
Rice Science ›› 2022, Vol. 29 ›› Issue (3): 268-276.DOI: 10.1016/j.rsci.2021.09.004
• Research Paper • Previous Articles Next Articles
Wang Chenjiaozi, Zhao Mei, Shu Canwei, Zhou Erxun( )
)
Received:2021-07-12
Accepted:2021-09-17
Online:2022-05-28
Published:2022-03-10
Contact:
Zhou Erxun
Wang Chenjiaozi, Zhao Mei, Shu Canwei, Zhou Erxun. Three Genes Related to Trehalose Metabolism Affect Sclerotial Development of Rhizoctonia solani AG-1 IA, Causal Agent of Rice Sheath Blight[J]. Rice Science, 2022, 29(3): 268-276.
Add to citation manager EndNote|Ris|BibTeX
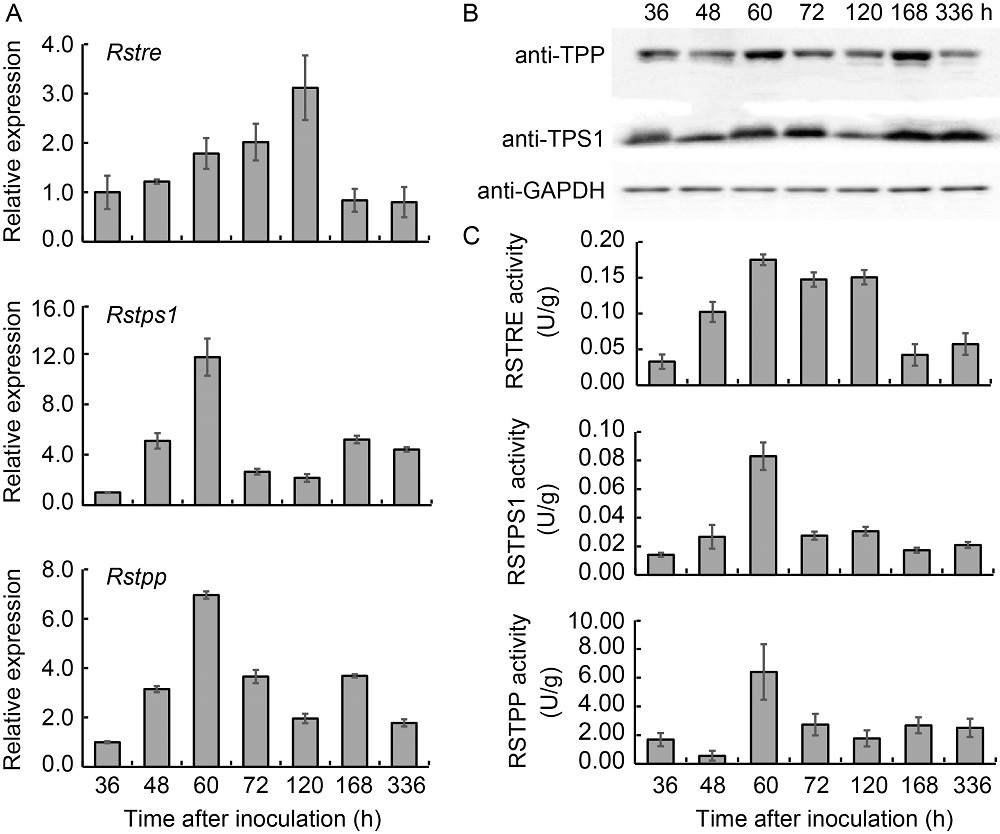
Fig. 1. Gene relative expression, Western blot and enzyme activities of Rstre, Rstps1 and Rstpp in different developmental stages of sclerotia in Rhizoctonia solani AG-1 IA. A, Relative expression levels of Rstre, Rstps1 and Rstpp at different sclerotial development stages. Samples were collected at seven different time points (36, 48, 60, 72, 120, 168 and 336 h of strain culture) during the sclerotial development. Data were normalized to transcripts encoding GAPDH, and shown as Mean ± SE (n = 3). B, Western blot of RSTPS1 and RSTPP at different sclerotial development stages. Anti- GAPDH was used as an internal control. C, Enzyme activities of RSTRE, RSTPS1 and RSTPP at different sclerotial development stages. Data are Mean ± SE (n = 3).
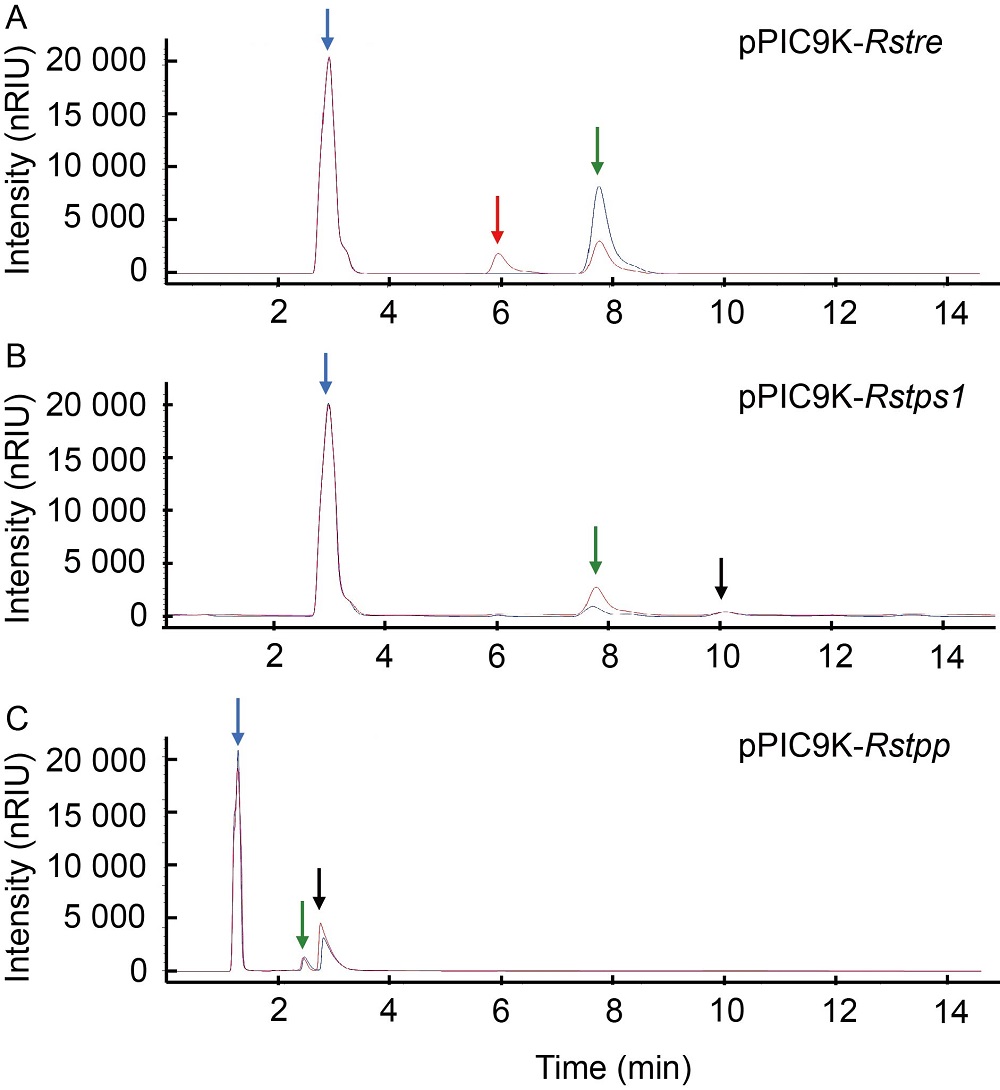
Fig. 2. Functional verification of Rstre (A), Rstps1 (B) and Rstpp (C) in Pichia pastoris GS115. pPIC9K-Rstre, pPIC9K-Rstps1 and pPIC9K-Rstpp are shown in red lines, and GS115 strain containing pPIC9K vector shown in blue line was used as a control. Solvent peak, glucose peak, trehalose peak and maltose peak are represented by blue, red, green and black arrows, respectively. nRIU is the refraction coefficient unit of differential refractive detector.
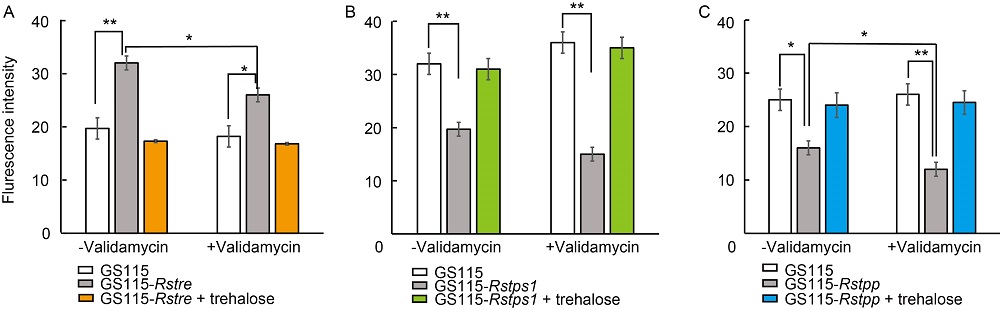
Fig. 3. Reactive oxygen species (ROS) contents in Pichia pastoris GS115 transformants of Rstre (A), Rstps1 (B) and Rstpp (C). ROS measurements were performed by the fluorescence assay. Mycelia were stained with 2’,7’-dichloro-dihydro-fluorescein diacetate for 20 min and visualized under a fluorescence microscope at 405 nm. Data are Mean ± SE (n = 3). * and ** indicate significant differences at P < 0.05 and P < 0.01 by the Duncan’s multiple range test.
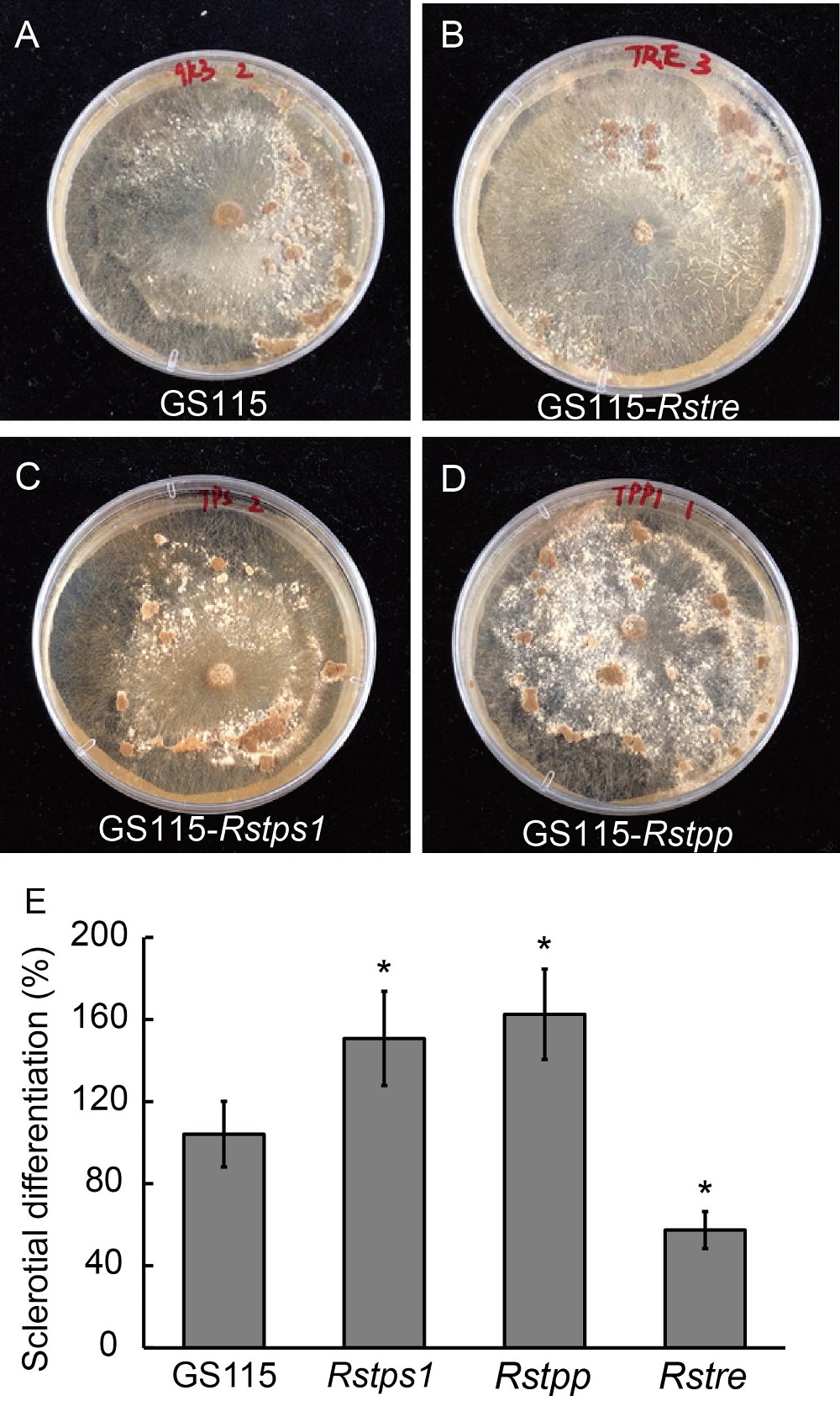
Fig. 4. Effects of fermentation broths of Pichia pastoris GS115 transformants of Rstre, Rstps1 and Rstpp on the amounts of sclerotia. A?D, Morphology of sclerotia on potato dextrose agar plate containing GS115 fermentation broth (A), GS115-Rstre yeast fermentation broth (B), GS115-Rstps1 yeast fermentation broth (C) and GS115-Rstpp yeast fermentation broth (D). E, Effect of fermentation broth of yeast inverters on sclerotia of R. solani. Sclerotial di?erentiation (SDIF) was defined as percent of dry weight of sclerotia (in respect to 100% SDIF of the control) per dish. Data are Mean ± SE (n = 3). * indicates significant difference at P < 0.05 by the Duncan's multiple range test.
| [1] |
Benaroudj N, Lee D H, Goldberg A L. 2001. Trehalose accumulation during cellular stress protects cells and cellular proteins from damage by oxygen radicals. J Biol Chem, 276: 24261-24267.
PMID |
| [2] | Bookout A L, Cummins C L, Mangelsdorf D J, Pesola J M, Kramer M F. 2006. High-throughput real-time quantitative reverse transcription PCR. Curr Protoc Mol Biol, 15(8): 15.8. 1-15.8.21. |
| [3] | da Costa Morato Nery D, da Silva C G, Mariani D, Fernandes P N, Pereira M D, Panek A D, Eleutherio E C A. 2008. The role of trehalose and its transporter in protection against reactive oxygen species. Biochim Biophys Acta, 1780: 1408-1411. |
| [4] | Domingo D D, Bawingan P A, Bharathan S, Bharathan N. 2014. Molecular screening and characterization of dsRNA from wild- type and mutant strains of Rhizoctonia solani Kühn isolates. Philipp J Sci, 143: 61-72. |
| [5] | Fan K Q, Jin L Q, Zheng Y G. 2009. The enzymatic properties of trehalase and its exploitation as a target of new pesticides. Chem Bioeng, 26(4): 7-11. |
| [6] | Fang Y Z, Yang S, Wu G Y. 2002. Free radicals, antioxidants, and nutrition. Nutrition, 18: 872-879. |
| [7] | Feng S J, Shu C W, Wang C J Z, Jiang S F, Zhou E X. 2017. Survival of Rhizoctonia solani AG-1 IA, the causal agent of rice sheath blight, under different environmental conditions. J Phytopathol, 165(1): 44-52. |
| [8] | Georgiou C D, Tairis N, Sotiropoulou A. 2000. Hydroxyl radical scavengers inhibit sclerotial differentiation and growth in Sclerotinia sclerotiorum and Rhizoctonia solani. Mycol Res, 104: 1191-1196. |
| [9] | Georgiou C D, Patsoukis N, Papapostolou I, Zervoudakis G. 2006. Sclerotial metamorphosis in filamentous fungi is induced by oxidative stress. Integr Comp Biol, 46: 691-712. |
| [10] | Harlow E, Lane D. 1988. Antibodies:A Laboratory Manual. New York, USA: Cold Spring Harbor Laboratory Publishing. |
| [11] |
Hottiger T, Schmutz P, Wiemken A. 1987. Heat-induced accumulation and futile cycling of trehalose in Saccharomyces cerevisiae. J Bacteriol, 169: 5518-5522.
PMID |
| [12] | Jin K, Peng G X, Liu Y C, Xia Y X. 2015. The acid trehalase, ATM1, contributes to the in vivo growth and virulence of the entomopathogenic fungus, Metarhizium acridum. Fungal Genet Biol, 77: 61-67. |
| [13] |
Lara-Ortíz T, Riveros-Rosas H, Aguirre J. 2003. Reactive oxygen species generated by microbial NADPH oxidase NoxA regulate sexual development in Aspergillus nidulans. Mol Microbiol, 50(4): 1241-1255.
PMID |
| [14] |
Leite F C B, da Rocha Leite D V, Pereira L F, de Barros Pita W, de Morais Junior M A. 2016. High intracellular trehalase activity prevents the storage of trehalose in the yeast Dekkera bruxellensis. Lett Appl Microbiol, 63: 210-214.
PMID |
| [15] | Lu L, Shu C W, Liu C, Wang C J Z, Zhou E X. 2016. The impacts of natural antioxidants on sclerotial differentiation and development in Rhizoctonia solani AG-1 IA. Eur J Plant Pathol, 146(4): 729-740. |
| [16] | Luo Y, Li W M, Wang W. 2008. Trehalose: Protector of antioxidant enzymes or reactive oxygen species scavenger under heat stress? Environ Exp Bot, 63: 378-384. |
| [17] | Pan L, Zhang X Q, Wang J P, Ma X, Zhou M L, Huang L K, Nie G, Wang P X, Yang Z F, Li J. 2016. Transcriptional profiles of drought- related genes in modulating metabolic processes and antioxidant defenses in Lolium multiflorum. Front Plant Sci, 7: 519. |
| [18] | Pedreño Y, González-Párraga P, Martínez-Esparza M, Sentandreu R, Valentín E, Argüelles J C. 2007. Disruption of the Candida albicans ATC1 gene encoding a cell-linked acid trehalase decreases hypha formation and infectivity without affecting resistance to oxidative stress. Microbiology, 153: 1372-1381. |
| [19] |
Richards A B, Krakowka S, Dexter L B, Schmid H, Wolterbeek A P M, Waalkens-Berendsen D H, Shigoyuki A, Kurimoto M. 2002. Trehalose: A review of properties, history of use and human tolerance, and results of multiple safety studies. Food Chem Toxicol, 40: 871-898.
PMID |
| [20] | Shu C W, Sun S, Chen J L, Chen J Y, Zhou E X. 2014. Comparison of different methods for total RNA extraction from sclerotia of Rhizoctonia solani. Electron J Biotechnol, 17(1): 50-54. |
| [21] | Shu C W, Chen J L, Sun S, Zhang M L, Wang C J Z, Zhou E X. 2015. Two distinct classes of protein related to GTB and RRM are critical in the sclerotial metamorphosis process of Rhizoctonia solani AG-1 IA. Funct Integr Genomics, 15(4): 449-459. |
| [22] | Shu C W, Zhao M, Anderson J P, Garg G, Singh K B, Zheng W B, Wang C J Z, Yang M, Zhou E X. 2019. Transcriptome analysis reveals molecular mechanisms of sclerotial development in the rice sheath blight pathogen, Rhizoctonia solani AG1-IA. Funct Integr Genomics, 19: 743-758. |
| [23] | Suzuki S, Koga M, Niizeki N, Furuya A, Matsuo K, Tanahashi Y, Tsuchida E, Nohara F, Okamoto T, Nagaya K, Azuma H. 2013. Evaluation of glycated hemoglobin and fetal hemoglobin- adjusted HbA1c measurements in infants. Pediatr Diabetes, 14(4): 267-272. |
| [24] |
Svanström A, Melin P. 2013. Intracellular trehalase activity is required for development, germination and heat-stress resistance of Aspergillus niger conidia. Res Microbiol, 164: 91-99.
PMID |
| [25] | Theerakulpisut P, Gunnula W. 2012. Exogenous sorbitol and trehalose mitigated salt stress damage in salt-sensitive but not salt-tolerant rice seedlings. Asian J Crop Sci, 4: 165-170. |
| [26] | Tournu H, Fiori A, van Dijck P. 2013. Relevance of trehalose in pathogenicity: Some general rules, yet many exceptions. PLoS Pathog, 9(8): e1003447. |
| [27] | Wang C J Z, Pi L, Jiang S F, Yang M, Shu C W, Zhou E X. 2018. ROS and trehalose regulate sclerotial development in Rhizoctonia solani AG-1 IA. Fungal Biol, 122(5): 322-332. |
| [28] | Wibberg D, Jelonek L, Rupp O, Kröber M, Goesmann A, Grosch R, Pühler A, Schlüter A. 2014. Transcriptome analysis of the phytopathogenic fungus Rhizoctonia solani AG1-IB 7/3/14 applying high-throughput sequencing of expressed sequence tags (ESTs). Fungal Biol, 118: 800-813. |
| [29] | Xu L J, Zhao W, Cao C, Wang J, Wang Q, Wang Q G. 2015. Research progress of Jinggangmycin. Chin Agric Sci Bull, 31: 191-198. (in Chinese with English abstract) |
| [30] | Zou Y P, Ma L J, Dong H T, Tao F, Feng X J, Yuan D Z, Fan D S, Hu X P. 2017. PstTPS1, the trehalose-6-phosphate synthase gene of Puccinia striiformis f. sp. tritici, involves in cold stress response and hyphae development. Physiol Mol Plant Pathol, 100: 201-208. |
| [1] | XIA Xiaodong, ZHANG Xiaobo, WANG Zhonghao, CHENG Benyi, Sun Huifeng, XU Xia, GONG Junyi, YANG Shihua, WU Jianli, SHI Yongfeng, XU Rugen. Mapping and Functional Analysis of LE Gene in a Lethal Etiolated Rice Mutant at Seedling Stage [J]. Rice Science, 2023, 30(6): 13-. |
| [2] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [3] | Jiang Changjie, Liang Zhengwei, Xie Xianzhi. Priming for Saline-Alkaline Tolerance in Rice: Current Knowledge and Future Challenges [J]. Rice Science, 2023, 30(5): 417-425. |
| [4] | Hyeran Moon, Young-Ah Kim, Ryoung Shin, Chang-Jin Park. Nucleus-Encoded Thylakoid Protein, OsY3IP1, Confers Enhanced Tolerance to Saline and Alkaline Stresses in Rice [J]. Rice Science, 2022, 29(3): 225-236. |
| [5] | Singh Priyanka, Pokharia Chitra, Shah Kavita. Exogenous Peroxidase Mitigates Cadmium Toxicity, Enhances Rhizobial Population and Lowers Root Knot Formation in Rice Seedlings [J]. Rice Science, 2021, 28(2): 166-177. |
| [6] | Ting Chen, Zheng Chen, Prakash Sathe Atul, Zhihong Zhang, Liangjian Li, Huihui Shang, Shaoqing Tang, Xiaobo Zhang, Jianli Wu. Characterization of a Novel Gain-of-Function Spotted-Leaf Mutant with Enhanced Disease Resistance in Rice [J]. Rice Science, 2019, 26(6): 372-383. |
| [7] | Li Zheng, Chen Liu, Meiling Zhang, Mei Yang, Erxun Zhou. Diversity of dsRNA Viruses Infecting Rice Sheath Blight Fungus Rhizoctonia solani AG-1 IA [J]. Rice Science, 2018, 25(1): 57-60. |
| [8] | YANG Ying-qing1, 2, #, YANG Mei1, #, LI Ming-hai1, LI Yong1, HE Xiao-xia1, ZHOU Er-xun. Establishment of Agrobacterium tumefaciens-Mediated Transformation System for Rice Sheath Blight Pathogen Rhizoctonia solani AG-1 IA [J]. RICE SCIENCE, 2011, 18(4): 297-303. |
| [9] | Shigeto MORITA, Shinya NAKATANI, Tomokazu KOSHIBA, Takehiro MASUMURA, Yasunari OGIHARA, Kunisuke TANAKA. Differential Expression of Two Cytosolic Ascorbate Peroxidase and Two Superoxide Dismutase Genes in Response to Abiotic Stress in Rice [J]. RICE SCIENCE, 2011, 18(3): 157-166. |
| [10] |
YUAN Hong-xu , XU Xin-ping , ZHANG Jian-zhong , GUO Jian-fu , LI Bao-jian .
Characteristics of Resistance to Rice Sheath Blight of Zhongda 2, a Transgenic Rice Line as Modified by Gene “RC24” [J]. RICE SCIENCE, 2004, 11(4): 177-180 . |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||