
Rice Science ›› 2017, Vol. 24 ›› Issue (4): 207-217.DOI: 10.1016/j.rsci.2017.04.001
• Orginal Article • Previous Articles Next Articles
Sakambari Mishra Swati, Panda Debabrata( )
)
Received:2017-02-15
Accepted:2017-04-26
Online:2017-07-10
Published:2017-04-28
Sakambari Mishra Swati, Panda Debabrata. Leaf Traits and Antioxidant Defense for Drought Tolerance During Early Growth Stage in Some Popular Traditional Rice Landraces from Koraput, India[J]. Rice Science, 2017, 24(4): 207-217.
Add to citation manager EndNote|Ris|BibTeX
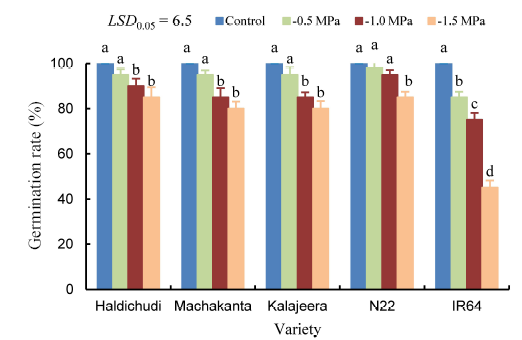
Fig. 1. Germination rate of rice in different concentration of polyethylene glycol (PEG) induced drought stress (Mean ± SD, n = 3).LSD, Least significant difference. Means followed by a common letter for the same variety are not significantly different at the 5% level by the Duncan’s multiple range test.
| Variety | Shoot length (cm) | Root length (cm) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | ||
| Haldichudi | 5.50 ± 0.14 a | 6.20 ± 0.21 a | 4.20 ± 0.14 b | 4.00 ± 0.14 b | 5.30 ± 0.16 a | 4.65 ± 0.21 b | 4.55 ± 0.07 b | 3.15 ± 0.07 c | |
| Machakanta | 6.35 ± 0.07 a | 6.20 ± 0.14 a | 5.15 ± 0.07 b | 4.55 ± 0.21 c | 10.80 ± 0.15 a | 9.80 ± 0.15 b | 6.00 ± 0.03 c | 3.65 ± 0.21 d | |
| Kalajeera | 5.40 ± 0.03 b | 5.80 ± 0.14 a | 5.55 ± 0.07 a | 4.35 ± 0.07 c | 6.20 ± 0.12 a | 5.55 ± 0.07 b | 5.35 ± 0.21 b | 3.70 ± 0.14 c | |
| N22 | 5.40 ± 0.14 b | 6.00 ± 0.06 a | 5.00 ± 0.14 c | 4.35 ± 0.21 d | 7.80 ± 0.20 a | 6.15 ± 0.29 b | 4.25 ± 0.07 c | 3.95 ± 0.18 c | |
| IR64 | 7.00 ± 0.02 a | 5.60 ± 0.05 b | 4.40 ± 0.03 c | 4.15 ± 0.07 c | 8.20 ± 0.18 a | 6.30 ± 0.14 b | 4.55 ± 0.05 c | 3.10 ± 0.02 d | |
| LSD0.05 | 0.27 | 0.34 | |||||||
| Variety | Fresh weight (g) | Dry weight (g) | |||||||
| Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | ||
| Haldichudi | 2.97 ± 0.18 a | 2.86 ± 0.07 a | 2.16 ± 0.18 b | 1.55 ± 0.08 c | 1.56 ± 0.02 a | 1.43 ± 0.01 a | 1.21 ± 0.01 b | 0.85 ± 0.07 c | |
| Machakanta | 3.41 ± 0.14 a | 3.25 ± 0.01 a | 3.05 ± 0.15 a | 2.60 ± 0.28 b | 2.14 ± 0.01 a | 2.09 ± 0.02 a | 1.90 ± 0.03 b | 1.65 ± 0.08 c | |
| Kalajeera | 3.17 ± 0.07 a | 3.08 ± 0.02 a | 3.21 ± 0.14 a | 2.45 ± 0.07 b | 2.21 ± 0.05 a | 1.19 ± 0.03 a | 2.03 ± 0.06 b | 1.70 ± 0.12 c | |
| N22 | 3.14 ± 0.15 a | 3.27 ± 0.25 a | 2.90 ± 0.03 a | 2.45 ± 0.09 b | 1.86 ± 0.01 a | 1.48 ± 0.02 b | 1.32 ± 0.04 b | 1.25 ± 0.07 c | |
| IR64 | 3.53 ± 0.12 a | 2.44 ± 0.28 b | 2.20 ± 0.14 b | 1.50 ± 0.14 c | 2.45 ± 0.03 a | 2.00 ± 0.04 b | 1.35 ± 0.12 c | 0.80 ± 0.03 d | |
| LSD0.05 | 0.38 | 0.15 | |||||||
Table 1 Changes of growth parameters of rice seedlings grown under different levels of polyethylene glycol (PEG) induced drought stress.
| Variety | Shoot length (cm) | Root length (cm) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | ||
| Haldichudi | 5.50 ± 0.14 a | 6.20 ± 0.21 a | 4.20 ± 0.14 b | 4.00 ± 0.14 b | 5.30 ± 0.16 a | 4.65 ± 0.21 b | 4.55 ± 0.07 b | 3.15 ± 0.07 c | |
| Machakanta | 6.35 ± 0.07 a | 6.20 ± 0.14 a | 5.15 ± 0.07 b | 4.55 ± 0.21 c | 10.80 ± 0.15 a | 9.80 ± 0.15 b | 6.00 ± 0.03 c | 3.65 ± 0.21 d | |
| Kalajeera | 5.40 ± 0.03 b | 5.80 ± 0.14 a | 5.55 ± 0.07 a | 4.35 ± 0.07 c | 6.20 ± 0.12 a | 5.55 ± 0.07 b | 5.35 ± 0.21 b | 3.70 ± 0.14 c | |
| N22 | 5.40 ± 0.14 b | 6.00 ± 0.06 a | 5.00 ± 0.14 c | 4.35 ± 0.21 d | 7.80 ± 0.20 a | 6.15 ± 0.29 b | 4.25 ± 0.07 c | 3.95 ± 0.18 c | |
| IR64 | 7.00 ± 0.02 a | 5.60 ± 0.05 b | 4.40 ± 0.03 c | 4.15 ± 0.07 c | 8.20 ± 0.18 a | 6.30 ± 0.14 b | 4.55 ± 0.05 c | 3.10 ± 0.02 d | |
| LSD0.05 | 0.27 | 0.34 | |||||||
| Variety | Fresh weight (g) | Dry weight (g) | |||||||
| Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | Control | -0.5 MPa | -1.0 MPa | -1.5 MPa | ||
| Haldichudi | 2.97 ± 0.18 a | 2.86 ± 0.07 a | 2.16 ± 0.18 b | 1.55 ± 0.08 c | 1.56 ± 0.02 a | 1.43 ± 0.01 a | 1.21 ± 0.01 b | 0.85 ± 0.07 c | |
| Machakanta | 3.41 ± 0.14 a | 3.25 ± 0.01 a | 3.05 ± 0.15 a | 2.60 ± 0.28 b | 2.14 ± 0.01 a | 2.09 ± 0.02 a | 1.90 ± 0.03 b | 1.65 ± 0.08 c | |
| Kalajeera | 3.17 ± 0.07 a | 3.08 ± 0.02 a | 3.21 ± 0.14 a | 2.45 ± 0.07 b | 2.21 ± 0.05 a | 1.19 ± 0.03 a | 2.03 ± 0.06 b | 1.70 ± 0.12 c | |
| N22 | 3.14 ± 0.15 a | 3.27 ± 0.25 a | 2.90 ± 0.03 a | 2.45 ± 0.09 b | 1.86 ± 0.01 a | 1.48 ± 0.02 b | 1.32 ± 0.04 b | 1.25 ± 0.07 c | |
| IR64 | 3.53 ± 0.12 a | 2.44 ± 0.28 b | 2.20 ± 0.14 b | 1.50 ± 0.14 c | 2.45 ± 0.03 a | 2.00 ± 0.04 b | 1.35 ± 0.12 c | 0.80 ± 0.03 d | |
| LSD0.05 | 0.38 | 0.15 | |||||||
| Variety | Shoot length | Root length | Fresh weight | Dry weight | Fo | Fm | Fv/Fm | qP |
|---|---|---|---|---|---|---|---|---|
| Haldichudi | 0.73 ± 0.03 a | 0.59 ± 0.03 a | 0.52 ± 0.01 b | 0.54 ± 0.03 b | 1.15 ± 0.03 b | 0.94 ± 0.02 a | 0.92 ± 0.03 a | 0.96 ± 0.01 a |
| Machakanta | 0.72 ± 0.03 b | 0.34 ± 0.06 b | 0.76 ± 0.02 a | 0.77 ± 0.03 a | 1.06 ± 0.02 c | 0.89 ± 0.01 a | 0.93 ± 0.02 a | 0.93 ± 0.02 a |
| Kalajeera | 0.80 ± 0.04 a | 0.60 ± 0.04 a | 0.77 ± 0.01 a | 0.77 ± 0.02 a | 1.20 ± 0.00 b | 0.89 ± 0.01 a | 0.92 ± 0.01 a | 0.93 ± 0.03 a |
| N22 | 0.81 ± 0.03 a | 0.51 ± 0.03 a | 0.78 ± 0.02 a | 0.67 ± 0.00 a | 1.06 ± 0.03 c | 0.95 ± 0.02 a | 0.96 ± 0.01 a | 0.98 ± 0.02 a |
| IR64 | 0.63 ± 0.02 c | 0.38 ± 0.01 b | 0.42 ± 0.02 c | 0.33 ± 0.01 c | 1.41 ± 0.02 a | 0.63 ± 0.04 b | 0.88 ± 0.02 b | 0.68 ± 0.01 b |
| LSD0.05 | 0.07 | 0.11 | 0.04 | 0.12 | 0.04 | 0.06 | 0.04 | 0.04 |
| Variety | NPQ | SPAD | SOD | APX | GPX | CAT | Protein | Proline |
| Haldichudi | 1.32 ± 0.01 b | 0.54 ± 0.02 b | 1.81 ± 0.03 b | 2.02 ± 0.01 b | 2.47 ± 0.04 a | 4.47 ± 0.09 a | 1.09 ± 0.02 b | 1.12 ± 0.03 b |
| Machakanta | 1.26 ± 0.02 c | 0.75 ± 0.01 a | 2.08 ± 0.04 a | 2.34 ± 0.02 a | 0.86 ± 0.03 d | 3.60 ± 0.04 b | 1.06 ± 0.02 b | 1.75 ± 0.04 a |
| Kalajeera | 1.33 ± 0.01 b | 0.73 ± 0.02 a | 1.92 ± 0.01 b | 2.51 ± 0.03 a | 1.62 ± 0.02 c | 4.60 ± 0.08 a | 0.94 ± 0.03 c | 1.65 ± 0.03 a |
| N22 | 1.32 ± 0.00 b | 0.83 ± 0.06 a | 1.64 ± 0.02 c | 2.30 ± 0.01 a | 2.03 ± 0.05 b | 1.99 ± 0.14 c | 1.55 ± 0.03 a | 1.59 ± 0.02 a |
| IR64 | 2.03 ± 0.01 a | 0.51 ± 0.06 b | 0.86 ± 0.01 d | 0.60 ± 0.06 c | 0.82 ± 0.06 d | 1.23 ± 0.18 d | 0.89 ± 0.06 c | 0.56 ± 0.02 c |
| LSD0.05 | 0.02 | 0.12 | 0.13 | 0.25 | 0.22 | 0.33 | 0.08 | 0.16 |
Table 2 Relative ratios of different parameters in rice seedlings grown in -1.5 MPa drought level for 15 d.
| Variety | Shoot length | Root length | Fresh weight | Dry weight | Fo | Fm | Fv/Fm | qP |
|---|---|---|---|---|---|---|---|---|
| Haldichudi | 0.73 ± 0.03 a | 0.59 ± 0.03 a | 0.52 ± 0.01 b | 0.54 ± 0.03 b | 1.15 ± 0.03 b | 0.94 ± 0.02 a | 0.92 ± 0.03 a | 0.96 ± 0.01 a |
| Machakanta | 0.72 ± 0.03 b | 0.34 ± 0.06 b | 0.76 ± 0.02 a | 0.77 ± 0.03 a | 1.06 ± 0.02 c | 0.89 ± 0.01 a | 0.93 ± 0.02 a | 0.93 ± 0.02 a |
| Kalajeera | 0.80 ± 0.04 a | 0.60 ± 0.04 a | 0.77 ± 0.01 a | 0.77 ± 0.02 a | 1.20 ± 0.00 b | 0.89 ± 0.01 a | 0.92 ± 0.01 a | 0.93 ± 0.03 a |
| N22 | 0.81 ± 0.03 a | 0.51 ± 0.03 a | 0.78 ± 0.02 a | 0.67 ± 0.00 a | 1.06 ± 0.03 c | 0.95 ± 0.02 a | 0.96 ± 0.01 a | 0.98 ± 0.02 a |
| IR64 | 0.63 ± 0.02 c | 0.38 ± 0.01 b | 0.42 ± 0.02 c | 0.33 ± 0.01 c | 1.41 ± 0.02 a | 0.63 ± 0.04 b | 0.88 ± 0.02 b | 0.68 ± 0.01 b |
| LSD0.05 | 0.07 | 0.11 | 0.04 | 0.12 | 0.04 | 0.06 | 0.04 | 0.04 |
| Variety | NPQ | SPAD | SOD | APX | GPX | CAT | Protein | Proline |
| Haldichudi | 1.32 ± 0.01 b | 0.54 ± 0.02 b | 1.81 ± 0.03 b | 2.02 ± 0.01 b | 2.47 ± 0.04 a | 4.47 ± 0.09 a | 1.09 ± 0.02 b | 1.12 ± 0.03 b |
| Machakanta | 1.26 ± 0.02 c | 0.75 ± 0.01 a | 2.08 ± 0.04 a | 2.34 ± 0.02 a | 0.86 ± 0.03 d | 3.60 ± 0.04 b | 1.06 ± 0.02 b | 1.75 ± 0.04 a |
| Kalajeera | 1.33 ± 0.01 b | 0.73 ± 0.02 a | 1.92 ± 0.01 b | 2.51 ± 0.03 a | 1.62 ± 0.02 c | 4.60 ± 0.08 a | 0.94 ± 0.03 c | 1.65 ± 0.03 a |
| N22 | 1.32 ± 0.00 b | 0.83 ± 0.06 a | 1.64 ± 0.02 c | 2.30 ± 0.01 a | 2.03 ± 0.05 b | 1.99 ± 0.14 c | 1.55 ± 0.03 a | 1.59 ± 0.02 a |
| IR64 | 2.03 ± 0.01 a | 0.51 ± 0.06 b | 0.86 ± 0.01 d | 0.60 ± 0.06 c | 0.82 ± 0.06 d | 1.23 ± 0.18 d | 0.89 ± 0.06 c | 0.56 ± 0.02 c |
| LSD0.05 | 0.02 | 0.12 | 0.13 | 0.25 | 0.22 | 0.33 | 0.08 | 0.16 |

Fig. 2. Minimum fluorescence (Fo), maximum fluorescence (Fm), photochemical efficiency of PSII (Fv/Fm), photochemical quenching (qP), non-photochemical quenching (NPQ) and soil and plant analyzer development (SPAD) chlorophyll index in rice leaves grown in different concentrations of polyethylene glycol (PEG) (Mean ± SD, n = 3).LSD, Least significant difference. Means followed by the same letter are not significantly different at the 5% level by the Duncan’s multiple range test.
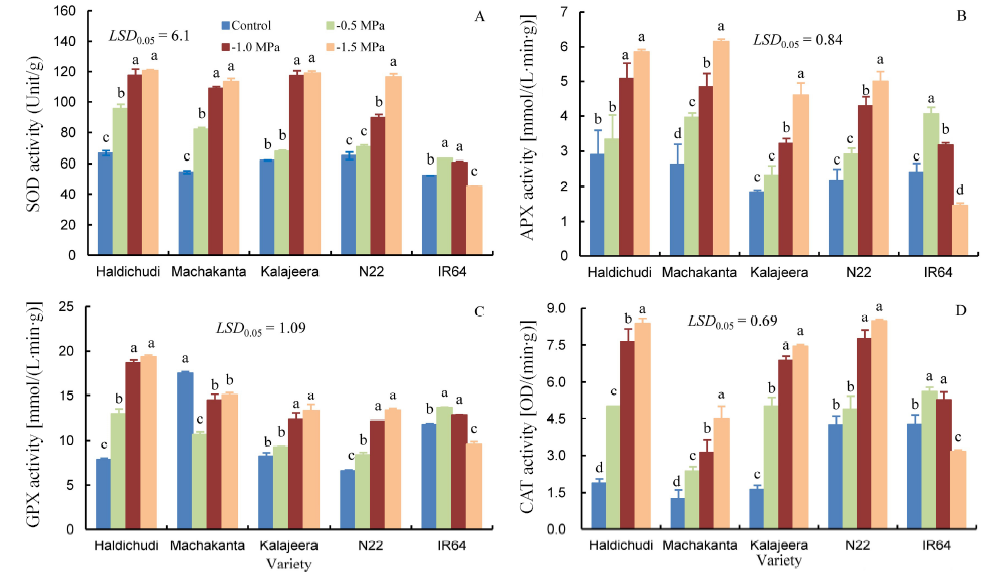
Fig. 3. Changes of superoxide dismutase (SOD), ascorbate peroxidase (APX), guaiacol peroxidase (GPX) and catalase (CAT) in rice leaves grown in different concentrations of polyethylene glycol (PEG) (Mean ± SD, n = 3).LSD, Least significant difference. Means followed by the same letter are not significantly different at the 5% level by the Duncan’s multiple range test.
| Parameter | Treatment (df = 3) | Variety (df = 4) | Variety × Treatment (df = 12) |
|---|---|---|---|
| Shoot length | 350** | 25.45** | 45.41** |
| Root length | 250** | 345** | 85.01** |
| Fresh weight | 71.8** | 26.4** | 5.35** |
| Dry weight | 227** | 158** | 20** |
| Fo | 18 422.5** | 1 625.7* | 10 540.2** |
| Fm | 281 083** | 183 768** | 260 695** |
| Fv/Fm | 0.048** | 0.024* | 0.041** |
| qP | 0.048** | 0.077** | 0.062** |
| NPQ | 0.018** | 0.006** | 0.006* |
| SPAD index | 120.0** | 170.4** | 35.30* |
| SOD activity | 11 413** | 9 358** | 5 020* |
| APX activity | 29.9** | 15.24* | 22.97** |
| GPX activity | 230.33** | 99.07* | 133.67** |
| CAT activity | 88.61** | 57.81** | 49.21** |
| Proline content | 4.04* | 21.85** | 8.25* |
| Protein content | 37.88** | 40.48** | 36.09* |
Table 3 Analysis of variance of studied parameters in rice seedlings grown in different concentrations of polyethylene glycol (PEG).
| Parameter | Treatment (df = 3) | Variety (df = 4) | Variety × Treatment (df = 12) |
|---|---|---|---|
| Shoot length | 350** | 25.45** | 45.41** |
| Root length | 250** | 345** | 85.01** |
| Fresh weight | 71.8** | 26.4** | 5.35** |
| Dry weight | 227** | 158** | 20** |
| Fo | 18 422.5** | 1 625.7* | 10 540.2** |
| Fm | 281 083** | 183 768** | 260 695** |
| Fv/Fm | 0.048** | 0.024* | 0.041** |
| qP | 0.048** | 0.077** | 0.062** |
| NPQ | 0.018** | 0.006** | 0.006* |
| SPAD index | 120.0** | 170.4** | 35.30* |
| SOD activity | 11 413** | 9 358** | 5 020* |
| APX activity | 29.9** | 15.24* | 22.97** |
| GPX activity | 230.33** | 99.07* | 133.67** |
| CAT activity | 88.61** | 57.81** | 49.21** |
| Proline content | 4.04* | 21.85** | 8.25* |
| Protein content | 37.88** | 40.48** | 36.09* |
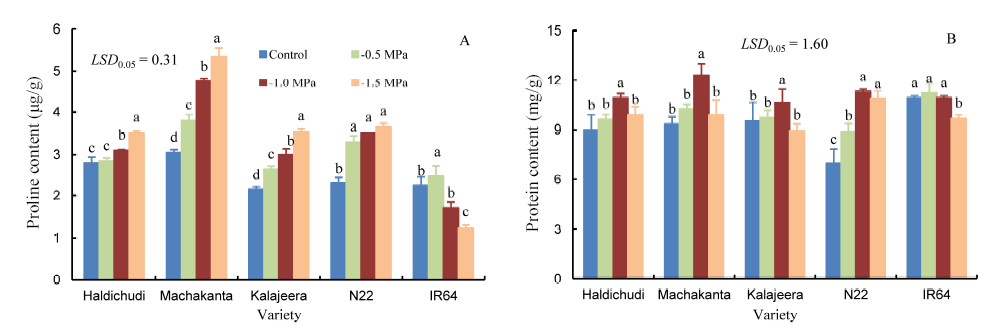
Fig. 4. Changes of proline and protein contents in rice leaves grown in different concentrations of polyethylene glycol (PEG).LSD, Least significant difference.Means followed by a common letter are not significantly different at the 5% level by the Duncan’s multiple range test (n = 3).
| Parameter | Fo | Fm | Fv | Fv/Fm | qP | NPQ | SPAD |
|---|---|---|---|---|---|---|---|
| APX activity | -0.379** | 0.496** | 0.513** | 0.629** | 0.469** | -0.361** | 0.431** |
| GPX activity | -0.380** | 0.447** | 0.459** | 0.596** | 0.353** | -0.314** | 0.576** |
| CAT activity | -0.239* | 0.397** | 0.625** | 0.698** | 0.202* | -0.257* | 0.439** |
| SOD activity | -0.181* | 0.469** | 0.438** | 0.430** | 0.179* | -0.214* | 0.586** |
| Protein content | -0.228* | 0.328** | 0.301** | 0.251* | 0.333** | -0.279* | 0.486** |
| Proline content | -0.172* | 0.265* | 0.238* | 0.280* | 0.395** | -0.279* | 0.202* |
Table 4 Correlation coefficient (r value) between leaf photosynthetic parameters with antioxidant enzymes as well as protein and proline contents in rice seedlings grown in different concentrations of polyethylene glycol (PEG).
| Parameter | Fo | Fm | Fv | Fv/Fm | qP | NPQ | SPAD |
|---|---|---|---|---|---|---|---|
| APX activity | -0.379** | 0.496** | 0.513** | 0.629** | 0.469** | -0.361** | 0.431** |
| GPX activity | -0.380** | 0.447** | 0.459** | 0.596** | 0.353** | -0.314** | 0.576** |
| CAT activity | -0.239* | 0.397** | 0.625** | 0.698** | 0.202* | -0.257* | 0.439** |
| SOD activity | -0.181* | 0.469** | 0.438** | 0.430** | 0.179* | -0.214* | 0.586** |
| Protein content | -0.228* | 0.328** | 0.301** | 0.251* | 0.333** | -0.279* | 0.486** |
| Proline content | -0.172* | 0.265* | 0.238* | 0.280* | 0.395** | -0.279* | 0.202* |
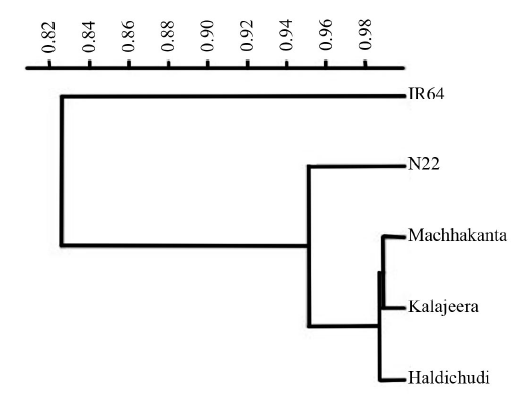
Fig. 5. Dendrogram showing similarity of rice landraces with tolerant (N22) and susceptible (IR64) check varieties using leaf phenotyping parameters under drought stress (-1.5 MPa) at the early growth stage.
| 1 | Abenavoli M R, Leone M, Sunseri F, Bacchi M, Sorgona A.2016. Root phenotyping for drought tolerance in bean landraces from Calabria (Italy).J Agron Crop Sci, 202(1): 1-12. |
| 2 | Agnihotri R K, Palni L M S, Pandey D K.2007. Germination and seedling growth under moisture stress: Screening of landraces of rice (Oryza sativa L.) from Kumaun region of Indian Central Himalaya.J Plant Biol, 34(1): 21-27. |
| 3 | Alonso-Blanco C, Aarts M G M, Bentsink L, Keurentjes J J B, Reymond M, Vreugdenhil D, Koornneef M.2009. What has natural variation taught us about plant development, physiology, and adaptation? Plant Cell, 21(7): 1877-1896. |
| 4 | Anower M R, Boe A, Auger D, Mott I W, Peel M D, Xu L, Kanchupati P, Wu Y J.2015. Comparative drought response in eleven diverse Alfalfa accessions.J Agron Crop Sci, 203: 1-13. |
| 5 | Aro E M, Virgin I, Anderson B.1993. Photoinhibition of photosystem II, inactivation, protein damage and turnover.Biochim Biophys Acta, 1143(2): 113-134. |
| 6 | Arora A, Sairam R K, Srivastava G C.2002. Oxidative stress and antioxidative system in plants.Curr Sci, 82(10): 1227-1238. |
| 7 | Arunachalam V, Chaudhury S S, Sarangi S K, Ray T, Mohanty B P, Nambi V A, Mishra S.2006. Rising on Rice: The Story of Jeypore. Chennai, India: Swaminathan Research Foundation: 1-37. |
| 8 | Ashraf M, Foolad M R.2007. Roles of glycine betaine and proline in improving plant abiotic stress resistance.Environ Exp Bot, 59(2): 206-216. |
| 9 | Athar H, Khan A, Ashraf M.2008. Exogenously applied ascorbic acid alleviates salt-induced oxidative stress in wheat.Environ Exp Bot, 63: 224-231. |
| 10 | Bates L S, Waldren R P, Teare I D.1973. Rapid determination of free proline for water stress studies.Plant Soil, 39(1): 205-207. |
| 11 | Batra N G, Kumari N, Sharma V.2016. Photosynthetic performance of Ocimum sanctum morphotypes in a semiarid region.J Herbs Spices Med Plants, 22(3): 211-224. |
| 12 | Beena R, Thandapani V, Chandrababu R.2012. Physio-morphological and biochemical characterization of selected recombinant inbred lines of rice for drought resistance.Ind J Plant Physiol, 17(2): 189-193. |
| 13 | Bray E A, Bailey-Serres J, Weretilnyk E.2000. Responses to abiotic stresses. In: Gruissem W, Buchannan B, Jones R. Biochemistry and Molecular Biology of Plants. Maryland, USA: American Society of Plant Physiologists: 1158-1249. |
| 14 | Cakmak I, Marschner H.1992. Magnesium deficiency and highlight intensity enhance activities of superoxide dismutase, ascorbate peroxidase and glutathione reductase in bean leaves.Plant Physiol, 98(4): 1222-1227. |
| 15 | Calatayud A, Roca D, Martínez P F.2006. Spatial-temporal variations in rose leaves under water stress conditions studied by chlorophyll fluorescence imaging.Plant Physiol Biochem, 44(10): 564-573. |
| 16 | Choudhury S R, Choudhuri M A.1985. Hydrogen peroxide metabolism as an index of water stress tolerance in jute.Physiol Plant, 65(4): 467-480. |
| 17 | Dudeja S S, Chaudhary P.2005. Fast chlorophyll fluorescence transient and nitrogen fixing ability of chickpea nodulation variants.Photosynthetica, 43(2): 253-259. |
| 18 | Faize M, Burgos L, Faize L, Piqueras A, Nicolas E, Barba-Espin G, Clemente-Moreno M J, Alcobendas R, Artlip T, Hernandez J A.2011. Involvement of cytosolic ascorbate peroxidase and Cu/Zn-superoxide dismutase for improved tolerance against drought stress.J Exp Bot, 62(8): 2599-2613. |
| 19 | Fu W G, Li P P, Wu Y Y.2012. Effects of different light intensities on chlorophyll fluorescence characteristics and yield in lettuce.Sci Hort, 135: 45-51. |
| 20 | Gao J M, Xiao Q, Ding L P, Chen M J, Yin L, Li J Z, Zhou S Y, He G Y.2008. Differential responses of lipid peroxidation and antioxidants in Alternanthera philoxeroides and Oryza sativa subjected to drought stress.Plant Growth Regul, 56: 89-95. |
| 21 | Giannopolitis C N, Ries S K.1977. Superoxide dismutases: I. Occurrence in higher plants.Plant Physiol, 59(2): 309-314. |
| 22 | Guan X K, Song L, Wang T C, Turner N C, Li F M.2015. Effect of drought on the gas exchange, chlorophyll fluorescence and yield of six different-era spring wheat cultivars.J Agron Crop Sci, 201(4): 253-266. |
| 23 | Hazrati S, Tahmasebi-Sarvestani Z, Modarres-Sanavy S A M, Mokhtassi-Bidgoli A, Nicola S.2016. Effects of water stress and light intensity on chlorophyll fluorescence parameters and pigments of Aloe vera L.Plant Physiol Biochem, 106: 141-148. |
| 24 | Jagadish S V K, Muthurajan R, Oane R, Wheeler T R, Heuer S, Bennett J, Craufurd P Q.2010. Physiological and proteomic approaches to address heat tolerance during anthesis in rice.J Exp Bot, 61(1): 143-156. |
| 25 | Karmakar J, Roychowdhury R, Kar R K, Deb D, Dey N.2012. Profiling of selected indigenous rice (Oryza sativa L.) landraces of Rarh Bengal in relation to osmotic stress tolerance.Physiol Mol Biol Plants, 18(2): 125-132. |
| 26 | Krause G H, Weis E.1991. Chlorophyll fluorescence and photosynthesis: The basics.Annu Rev Plant Physiol, 42: 313-349. |
| 27 | Kumar R R, Karajol K, Naik G R.2011. Effect of polyethylene glycol induced water stress on physiological and biochemical responses in pigeonpea (Cajanus cajan L. Millsp.).Rec Res Sci Technol, 3(1): 148-152. |
| 28 | Kumbhar S D, Kulwal P L, Patil J V, Sarawate C D, Gaikwad A P, Jadhav A S.2015. Genetic diversity and population structure in landraces and improved rice varieties from India.Rice Sci, 22(3): 99-107. |
| 29 | Lowry O H, Rosebrough N J, Farr A L, Randall R J.1951. Protein measurement with Folin phenol reagent.J Biol Chem, 193: 265-275. |
| 30 | Lum M S, Hanafi M M, Rafii Y M, Akmar A S N.2014. Effect of drought stress on growth, proline and antioxidant enzyme activities of upland rice.J Anim Plant Sci, 24(5): 1487-1493. |
| 31 | Mathobo R, Marais D, Steyn J M.2017. The effect of drought stress on yield, leaf gaseous exchange and chlorophyll fluorescence of dry beans (Phaseolus vulgaris L.).Agric Water Manag, 180: 118-125. |
| 32 | Maxwell K, Johnson G N.2000. Chlorophyll fluorescence practical guide.J Exp Bot, 51: 659-668. |
| 33 | Mishra S, Chaudhury S S, Nambi V A.2012. Strengthening of traditional paddy seed selection practices of tribal farm families with improved knowledge and skills in Koraput district, Odisha.Ind J Trad Knowl, 11(3): 461-470. |
| 34 | Murchie E H, Lawson T.2013. Chlorophyll fluorescence analysis: A guide to good practice and understanding some new applications.J Exp Bot, 64(13): 3983-3998. |
| 35 | Nakano Y, Asada K.1981. Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts.Plant Cell Physiol, 22(5): 867-880. |
| 36 | Pandey V, Shukla A.2015. Acclimation and tolerance strategies of rice under drought stress.Rice Sci, 22(4): 147-161. |
| 37 | Parida A K, Das A B.2005. Salt tolerance and salinity effects on plants.Ecotoxicol Environ Safety, 60(3): 324-349. |
| 38 | Patra B C, Dhua S R.2003. Agro-morphological diversity scenario in upland rice germplasm of Jeypore tract.Genet Resour Crop Evol, 50(8): 825-828. |
| 39 | Pinnola A, Dall’Osto L, Gerotto C, Morosinotto T, Bassi R, Alboresi A.2013. Zeaxanthin binds to light-harvesting complex stress-related protein to enhance non-photochemical quenching in Physcomitrella patens. Plant Cell, 25(9): 3519-3534. |
| 40 | Prashanth S R, Parani M, Mohanty B P, Talame V, Tuberosa R, Parida A.2002. Genetic diversity in cultivars and landraces of Oryza sativa subsp. indica as revealed by AFLP markers.Genome, 45(3): 451-459. |
| 41 | Rao M V, Hale B A, Ormrod D P.1995. Amelioration of ozone- induced oxidative damage in wheat plants grown under high carbon dioxide: Role of antioxidant enzymes.Plant Physiol, 109(2): 421-432. |
| 42 | Rebolledo M C, Dingkuhn M, Clément-Vidal A, Rouan L, Luquet D.2012. Phenomics of rice early vigour and drought response: Are sugar related and morphogenetic traits relevant?Rice, 5(1): 22. |
| 43 | Roy P S, Patnaik A, Rao G J N, Patnaik S S C, Chaudhury S S, Sharma S G.2016. Participatory and molecular marker assisted pure line selection for refinement of three premium rice landraces of Koraput, India.Agroecol Sust Food Syst, 41(2): 167-185. |
| 44 | Sairam R K, Srivastava G C.2001. Water stress tolerance of wheat (Triticum aestivum L.): Variations in hydrogen peroxide accumulation and antioxidant activity in tolerant and susceptible genotypes.J Agron Crop Sci, 186(1): 63-70. |
| 45 | Sarkar R K, Bhattacharjee B.2011. Rice genotypes with SUB1 QTL differ in submergence tolerance, elongation ability during submergence and re-generation growth at re-emergence.Rice, 5(7): 1-11. |
| 46 | Sarkar R K, Mahata K R, Singh D P.2013. Differential responses of antioxidant system and photosynthetic characteristics in four rice cultivars differing in sensitivity to sodium chloride stress.Acta Physiol Plant, 35(10): 2915-2926. |
| 47 | Sayed O H.2003. Chlorophyll fluorescence as a tool in cereal crop research.Photosynthetica, 41(3): 321-330. |
| 48 | Shrestha S, Brueck H, Asch F.2012. Chlorophyll index, photochemical reflectance index and chlorophyll fluorescence measurements of rice leaves supplied with different N levels.J Photochem Photobiol, 113: 7-13. |
| 49 | Singh A, Sengar K, Sengar R S.2013. Gene regulation and biotechnology of drought tolerance in rice.Int J Biotechnol Bioeng Res, 4(6): 547-552. |
| 50 | Soleymani A, Shahrajabian M H.2012. Study of cold stress on the germination and seedling stage and determination of recovery in rice varieties.Int J Biol, 4(4): 23-30. |
| 51 | Souza R P, Machado E C, Silva J A B, Lagôa A M M A, Silveira J A G.2004. Photosynthetic gas exchange, chlorophyll fluorescence and some associated metabolic changes in cowpea (Vigna unguiculata) during water stress and recovery.Environ Exp Bot, 51(1): 45-56. |
| 52 | Stirbet A, Govindjee.2011. On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and photosystem: II. Basics and applications of the OJIP fluorescence transient.J Photochem Photobiol Biol, 104(1/2): 236-257. |
| 53 | Strasser R J, Tsimilli-Michael M, Srivastava A.2004. Analysis of the chlorophyll fluorescence transient. In: Papageorgiou G C, Govindjee. Chlorophyll a Fluorescence: A Signature of Photosynthesis. Dordrecht, the Netherlands: Springer: 321-362. |
| 54 | Tripathy S, Gurung P, Sharma S D.2005. Intellectual property contribution with regard to rice genetic resources by tribes of south Orissa, India.Plant Genet Resour Newsl, 141: 70-73. |
| 55 | Vikram P, Kadam S, Singh B P, Lee Y J, Pal J K, Singh S, Singh O N, Swamy B P M, Thiyagarajan K, Singh S, Singh N K.2016. Genetic diversity analysis reveals importance of green revolution gene (Sd1 locus) for drought tolerance in rice.Agric Res, 5(1): 1-12. |
| 56 | Yang P M, Huang Q C, Qin G Y, Zhao S P, Zhou J G.2014. Different drought-stress responses in photosynthesis and reactive oxygen metabolism between autotetraploid and diploid rice. Photosynthetica, 52(2): 193-202. |
| 57 | Zhang H W, Liu W, Wan L Y, Li F, Dai L Y, Li D J, Zhang Z J, Huang R F.2010. Functional analyses of ethylene response factor JERF3 with the aim of improving tolerance to drought and osmotic stress in transgenic rice.Transg Res, 19(5): 809-818. |
| 58 | Zhang L D, Yu S W, Zuo K J, Luo L J, Tang K X.2012. Identification of gene modules associated with drought response in rice by network-based analysis.PLoS One, 7(5): 1-12. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [13] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| [14] | Lu Xuedan, Li Fan, Xiao Yunhua, Wang Feng, Zhang Guilian, Deng Huabing, Tang Wenbang. Grain Shape Genes: Shaping the Future of Rice Breeding [J]. Rice Science, 2023, 30(5): 379-404. |
| [15] | Zhang Guomei, Li Han, Liu Shanshan, Zhou Xuming, Lu Mingyang, Tang Liang, Sun Lihua. Water Extract of Rice False Smut Balls Activates Nrf2/HO-1 and Apoptosis Pathways, Causing Liver Injury [J]. Rice Science, 2023, 30(5): 473-485. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||