
Rice Science ›› 2024, Vol. 31 ›› Issue (5): 587-602.DOI: 10.1016/j.rsci.2024.02.007
收稿日期:2023-12-04
接受日期:2024-02-05
出版日期:2024-09-28
发布日期:2024-10-11
. [J]. Rice Science, 2024, 31(5): 587-602.
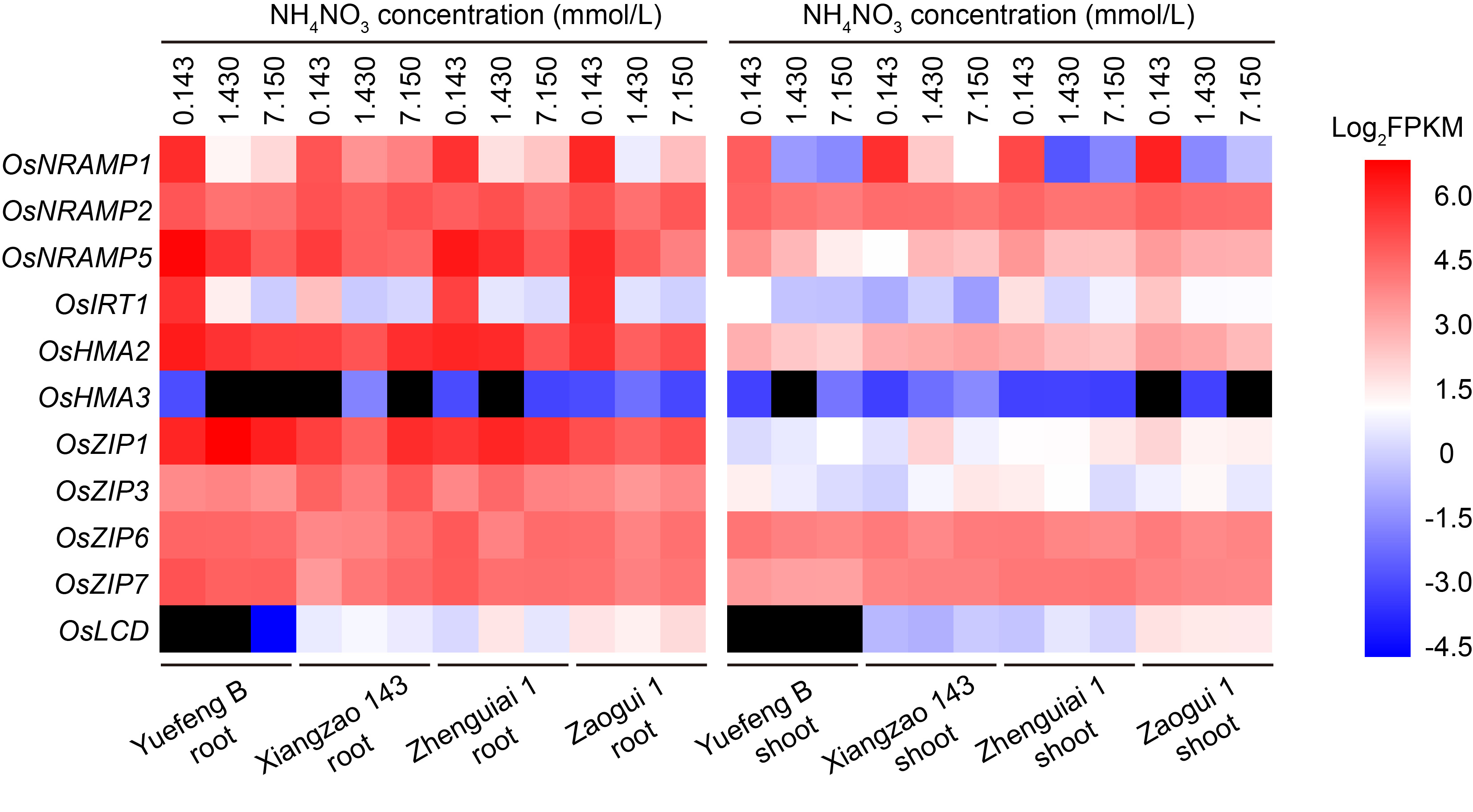
Fig. 1. Effects of different NH4NO3 dosages on expression of cadmium (Cd) absorption and transport-related genes in roots and shoots of four indica rice accession based on RNA-seq data. Gradient color blocks represent log2-transformed FPKM values. The black blocks indicate that no FPKM value is available. FPKM, Fragments per kilo bases per million fragments.
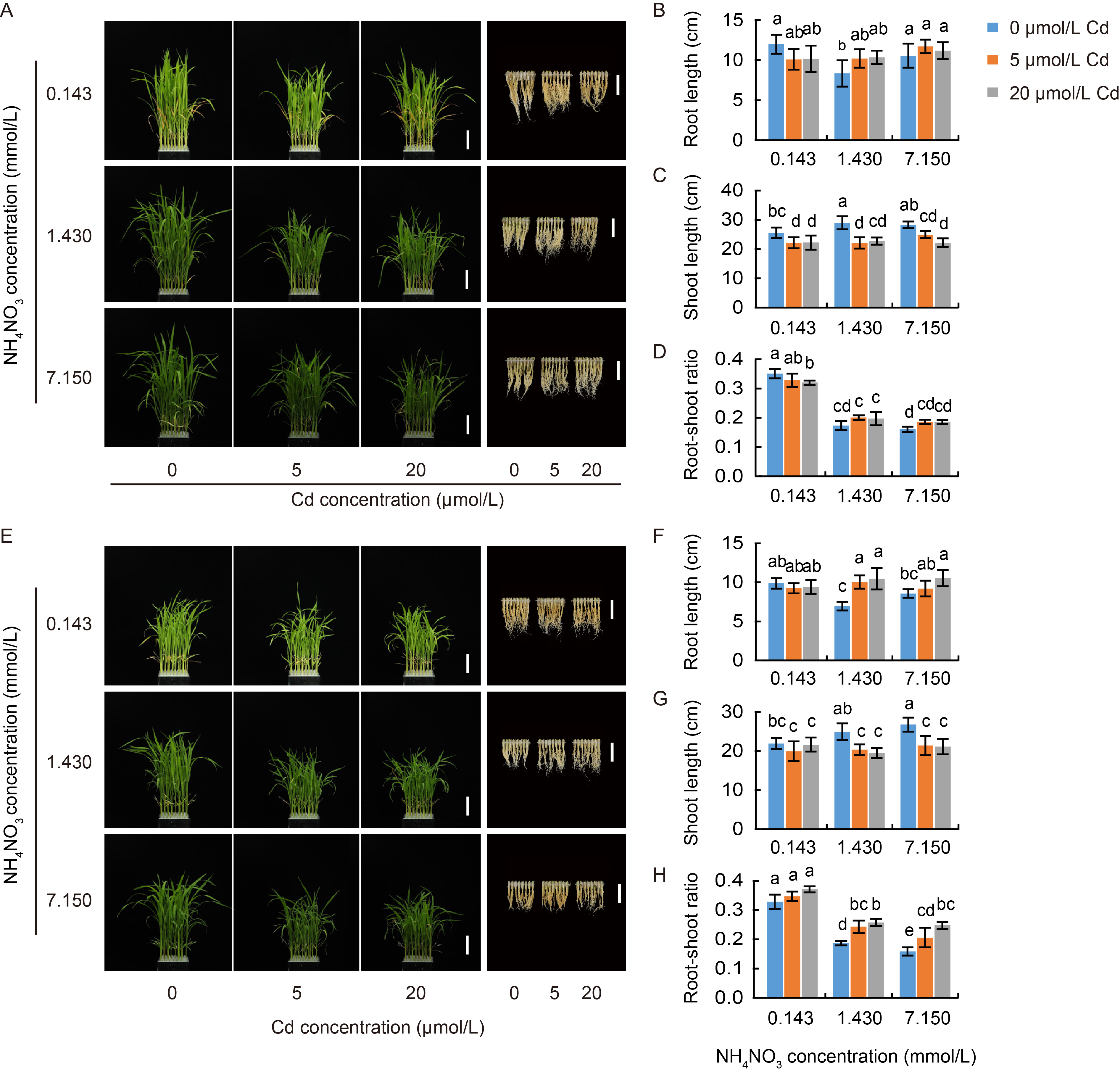
Fig. 2. Phenotypic characterization of N-sensitive Yuefeng B (A-D) and N-insensitive Zaogui 1 (E-H) rice seedlings grown under different cadmium (Cd) stress conditions with three NH4NO3 levels. A, Growth phenotypes of Yuefeng B treated with varying levels of Cd and NH4NO3 cultured outdoors from July to September 2023 in Hangzhou, China. B and C, Root length (B, n = 8) and shoot length (C, n = 8) of Yuefeng B, as shown in panel A. D, Root-shoot ratio (root dry weight / shoot dry weight, n = 4) of Yuefeng B, as shown in panel A. E, Growth phenotypes of Zaogui 1 treated with varying levels of Cd and NH4NO3, cultured outdoors from July to September 2023 in Hangzhou, China. F and G, Root length (F, n = 8) and shoot length (G, n = 8) of Zaogui 1, as shown in panel E. H, Root-shoot ratio (root dry weight / shoot dry weight, n = 4) of Zaogui 1, as shown in panel E. In A and E, the roots in the right panels are cropped and combined. Scale bars are 5 cm. In B-D and F-H, data are Mean ± SD. Different lowercase letters above the bars indicate significant differences at the P < 0.05 level by Tukey’s test following a one-way analysis of variance.

Fig. 3. Effects of three NH4NO3 levels on capacity of antioxidant enzyme system in N-sensitive Yuefeng B and N-insensitive Zaogui 1 rice seedlings under cadmium (Cd) stress. A-F, H2O2 content (A), malondialdehyde (MDA) content (B), superoxide dismutase (SOD) activity (C), peroxidase (POD) activity (D), catalase (CAT) activity (E), and ascorbate peroxidase (APX) activity (F) per gram of fresh shoots under 5 μmol/L Cd treatment. Data are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences at the P < 0.05 level by Tukey’s test following a one-way analysis of variance.
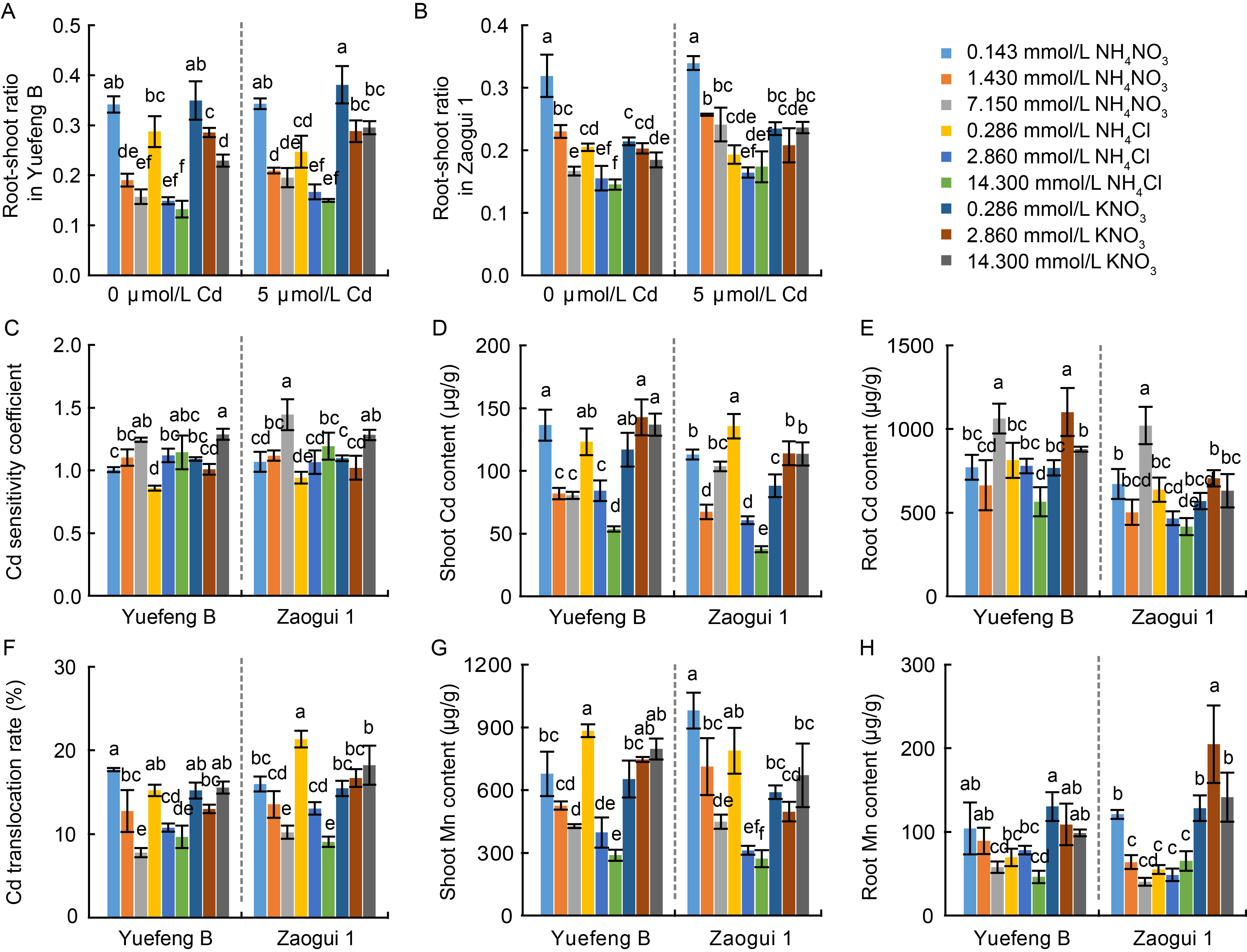
Fig. 4. Effects of different nitrogen forms and dosages on growth parameters, and cadmium (Cd) and manganese (Mn) content in N-sensitive Yuefeng B and N-insensitive Zaogui 1 rice seedlings. A and B, Root-shoot ratio (root dry weight / shoot dry weight, n = 3) of Yuefeng B (A) and Zaogui 1 (B) in the presence and absence of Cd treatment. C, Cd sensitivity coefficient (n = 3) for Yuefeng B and Zaogui 1 under 5 μmol/L Cd treatment. D and E, Cd content in shoots (D, n = 4) and roots (E, n = 4) per gram dry weight of rice seedlings under 5 μmol/L Cd treatment. F, Cd translocation rate (n = 4) in Yuefeng B and Zaogui 1 under 5 μmol/L Cd treatment. G and H, Mn content in shoots (G, n = 4) and roots (H, n = 4) per gram dry weight of rice seedlings under 5 μmol/L Cd treatment. Data are Mean ± SD. Different lowercase letters above the bars indicate significant differences at the P < 0.05 level by Tukey’s test following a one-way analysis of variance.
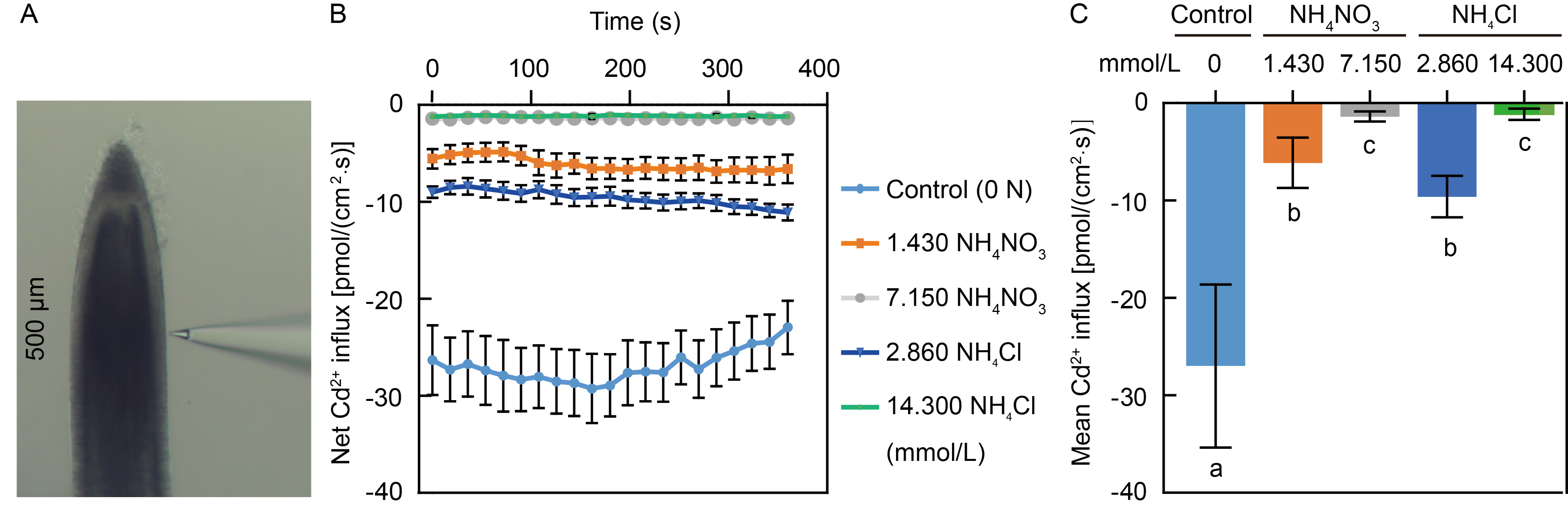
Fig. 5. Effects of NH4NO3 and NH4Cl on instantaneous uptake of cadmium ions (Cd2+) in N-sensitive Yuefeng B rice seedlings. A, Location of net Cd2+ flux detection at the elongation zone of rice roots (500 μm from the root tip). B, Net Cd2+ influx in Yuefeng B roots over time under different instantaneous treatments with NH4NO3 or NH4Cl. Data are Mean ± SE (n = 10). C, Mean Cd2+ influx in Yuefeng B roots as shown in panel B. Data are Mean ± SD (n = 10). Different lowercase letters indicate significant differences at the P < 0.05 level by Tukey’s test following to a one-way analysis of variance.
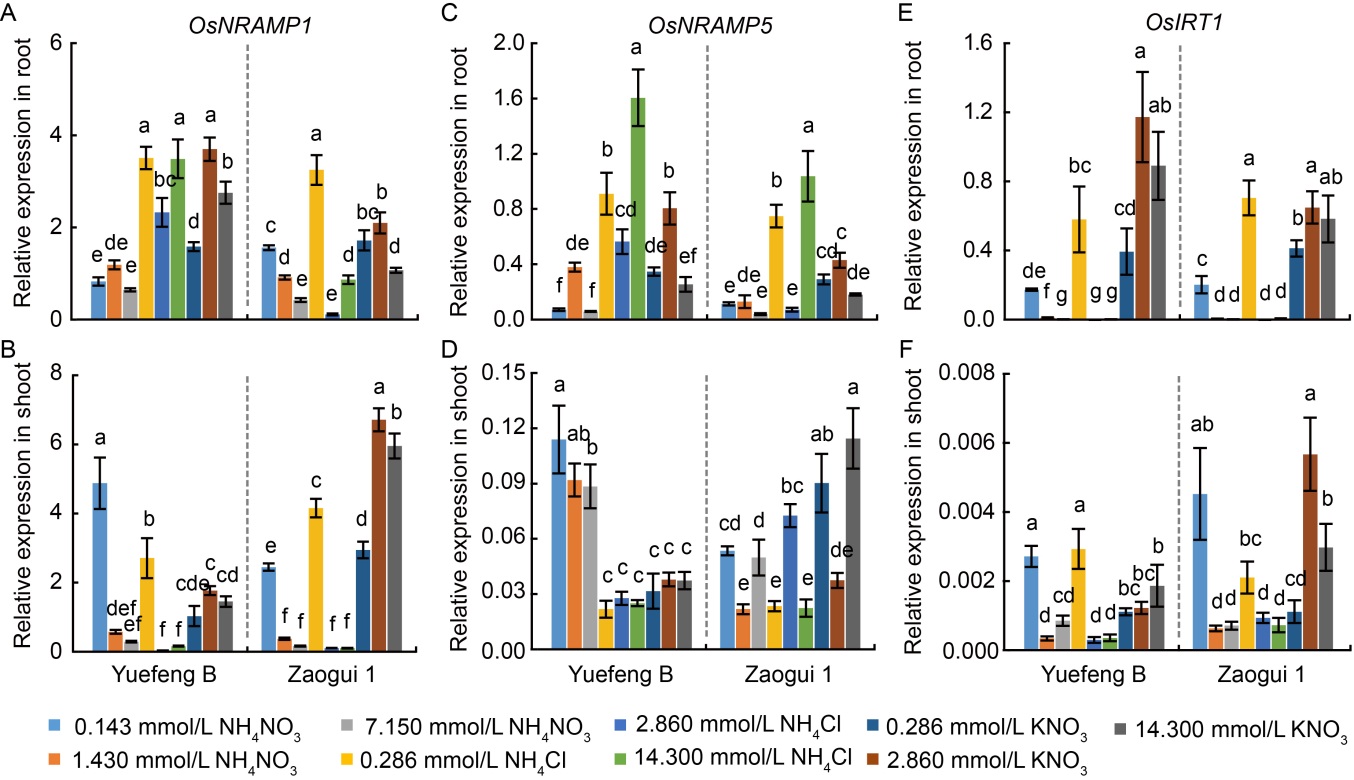
Fig. 6. Effects of different nitrogen forms and dosages on expression of Cd transport-related genes in cadmium (Cd)-stressed N-sensitive Yuefeng B and N-insensitive Zaogui 1 rice seedlings. A-F, Effects of various N forms on OsNRAMP1 expression in roots (A), OsNRAMP1 expression in shoots (B), OsNRAMP5 expression in roots (C), OsNRAMP5 expression in shoots (D), OsIRT1 expression in roots (E), and OsIRT1 expression in shoots (F) under 5 μmol/L Cd treatment. Transcripts of Cd transport-related genes are quantified from four replicates using the 2−∆CT method with OsACT2 as the reference gene. Data are Mean ± SD (n = 4). Different lowercase letters above the bars indicate significant differences at the P < 0.05 level by Tukey’s test following to a one-way analysis of variance.
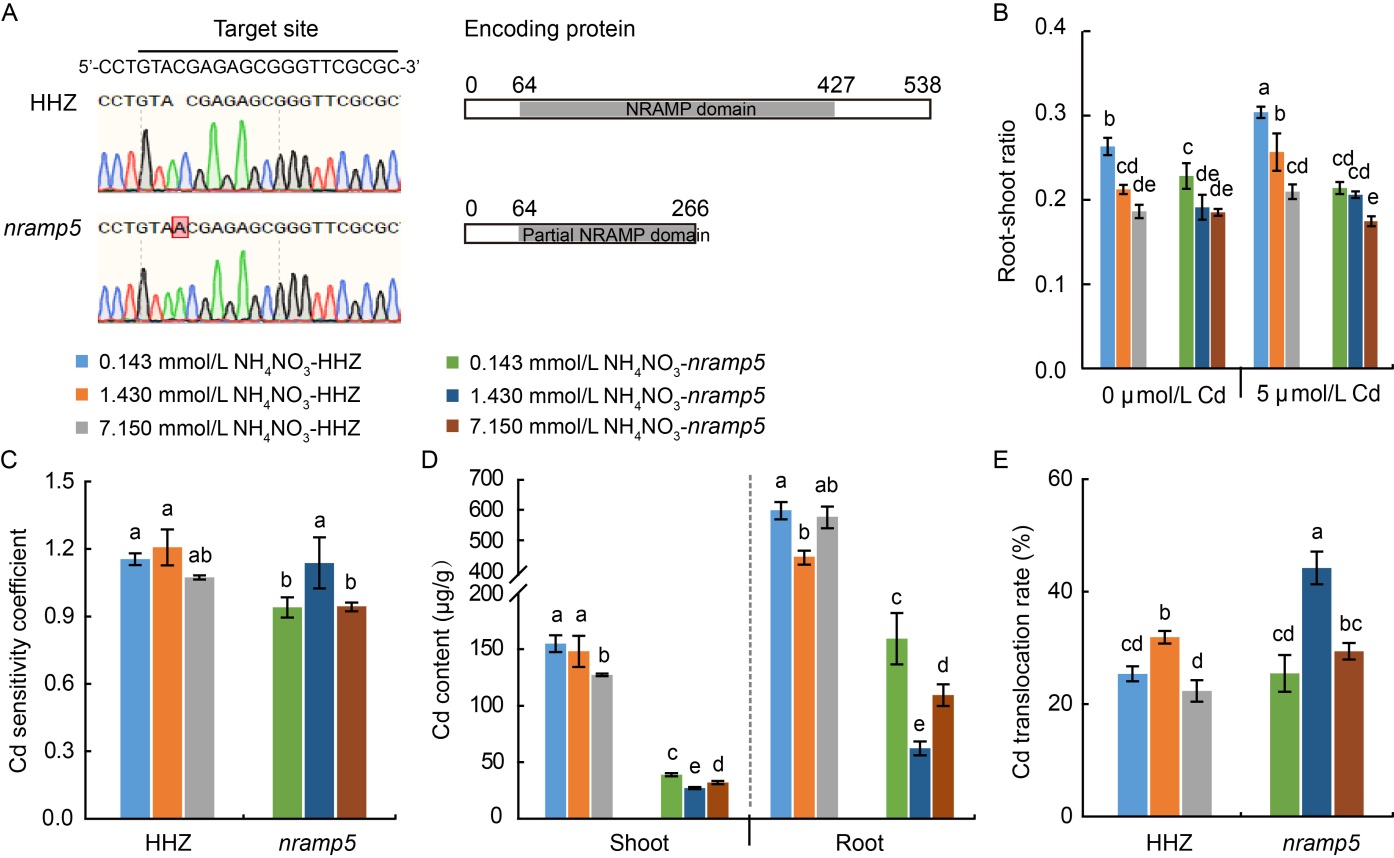
Fig. 7. Effects of three NH4NO3 levels on growth parameters and cadmium (Cd) concentration in nramp5 mutant and its wild type Huanghuazhan (HHZ). A, Mutation site in nramp5. B, Root-shoot ratio (root dry weight / shoot dry weight) of HHZ and nramp5 in the presence and absence of Cd. C, Cd sensitivity coefficient for HHZ and nramp5. D, Cd content per gram shoot or root dry weight under 5 μmol/L Cd treatment. E, Cd translocation rate under 5 μmol/L Cd treatment. Data are Mean ± SD (n = 4). Different lowercase letters above the bars indicate significant differences at the P < 0.05 level by Tukey’s test following a one-way analysis of variance.
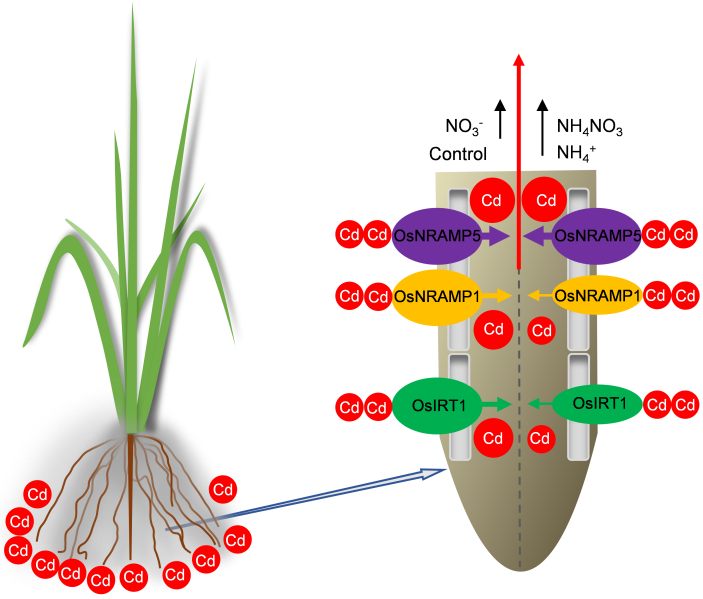
Fig. 8. Schematic diagram illustrating possible mechanism, by which NH4NO3 and NH4+-N alleviate cadmium (Cd) content. Compared with the control or with an increased supply of NO3-, increasing the supply of NH4NO3 and NH4+ decreased the gene expression of OsNRAMP1 and OsIRT1, thereby inhibiting Cd uptake and transport in roots, and ultimately reducing the Cd content in shoots. The red upward arrow represents the transfer of Cd from the root to shoot. The black upward arrow indicates an increased N dosage. The size of oval represents the corresponding gene expression level. The thickness of the arrow on the transporter indicates the flux of Cd absorption and transport. The size of red circle in the root represents the relative concentration of Cd.
| [1] | Adeosun A A, Price A H, Norton G J. 2023. Cadmium tolerance and accumulation in wild rice species. Rice Sci, 30(3): 181-185. |
| [2] | Åkesson A, Barregard L, Bergdahl I A, Nordberg G F, Nordberg M, Skerfving S. 2014. Non-renal effects and the risk assessment of environmental cadmium exposure. Environ Health Perspect, 122(5): 431-438. |
| [3] |
Anders S, Pyl P T, Huber W. 2015. HTSeq: A python framework to work with high-throughput sequencing data. Bioinformatics, 31(2): 166-169.
DOI PMID |
| [4] | Bai Z Q, Zhu L, Chang H X, Wu J W. 2021a. Enhancement of cadmium accumulation in sweet sorghum as affected by nitrate. Plant Biol, 23(1): 66-73. |
| [5] | Bai Z Q, Li D, Zhu L, Tang X Y, Wang Y F, Mao R J, Wu J W. 2021b. Nitrate increases cadmium accumulation in sweet sorghum for improving phytoextraction efficiency rather than ammonium. Front Plant Sci, 12: 643116. |
| [6] | Chang J D, Huang S, Yamaji N, Zhang W W, Ma J F, Zhao F J. 2020. OsNRAMP1 transporter contributes to cadmium and manganese uptake in rice. Plant Cell Environ, 43(10): 2476-2491. |
| [7] | Chanh T T, Tsutsumi M, Kurihara K. 1981. Comparative study on the response of indica and japonica rice plants to ammonium and nitrate nitrogen. Soil Sci Plant Nutr, 27(1): 83-92. |
| [8] | Chen A L, Liu T, Deng Y, Xiao R, Zhang T, Wang Y, Yang Y H, Lakshmanan P, Shi X J, Zhang F S, Chen X P. 2023. Nitrate-dependent suberization regulates cadmium uptake and accumulation in maize. Sci Total Environ, 878: 162848. |
| [9] | Chen J G, Zou W L, Meng L J, Fan X R, Xu G H, Ye G Y. 2019. Advances in the uptake and transport mechanisms and QTLs mapping of cadmium in rice. Int J Mol Sci, 20(14): 3417. |
| [10] |
Cheng M M, Wang P, Kopittke P M, Wang A N, Sale P W G, Tang C X. 2016. Cadmium accumulation is enhanced by ammonium compared to nitrate in two hyperaccumulators, without affecting speciation. J Exp Bot, 67(17): 5041-5050.
DOI PMID |
| [11] | Cheng M M, Wang A N, Tang C X. 2017. Ammonium-based fertilizers enhance Cd accumulation in Carpobrotus rossii grown in two soils differing in pH. Chemosphere, 188: 689-696. |
| [12] |
Clemens S, Aarts M G M, Thomine S, Verbruggen N. 2013. Plant science: The key to preventing slow cadmium poisoning. Trends Plant Sci, 18(2): 92-99.
DOI PMID |
| [13] | Cramer M D, Lewis O A M. 1993. The influence of NO3- and NH4+ nutrition on the carbon and nitrogen partitioning characteristics of wheat (Triticum aestivum L.) and maize (Zea mays L.) plants. Plant Soil, 154(2): 289-300. |
| [14] |
Ding Y F, Ye Y Y, Jiang Z H, Wang Y, Zhu C. 2016. MicroRNA390 is involved in cadmium tolerance and accumulation in rice. Front Plant Sci, 7: 235.
DOI PMID |
| [15] |
Du Q, Chen M X, Zhou R, Chao Z Y, Zhu Z W, Shao G S, Wang G M. 2009. Cd toxicity and accumulation in rice plants vary with soil nitrogen status and their genotypic difference can be partly attributed to nitrogen uptake capacity. Rice Sci, 16(4): 283-291.
DOI |
| [16] | Eriksson J E. 1990. Effects of nitrogen-containing fertilizers on solubility and plant uptake of cadmium. Water Air Soil Pollut, 49(3): 355-368. |
| [17] | Guo S, Zhou Y, Shen Q, Zhang F. 2007. Effect of ammonium and nitrate nutrition on some physiological processes in higher plants-growth, photosynthesis, photorespiration, and water relations. Plant Biol, 9(1): 21-29. |
| [18] |
Hasan S A, Fariduddin Q, Ali B, Hayat S, Ahmad A. 2009. Cadmium: Toxicity and tolerance in plants. J Environ Biol, 30(2): 165-174.
PMID |
| [19] | Hassan M J, Wang F, Ali S, Zhang G P. 2005. Toxic effect of cadmium on rice as affected by nitrogen fertilizer form. Plant Soil, 277(1): 359-365. |
| [20] | Hassan M J, Shafi M, Zhang G P, Zhu Z J, Qaisar M. 2008a. The growth and some physiological responses of rice to Cd toxicity as affected by nitrogen form. Plant Growth Regul, 54(2): 125-132. |
| [21] | Hassan M J, Zhang G P, Zhu Z J. 2008b. Influence of cadmium toxicity on plant growth and nitrogen uptake in rice as affected by nitrogen form. J Plant Nutr, 31(2): 251-262. |
| [22] | Hu P J, Yin Y G, Ishikawa S, Suzui N, Kawachi N, Fujimaki S, Igura M, Yuan C, Huang J X, Li Z, Makino T, Luo Y M, Christie P, Wu L H. 2013. Nitrate facilitates cadmium uptake, transport and accumulation in the hyperaccumulator Sedum plumbizincicola. Environ Sci Pollut Res Int, 20(9): 6306-6316. |
| [23] | Jalloh M A, Chen J H, Zhen F R, Zhang G P. 2009. Effect of different N fertilizer forms on antioxidant capacity and grain yield of rice growing under Cd stress. J Hazard Mater, 162(2/3): 1081-1085. |
| [24] | Järup L, Akesson A. 2009. Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol, 238(3): 201-208. |
| [25] | Jiang L L, Wu L, Wang Y, Xu Q, Xu Z J, Chen W F. 2022. Research progress on the divergence and genetic basis of agronomic traits in Xian and Geng rice. Crop J, 10(4): 924-931. |
| [26] |
Kim D, Langmead B, Salzberg S L. 2015. HISAT: A fast spliced aligner with low memory requirements. Nat Methods, 12(4): 357-360.
DOI PMID |
| [27] | Larsson Jönsson E H, Asp H. 2013. Effects of pH and nitrogen on cadmium uptake in potato. Biol Plant, 57(4): 788-792. |
| [28] | Lee S, An G. 2009. Over-expression of OsIRT1 leads to increased iron and zinc accumulations in rice. Plant Cell Environ, 32(4): 408-416. |
| [29] | Li Q, Lu X L, Wang C J, Shen L, Dai L P, He J L, Yang L, Li P Y, Hong Y F, Zhang Q, Dong G J, Hu J, Zhang G H, Ren D Y, Gao Z Y, Guo L B, Qian Q, Zhu L, Zeng D L. 2022. Genome-wide association study and transcriptome analysis reveal new QTL and candidate genes for nitrogen-deficiency tolerance in rice. Crop J, 10(4): 942-951. |
| [30] | Lin Y L, Chao Y Y, Huang W D, Kao C H. 2011. Effect of nitrogen deficiency on antioxidant status and Cd toxicity in rice seedlings. Plant Growth Regul, 64(3): 263-273. |
| [31] | Liu Z Y, Zhu C S, Jiang Y, Tian Y L, Yu J, An H Z, Tang W J, Sun J, Tang J P, Chen G M, Zhai H Q, Wang C M, Wan J M. 2016. Association mapping and genetic dissection of nitrogen use efficiency-related traits in rice (Oryza sativa L.). Funct Integr Genomic, 16(3): 323-333. |
| [32] |
Luo B F, Du S T, Lu K X, Liu W J, Lin X Y, Jin C W. 2012. Iron uptake system mediates nitrate-facilitated cadmium accumulation in tomato (Solanum lycopersicum) plants. J Exp Bot, 63(8): 3127-3136.
DOI PMID |
| [33] | Maier N A, McLaughlin M J, Heap M, Butt M, Smart M K. 2002. Effect of nitrogen source and calcitic lime on soil pH and potato yield, leaf chemical composition, and tuber cadmium concentrations. J Plant Nutr, 25(3): 523-544. |
| [34] | Morishita T, Fumoto N, Yoshizawa T, Kagawa K. 1987. Varietal differences in cadmium levels of rice grains of japonica, indica, Javanica, and hybrid varieties produced in the same plot of a field. Soil Sci Plant Nutr, 33(4): 629-637. |
| [35] |
Nawrot T S, Staessen J A, Roels H A, Munters E, Cuypers A, Richart T, Ruttens A, Smeets K, Clijsters H, Vangronsveld J. 2010. Cadmium exposure in the population: From health risks to strategies of prevention. Biometals, 23(5): 769-782.
DOI PMID |
| [36] | Nordberg G F. 2009. Historical perspectives on cadmium toxicology. Toxicol Appl Pharmacol, 238(3): 192-200. |
| [37] | Rizwan M, Ali S, Adrees M, Rizvi H, Zia-Ur-Rehman M, Hannan F, Qayyum M F, Hafeez F, Ok Y S. 2016. Cadmium stress in rice: Toxic effects, tolerance mechanisms, and management: A critical review. Environ Sci Pollut Res Int, 23(18): 17859-17879. |
| [38] | Sasaki A, Yamaji N, Yokosho K, Ma J F. 2012. Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell, 24(5): 2155-2167. |
| [39] | Satarug S, Garrett S H, Sens M A, Sens D A. 2010. Cadmium, environmental exposure, and health outcomes. Environ Health Perspect, 118(2): 182-190. |
| [40] | Song Y, Wang Y, Mao W F, Sui H X, Yong L, Yang D J, Jiang D G, Zhang L, Gong Y Y. 2017. Dietary cadmium exposure assessment among the Chinese population. PLoS One, 12(5): e0177978. |
| [41] |
Takahashi R, Ishimaru Y, Senoura T, Shimo H, Ishikawa S, Arao T, Nakanishi H, Nishizawa N K. 2011. The OsNRAMP1 iron transporter is involved in Cd accumulation in rice. J Exp Bot, 62(14): 4843-4850.
DOI PMID |
| [42] | Tang W J, Ye J, Yao X M, Zhao P Z, Xuan W, Tian Y L, Zhang Y Y, Xu S, An H Z, Chen G M, Yu J, Wu W, Ge Y W, Liu X L, Li J, Zhang H Z, Zhao Y Q, Yang B, Jiang X Z, Peng C, Zhou C, Terzaghi W, Wang C M, Wan J M. 2019. Genome-wide associated study identifies NAC42-activated nitrate transporter conferring high nitrogen use efficiency in rice. Nat Commun, 10(1): 5279. |
| [43] |
Tsadilas C D, Karaivazoglou N A, Tsotsolis N C, Stamatiadis S, Samaras V. 2005. Cadmium uptake by tobacco as affected by liming, N form, and year of cultivation. Environ Pollut, 134(2): 239-246.
DOI PMID |
| [44] | Uraguchi S, Fujiwara T. 2012. Cadmium transport and tolerance in rice: Perspectives for reducing grain cadmium accumulation. Rice, 5(1): 5. |
| [45] | Vazquez A, Recalde L, Cabrera A, Groppa M D, Benavides M P. 2020. Does nitrogen source influence cadmium distribution in Arabidopsis plants? Ecotoxicol Environ Saf, 191: 110163. |
| [46] | Wang J F, Li W L, Li Q S, Wang L L, He T, Wang F P, Xu Z M. 2021. Nitrogen fertilizer management affects remobilization of the immobilized cadmium in soil and its accumulation in crop tissues. Environ Sci Pollut Res Int, 28(24): 31640-31652. |
| [47] | Wang T K, Li Y X, Fu Y F, Xie H J, Song S F, Qiu M D, Wen J, Chen M W, Chen G, Tian Y, Li C X, Yuan D Y, Wang J L, Li L. 2019. Mutation at different sites of metal transporter gene OsNramp5 affects Cd accumulation and related agronomic traits in rice (Oryza sativa L.). Front Plant Sci, 10: 1081. |
| [48] | Wångstrand H, Eriksson J, Öborn I. 2007. Cadmium concentration in winter wheat as affected by nitrogen fertilization. Eur J Agron, 26(3): 209-214. |
| [49] | Wu Z C, Zhang W J, Xu S J, Shi H Z, Wen D, Huang Y D, Peng L J, Deng T H B, Du R Y, Li F R, Wang X, Wang F H. 2018. Increasing ammonium nutrition as a strategy for inhibition of cadmium uptake and xylem transport in rice (Oryza sativa L.) exposed to cadmium stress. Environ Exp Bot, 155: 734-741. |
| [50] | Xie H L, Jiang R F, Zhang F S, McGrath S P, Zhao F J. 2009. Effect of nitrogen form on the rhizosphere dynamics and uptake of cadmium and zinc by the hyperaccumulator Thlaspi caerulescens. Plant Soil, 318(1): 205-215. |
| [51] | Yan H L, Xu W X, Xie J Y, Gao Y W, Wu L L, Sun L, Feng L, Chen X, Zhang T, Dai C H, Li T, Lin X N, Zhang Z Y, Wang X Q, Li F M, Zhu X Y, Li J J, Li Z C, Chen C Y, Ma M, Zhang H L, He Z Y. 2019. Variation of a major facilitator superfamily gene contributes to differential cadmium accumulation between rice subspecies. Nat Commun, 10(1): 2562. |
| [52] | Yang Y J, Xiong J, Chen R J, Fu G F, Chen T T, Tao L X. 2016. Excessive nitrate enhances cadmium (Cd) uptake by up-regulating the expression of OsIRT1 in rice (Oryza sativa). Environ Exp Bot, 122: 141-149. |
| [53] | Zaccheo P, Crippa L, Di Muzio Pasta V. 2006. Ammonium nutrition as a strategy for cadmium mobilisation in the rhizosphere of sunflower. Plant Soil, 283(1): 43-56. |
| [54] | Zeng X Y, Zou D S, Wang A D, Zhou Y Y, Liu Y H, Li Z H, Liu F, Wang H, Zeng Q R, Xiao Z H. 2020. Remediation of cadmium- contaminated soils using Brassica napus: Effect of nitrogen fertilizers. J Environ Manage, 255: 109885. |
| [55] | Zhang J L, Zhu Y C, Yu L J, Yang M, Zou X, Yin C X, Lin Y J. 2022. Research advances in cadmium uptake, transport and resistance in rice (Oryza sativa L.). Cells, 11(3): 569. |
| [56] | Zhang L D, Liu X, Wei M Y, Guo Z J, Zhao Z Z, Gao C H, Li J, Xu J X, Shen Z J, Zheng H L. 2022. Ammonium has stronger Cd detoxification ability than nitrate by reducing Cd influx and increasing Cd fixation in Solanum nigrum L. J Hazard Mater, 425: 127947. |
| [57] | Zhang Z H, Chu C C. 2020. Nitrogen-use divergence between indica and japonica rice: Variation at nitrate assimilation. Mol Plant, 13(1): 6-7. |
| [58] | Zhao J L, Yang W, Zhang S H, Yang T F, Liu Q, Dong J F, Fu H, Mao X X, Liu B. 2018. Genome-wide association study and candidate gene analysis of rice cadmium accumulation in grain in a diverse rice collection. Rice, 11(1): 61. |
| [59] | Zhu C Q, Cao X C, Zhu L F, Hu W J, Hu A Y, Bai Z G, Zhong C, Sun L M, Liang Q D, Huang J, Yang S X, Zhang J H, Jin Q Y. 2018. Ammonium mitigates Cd toxicity in rice (Oryza sativa) via putrescine-dependent alterations of cell wall composition. Plant Physiol Biochem, 132: 189-201. |
| [60] | Zhu D F, Zhang Y P, Chen H Z, Xiang J, Zhang Y K. 2015. Innovation and practice of high-yield rice cultivation technology in China. Sci Agric Sin, 48(17): 3404-3414. (in Chinese with English abstract) |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||