboxylic acid cycle to the GABA shunt under salt stress. New Phytol, 225(3): 1166-1180. |
| [7] | Corrêa-Ferreira M L, Viudes E B, de Magalhães A P, Paixão de Santana Filho P M, Sassaki G L, Pacheco A C, de Oliveira Petkowicz C L. 2019. Changes in the composition and structure of cell wall polysaccharides from Artemisia annua in response to salt stress. Carbohydr Res, 483: 107753. |
| [8] | Das P, Manna I, Sil P, Bandyopadhyay M, Biswas A K. 2021. Silicon augments salt tolerance through modulation of polyamine and GABA metabolism in two indica rice (Oryza sativa L.) cultivars. Plant Physiol Biochem, 166: 41-52. |
| [9] | El-Badri A M, Batool M, Wang C Y, Hashem A M, Tabl K M, Nishawy E, Kuai J, Zhou G S, Wang B. 2021. Selenium and zinc oxide nanoparticles modulate the molecular and morpho- physiological processes during seed germination of Brassica napus under salt stress. Ecotoxicol Environ Saf, 225: 112695. |
| [10] | Gebremeskel G, Gebremicael T G, Kifle M, Meresa E, Gebremedhin T, Girmay A. 2018. Salinization pattern and its spatial distribution in the irrigated agriculture of Northern Ethiopia: An integrated approach of quantitative and spatial analysis. Agric Water Manage, 206: 147-157. |
| [11] | He J X, Han W, Wang J, Qian Y C, Saito M, Bai W B, Song J Q, Lv G H. 2022. Functions of oligosaccharides in improving tomato seeding growth and chilling resistance. J Plant Growth Regul, 41(2): 535-545. |
| [12] | He J X, Kong M, Qian Y C, Gong M, Lv G H, Song J Q. 2023. Cellobiose elicits immunity in lettuce conferring resistance to Botrytis cinerea. J Exp Bot, 74(3): 1022-1038. |
| [13] | He M, Zhu C, Dong K, Zhang T, Cheng Z W, Li J R, Yan Y M. 2015. Comparative proteome analysis of embryo and endosperm reveals central differential expression proteins involved in wheat seed germination. BMC Plant Biol, 15: 97. |
| [14] | Huang Z B, Ying J F, Peng L L, Sun S, Huang C W, Li C, Wang Z F, He Y Q. 2021. A genome-wide association study reveals that the cytochrome b5 involved in seed reserve mobilization during seed germination in rice. Theor Appl Genet, 134(12): 4067-4076. |
| [15] | Jacoby R P, Millar A H, Taylor N L. 2013. Investigating the role of respiration in plant salinity tolerance by analyzing mitochondrial proteomes from wheat and a salinity-tolerant amphiploid (wheat × Lophopyrum elongatum). J Proteome Res, 12(11): 4807-4829. |
| [16] | Jiang C J, Liang Z W, Xie X Z. 2023. Priming for saline-alkaline tolerance in rice: Current knowledge and future challenges. Rice Sci, 30(5): 417-425. |
| [17] | Kong X Q, Luo Z, Zhang Y J, Li W J, Dong H Z. 2017. Soaking in H2O2 regulates ABA biosynthesis and GA catabolism in germinating cotton seeds under salt stress. Acta Physiol Plant, 39(1): 2. |
| [18] | Kosová K, Práil I T, Vítámvás P. 2013. Protein contribution to plant salinity response and tolerance acquisition. Int J Mol Sci, 14(4): 6757-6789. |
| [19] | Li J W, Liu J H, Zhao B P, Mi J Z, Wang J Y, Guo L C, Wang C L, Ren C Z. 2023. Differential analysis of oat leaf metabolism under salt and alkali stress. Pratac Sci, 40(10): 2607-2618. |
| [20] | Liu C X, Li C X, Bing H, Zhao J W, Li L, Sun P, Li T T, Du D D, Zhao J L, Wang X J, Xiang W S. 2023. Integrated physiological, transcriptomic, and metabolomic analysis reveals the mechanism of guvermectin promoting seed germination in direct-seeded rice under chilling stress. J Agric Food Chem, 71(19): 7348-7358. |
| [21] | Liu G X, Ouyang X, Li Z M, Fu Y Q, Shen H. 2022. Seaweed oligosaccharide synergistic silicate improves the resistance of rice plants to lodging stress under high nitrogen level. Agronomy, 12(8): 1750. |
| [22] | Lu X P, Min W F, Shi Y F, Tian L, Li P F, Ma T L, Zhang Y X, Luo C K. 2022. Exogenous melatonin alleviates alkaline stress by removing reactive oxygen species and promoting antioxidant defence in rice seedlings. Front Plant Sci, 13: 849553. |
| [23] | Lv Y S, Shao G N, Jiao G A, Sheng Z H, Xie L H, Hu S K, Tang S Q, Wei X J, Hu P S. 2021. Targeted mutagenesis of POLYAMINE OXIDASE 5 that negatively regulates mesocotyl elongation enables the generation of direct-seeding rice with improved grain yield. Mol Plant, 14(2): 344-351. |
| [24] | Ma J, Wang D H, She J, Li J M, Zhu J K, She Y M. 2016. Endoplasmic reticulum-associated N-glycan degradation of cold- upregulated glycoproteins in response to chilling stress in Arabidopsis. New Phytol, 212(1): 282-296. |
| [25] | McDowell G S, Gaun A, Steen H. 2013. iFASP: Combining isobaric mass tagging with filter-aided sample preparation. J Proteome Res, 12(8): 3809-3812. |
| [26] | Mohamed I A A, Shalby N, El-Badri A M, Batool M, Wang C Y, Wang Z K, Salah A, Rady M M, Jie K, Wang B, Zhou G S. 2022. RNA-seq analysis revealed key genes associated with salt tolerance in rapeseed germination through carbohydrate metabolism, hormone, and MAPK signaling pathways. Ind Crops Prod, 176: 114262. |
| [27] | Monajjem S, Soltani E, Zainali E, Esfahani M, Ghaderi-Far F, Chaleshtori M H, Rezaei A. 2023. Seed priming improves enzymatic and biochemical performances of rice during seed germination under low and high temperatures. Rice Sci, 30(4): 335-347. |
| [28] | Ojha R, Kaur S, Sinha K, Chawla K, Kaur S, Jadhav H, Kaur M, Bhunia R K. 2021. Characterization of oleosin genes from forage sorghum in Arabidopsis and yeast reveals their role in storage lipid stability. Planta, 254(5): 97. |
| [29] | Pring S, Kato H, Imano S, Camagna M, Tanaka A, Kimoto H, Chen P R, Shrotri A, Kobayashi H, Fukuoka A, Saito M, Suzuki T, Terauchi R, Sato I, Chiba S, Takemoto D. 2023. Induction of plant disease resistance by mixed oligosaccharide elicitors prepared from plant cell wall and crustacean shells. Physiol Plant, 175(5): e14052. |
| [30] | Pucciariello C. 2020. Molecular mechanisms supporting rice germination and coleoptile elongation under low oxygen. Plants, 9(8): 1037. |
| [31] | Rao M V, Paliyath G, Ormrod D P, Murr D P, Watkins C B. 1997. Influence of salicylic acid on H2O2 production, oxidative stress, and H2O2-metabolizing enzymes: Salicylic acid-mediated oxidative damage requires H2O2. Plant Physiol, 115(1): 137-149. |
| [32] | Sano N, Takebayashi Y, To A, Mhiri C, Rajjou L C, Nakagami H, Kanekatsu M. 2019. Shotgun proteomic analysis highlights the roles of long-lived mRNAs and de novo transcribed mRNAs in rice seeds upon imbibition. Plant Cell Physiol, 60(11): 2584-2596. |
| [33] | Shen Z, Li P, Ni R J, Ritchie M, Yang C P, Liu G F, Ma W, Liu G J, Ma L, Li S J, Wei Z G, Wang H X, Wang B C. 2009. Label-free quantitative proteomics analysis of etiolated maize seedling leaves during greening. Mol Cell Proteomics, 8(11): 2443-2460. |
| [34] | Singh A. 2021. Soil salinization management for sustainable development: A review. J Environ Manage, 277: 111383. |
| [35] | Song J, Shi W W, Liu R R, Xu Y G, Sui N, Zhou J C, Feng G. 2017. The role of the seed coat in adaptation of dimorphic seeds of the euhalophyte Suaeda salsa to salinity. Plant Species Biol, 32(2): 107-114. |
| [36] | Wang J, Zhou L, Shi H, Chern M, Yu H, Yi H, He M, Yin J J, Zhu X B, Li Y, Li W T, Liu J L, Wang J C, Chen X Q, Qing H, Wang Y P, Liu G F, Wang W M, Li P, Wu X J, Zhu L H, Zhou J M, Ronald P C, Li S G, Li J Y, Chen X W. 2018. A single transcription factor promotes both yield and immunity in rice. Science, 361: 1026-1028. |
| [37] | Wang N, Wang X R, Zhang H H, Liu X H, Shi J B, Dong Q, Xu Q H, Gui H P, Song M Z, Yan G T. 2021. Early ABA-stimulated maintenance of Cl-homeostasis by mepiquat chloride priming confers salt tolerance in cotton seeds. Crop J, 9(2): 387-399. |
| [38] | Wang N, Wang X R, Qi Q, Iqbal A, Zhang H H, Shi J B, Dong Q, Xu Q H, Liu X H, Gui H P, Song M Z, Zhang X L, Yan G T. 2022. Analysis of the effects of mepiquat chloride priming on the seedling growth-promoting in cotton under salt stress by multi-omics. Ind Crops Prod, 186: 115296. |
| [39] | Wang Q S, Zhou X, Liu Y, Han Y, Zuo J, Deng J, Yuan L Y, Gao L J, Bai W B. 2023. Mixed oligosaccharides-induced changes in bacterial assembly during cucumber (Cucumis sativus L.) growth. Front Microbiol, 14: 1195096. |
| [40] | Wang W H, Jing H F, Guo X X, Dou B Y, Zhang W S. 2023. Analysis of water and salt spatio-temporal distribution along irrigation canals in Ningxia Yellow River Irrigation Area, China. Sustainability, 15(16): 12114. |
| [41] | Wang Y G, Deng C Y, Liu Y, Niu Z R, Li Y. 2018. Identifying change in spatial accumulation of soil salinity in an inland river watershed, China. Sci Total Environ, 621: 177-185. |
| [42] | Yang X J, Chen Y Q, Hu Z C, Ma S, Zhang J E, Shen H. 2021. Alginate oligosaccharides alleviate the damage of rice leaves caused by acid rain and high temperature. Agronomy, 11(3): 500. |
| [43] | Yu Y C, Wang A M, Li X, Kou M, Wang W J, Chen X Y, Xu T, Zhu M K, Ma D F, Li Z Y, Sun J. 2018. Melatonin-stimulated triacylglycerol breakdown and energy turnover under salinity stress contributes to the maintenance of plasma membrane H+-ATPase activity and K+/Na+ homeostasis in sweet potato. Front Plant Sci, 9: 256. |
| [44] | Zhang N, Zhang H J, Sun Q Q, Cao Y Y, Li X S, Zhao B, Wu P, Guo Y D. 2017. Proteomic analysis reveals a role of melatonin in promoting cucumber seed germination under high salinity by regulating energy production. Sci Rep, 7(1): 503. |
| [45] | Zheng M M, Tao Y, Hussain S, Jiang Q W, Peng S B, Huang J L, Cui K H, Nie L X. 2016. Seed priming in dry direct-seeded rice: Consequences for emergence, seedling growth and associated metabolic events under drought stress. Plant Growth Regul, 78(2): 167-178. |
| [46] | Zhou M, Hassan M J, Peng Y, Liu L, Liu W, Zhang Y, Li Z. 2021. γ-Aminobutyric acid (GABA) priming improves seed germination and seedling stress tolerance associated with enhanced antioxidant metabolism, DREB expression, and dehydrin accumulation in white clover under water stress. Front Plant Sci, 12: 776939. |
| [47] | Zhou W, Guo Z, Chen J, Jiang J, Hui D F, Wang X, Sheng J, Chen L G, Luo Y Q, Zheng J C, Li S F, Zhang Y F. 2019. Direct seeding for rice production increased soil erosion and phosphorus runoff losses in subtropical China. Sci Total Environ, 695: 133845. |
| [48] | Zhou W G, Chen F, Zhao S H, Yang C Q, Meng Y J, Shuai H W, Luo X F, Dai Y J, Yin H, Du J B, Liu J, Fan G Q, Liu W G, Yang W Y, Shu K. 2019. DA-6 promotes germination and seedling establishment from aged soybean seeds by mediating fatty acid metabolism and glycometabolism. J Exp Bot, 70(1): 101-114. |
| [49] | Zhu J F, Yang X Y, Wu H W, Zhang H X. 2020. Research advances in salt and alkali tolerance improvement technology at the seed germination stage. Biotechnol Bull, 36(2): 158-168. |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||