
Rice Science ›› 2015, Vol. 22 ›› Issue (4): 171-179.DOI: 10.1016/S1672-6308(14)60295-X
收稿日期:2015-03-07
接受日期:2015-04-21
出版日期:2015-07-28
发布日期:2015-05-27
. [J]. Rice Science, 2015, 22(4): 171-179.
| M1 line | M2 line | Seed source | Mutagenesis | Phenotype of M3 | Single gene recessive mutation | Mutant designation |
| 6-1 | 6-1A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Nd | wsl3 |
| 6-1 | 6-1B | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy | Nd | |
| 7-17 | 7-17A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Yes | wsl4 |
| 11-39 | 11-39A | 2009 | 1 mmol/L NaN3 / 20 h presoak | Wet leaf/glossy | Nd | wsl5 |
| 26 | 26.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl6 |
| 264 | 264.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Nd | wsl7 |
| 524 | 524.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl8 |
| 680 | 680.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl9 |
| 843 | 843.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl10 |
| 1064 | 1064.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl11 |
| 1086 | 1086.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl12 |
| 1558 | 1558.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | |
| 1558 | 1558.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | wsl13 |
| Phenotypic ratios of segregating M3 populations were evaluated using Pearson’s Chi-square test for goodness-of-fit to the 3:1 (wild-type:mutant) ratio expected for single gene recessive mutations. Nd, Not determined; wsl, Wax crystal-sparse leaf. | ||||||
Table Genetic and physical characteristics of reduced epicuticular wax mutants.
| M1 line | M2 line | Seed source | Mutagenesis | Phenotype of M3 | Single gene recessive mutation | Mutant designation |
| 6-1 | 6-1A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Nd | wsl3 |
| 6-1 | 6-1B | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy | Nd | |
| 7-17 | 7-17A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Yes | wsl4 |
| 11-39 | 11-39A | 2009 | 1 mmol/L NaN3 / 20 h presoak | Wet leaf/glossy | Nd | wsl5 |
| 26 | 26.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl6 |
| 264 | 264.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Nd | wsl7 |
| 524 | 524.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl8 |
| 680 | 680.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl9 |
| 843 | 843.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl10 |
| 1064 | 1064.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl11 |
| 1086 | 1086.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl12 |
| 1558 | 1558.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | |
| 1558 | 1558.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | wsl13 |
| Phenotypic ratios of segregating M3 populations were evaluated using Pearson’s Chi-square test for goodness-of-fit to the 3:1 (wild-type:mutant) ratio expected for single gene recessive mutations. Nd, Not determined; wsl, Wax crystal-sparse leaf. | ||||||

Fig. 1. Wet leaf/glossy and intermediate-type mutant phenotypes. Leaves were subjected to a fine misting with water from a spray bottle. Water droplets adhere to the leaves of mutants and coalesce resulting in a wet, glossy appearance. A, Comparison of wild-type Sabine and the three mutants 6-1A, 7-17A, and 11-39A. Booting-stage flag leaves are shown; B, Comparison of the wild-type Sabine and the mutants 1558.2 and 6-1A. Fifth fully-expanded leaves from the base are shown. Unlike the other mutants, water droplets adhere to 1558.2 but do not coalesce and the leaves do not appear wet or glossy.
| Population | M3 seeds planted a | M3 plants phenotyped a | Segregation ratio (wild-type:mutant) | Pearson’s Chi-square test (3:1) | ||
| χ2 | P value | |||||
| 26.1 | 77 | 57 | 40:17 | 0.708 | 0.400 | |
| 680.2 | 77 | 61 | 43:18 | 0.661 | 0.416 | |
| 843.1 | 77 | 73 | 53:20 | 0.224 | 0.636 | |
| 524.2 | 95 | 68 | 53:15 | 0.314 | 0.575 | |
| a includes initial screening of M3 plants. | ||||||
Table 2 Segregation of wet leaf/glossyphenotype in four M3 populations.
| Population | M3 seeds planted a | M3 plants phenotyped a | Segregation ratio (wild-type:mutant) | Pearson’s Chi-square test (3:1) | ||
| χ2 | P value | |||||
| 26.1 | 77 | 57 | 40:17 | 0.708 | 0.400 | |
| 680.2 | 77 | 61 | 43:18 | 0.661 | 0.416 | |
| 843.1 | 77 | 73 | 53:20 | 0.224 | 0.636 | |
| 524.2 | 95 | 68 | 53:15 | 0.314 | 0.575 | |
| a includes initial screening of M3 plants. | ||||||
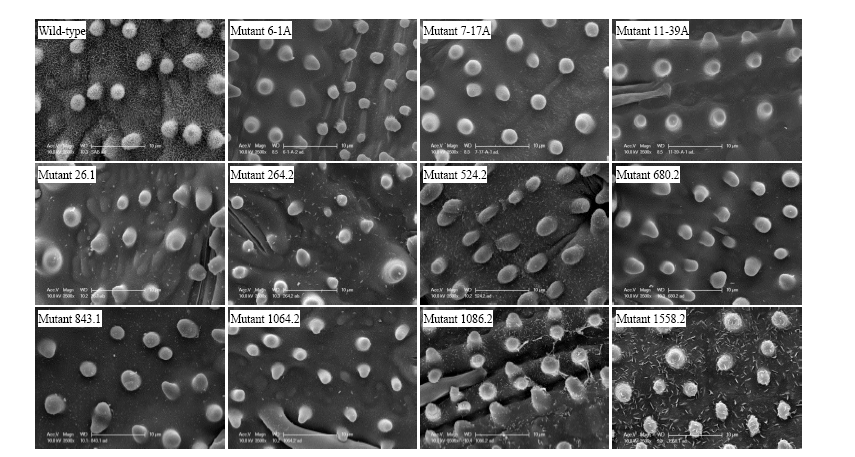
Fig. 2. Scanning electron micrographs of wild-type Sabine and 11 reduced epicuticular wax mutants.Adaxial leaf blade surface shown (knob-like structures are papillae). Scale bars, 10 μm.
| Line | Wax content (mg/g) | Reduction in wax content (%) |
| Sabine (wild-type) | 3.01 ± 0.49 | |
| 6-1A/B a | 0.91 ± 0.31 ** | 69.77 |
| 7-17A | 0.68 ± 0.42 ** | 77.41 |
| 11-39A | 1.38 ± 0.10 ** | 54.15 |
| Values are given as mean ± SD with four replicates except where noted. a, Due to insufficient tissue from 6-1A M3 mutants, additional samples were taken from 6-1B M3 mutants and content was determined using a total of five replicates (three from 6-1A plants, two from 6-1B plants included). ** means significant difference between mean values at P < 0.01 by t-test between the wild-type and each of the mutant line. | ||
Table 3 Total epicuticular wax content of wild-type and wsl mutant lines by weight method.
| Line | Wax content (mg/g) | Reduction in wax content (%) |
| Sabine (wild-type) | 3.01 ± 0.49 | |
| 6-1A/B a | 0.91 ± 0.31 ** | 69.77 |
| 7-17A | 0.68 ± 0.42 ** | 77.41 |
| 11-39A | 1.38 ± 0.10 ** | 54.15 |
| Values are given as mean ± SD with four replicates except where noted. a, Due to insufficient tissue from 6-1A M3 mutants, additional samples were taken from 6-1B M3 mutants and content was determined using a total of five replicates (three from 6-1A plants, two from 6-1B plants included). ** means significant difference between mean values at P < 0.01 by t-test between the wild-type and each of the mutant line. | ||
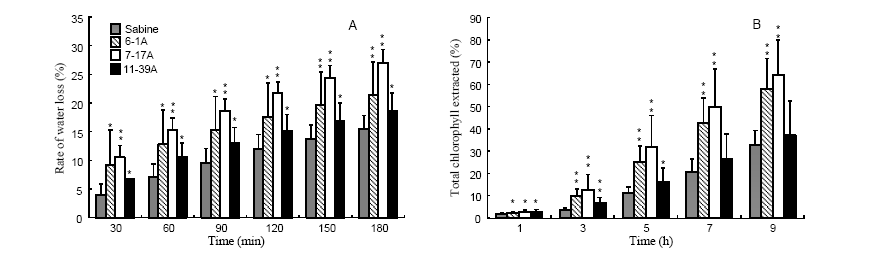
Fig. 3. Cuticle membrane permeability of three wax mutants. A, Water loss rate from detached leaves over time; B, Chlorophyll loss over time. Each bar represents the mean ± SD of two independent assays (two replicates for wild-type and four replicates for each mutant). Levels of significance between wild-type Sabine and each mutant were determined by t-test for unequal variance; * and ** mean significant difference from wild-type at P < 0.05 and P < 0.01, respectively.
| 1 | Abe A, Kosugi S, Yoshida K, Natsume S, Takagi H, Kanzaki H, Matsumura H, Yoshida K, Mitsuoka C, Tamiru M, Innan H, Cano L, Kamoun S, Terauchi R.2012. Genome sequencing reveals agronomically important loci in rice using MutMap.Nat Biotechnol, 30(2): 174-178. |
| 2 | Baker E A.1982. Chemistry and morphology of plant epicuticular waxes. In: Cutler D F, Alvin K L, Price C E. The Plant Cuticle. London: Academic Press: 139-165. |
| 3 | Clark J B, Lister G R.1975. Photosynthetic action spectra of trees: II. The relationship of cuticle structure to the visible and ultraviolet spectral properties of needles from four coniferous species.Plant Physiol, 55(2): 407-413. |
| 4 | Fiebig A, Mayfield J A, Miley N L, Chau S, Fischer R L, Preuss D.2000. Alterations in CER6, a gene identical to CUT1, differentially affect long-chain lipid content on the surface of pollen and stems.Plant Cell, 12(10): 2001-2008. |
| 5 | Islam M A, Du H, Ning J, Ye H Y, Xiong L Z.2009. Characterization of Glossy1-homologous genes in rice involved in leaf wax accumulation and drought resistance.Plant Mol Biol, 70(4): 443-456. |
| 6 | Jenks M A, Joly R J, Peters P J, Rich P J, Axtell J D, Ashworth E N.1994. Chemically induced cuticle mutation affecting epidermal conductance to water vapor and disease susceptibility in Sorghum bicolor (L.) Moench.Plant Physiol, 105(4): 1239-1245. |
| 7 | Jenks M A, Tuttle H A, Eigenbrode S D, Feldmann K A.1995. Leaf epicuticular waxes of the eceriferum mutant in Arabidopsis.Plant Physiol, 108: 369-377. |
| 8 | Jenks M A, Eigenbrode S D, Lemieux B.2002. Cuticular waxes of Arabidopsis. In: Somerville C R, Meyerowitz E M. The Arabidopsis book/American Society of Plant Biologists. |
| 9 | Jung K H, Han M J, Lee D Y, Lee Y S, Schreiber L, Franke R, Faust A, Yephremov A, Saedler H, Kim Y W, Hwang I, An G.2006. Wax-deficient anther1 is involved in cuticle and wax production in rice anther walls and is required for pollen development.Plant Cell, 18: 3015-3032. |
| 10 | Kerstiens G.1996. Cuticular water permeability and its physiological significance.J Exp Bot, 47(12): 1813-1832. |
| 11 | Lolle S J, Cheung A Y, Sussex I M.1992. Fiddlehead: An Arabidopsis mutant constitutively expressing an organ fusion program that involves interactions between epidermal cells.Dev Biol, 152: 383-392. |
| 12 | Lolle S J, Berlyn G P, Engstrom E M, Krolikowski K A, Reiter W D, Pruitt R E.1997. Developmental regulation of cell interactions in the Arabidopsis fiddlehead-1 mutant: A role for the epidermal cell wall and cuticle.Dev Biol, 189: 311-321. |
| 13 | Mao B G, Cheng Z J, Lei C L, Xu F H, Gao S W, Ren Y L, Wang J L, Zhang X, Wang J, Wu F Q, Guo X P, Liu X P, Wu C Y, Wang H Y, Wan J M.2012. Wax crystal-sparse leaf2, a rice homologue of WAX2/GL1, is involved in synthesis of leaf cuticular wax.Planta, 235: 39-52. |
| 14 | Nordström K J V, Albani M C, Velikkakam James G, Gutjahr C, Hartwig B, Turck F, Paszkowski U, Coupland G, Schneeberger K.2013. Mutation identification by direct comparison of whole-genome sequencing data from mutant and wild-type individuals using k-mers.Nat Biotechnol, 31(4): 325-330. |
| 15 | Preuss D, Lemieux B, Yen G, Davis R W.1993. A conditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signaling during fertilization.Genes Dev, 7(6): 974-985. |
| 16 | Post-Beittenmiller D.1996. Biochemistry and molecular biology of wax production in plants.Annu Rev Plant Physiol Plant Mol Biol, 47: 405-430. |
| 17 | Qin B X, Tang D, Huang J, Li M, Wu X R, Lu L L, Wang K J, Yu H X, Chen J M, Gu M H, Cheng Z K.2011. Rice OsGL1-1 is involved in leaf cuticular wax and cuticle membrane.Mol Plant, 4: 985-995. |
| 18 | Riederer M.2006. Biology of the Plant Cuticle. Oxford, Blackwell: 1-8. |
| 19 | Samuels L, Kunst L, Jetter R.2008. Sealing plant surfaces: Cutiicular wax formation by epidermal cells.Plant Biol, 59: 683-707. |
| 20 | Schneeberger K, Weigel D.2011. Fast-forward genetics enabled by new sequencing technologies.Trends Plant Sci, 16: 282-288. |
| 21 | Schneeberger K.2014. Using next-generation sequencing to isolate mutant genes from forward genetic screens.Nat Rev Genet, 15: 662-676. |
| 22 | Sieber P, Schorderet M, Ryser U, Buchala A, Kolattukudy P, Métraux J P, Nawrath C.2000. Transgenic Arabidopsis plants expressing a fungal cutinase show alterations in the structure and properties of the cuticle and postgenital organ fusions.Plant Cell, 12: 721-737. |
| 23 | Serrano M, Coluccia F, Torres M, L’Haridon F, Métraux J P.2014. The cuticle and plant defense to pathogens.Front Plant Sci, 5: 1-8. |
| 24 | Yeats T H, Rose J K C.2013. The formation and function of plant cuticles.Plant Physiol, 163: 5-20. |
| 25 | Yu D, Ranathunge K, Huang H, Pei Z, Franke R, Schreiber L, He C.2008. Wax crystal-sparse leaf1 encodes a β-ketoacyl CoA synthase involved in biosynthesis of cuticular waxes on rice leaf.Planta, 228: 675-685. |
| 26 | Zhou L Y, Ni E D, Yang J W, Zhou H, Liang H, Li J, Jiang D G, Wang Z H, Liu Z L, Zhuang C X.2013. Rice OsGL1-6 is involved in leaf cuticular wax accumulation and drought resistance.PLoS One, 8: e65139. |
| 27 | (Managing Editor: Wang Caihong) |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||