
Rice Science ›› 2022, Vol. 29 ›› Issue (3): 225-236.DOI: 10.1016/j.rsci.2021.08.004
• Research Paper • Previous Articles Next Articles
Hyeran Moon1,#, Young-Ah Kim2,#, Ryoung Shin3, Chang-Jin Park1,2,4( )
)
Received:2021-06-21
Accepted:2021-08-02
Online:2022-05-28
Published:2022-03-10
Contact:
Chang-Jin Park
About author:First author contact:#These authors contributed equally to this work
Hyeran Moon, Young-Ah Kim, Ryoung Shin, Chang-Jin Park. Nucleus-Encoded Thylakoid Protein, OsY3IP1, Confers Enhanced Tolerance to Saline and Alkaline Stresses in Rice[J]. Rice Science, 2022, 29(3): 225-236.
Add to citation manager EndNote|Ris|BibTeX
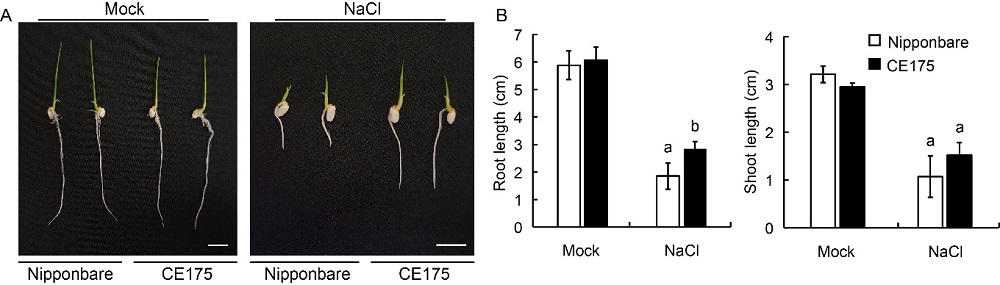
Fig. 1. Salinity-tolerant phenotypes of one-week-old FOX-rice line CE175 overexpressing OsY3IP1 on 1/2 Murashige and Skoog medium containing 150 mmol/L NaCl. A, Seedling growth of CE175 and Nipponbare at 4 d after NaCl treatment. Scale bars, 1 cm. B, Shoot and root lengths of CE175 and Nipponbare at 4 d after NaCl treatment. Error bars represent standard error of at least four plants. Different lowercase letters above the bars indicate significant differences at P < 0.01.

Fig. 2. Amino acid sequence alignment of plant Y3IP1 homologues. A predicted chloroplast transfer peptide by the TargetP-2.0 and ChloroP 1.1 is marked with a red solid box in the O. sativa sequence. A putative transmembrane region identified using SOSUI 1.11 is indicated with a red dashed box in the O. sativa sequence. Amino acid residues conserved in all species are marked with asterisks.
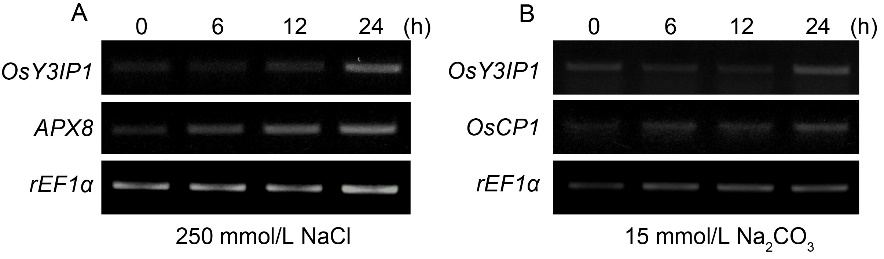
Fig. 3. Induction of OsY3IP1 expression of 4-week-old Kitaake plants by saline (A, 250 mmol/L NaCl) and alkaline (B, 15 mmol/L Na2CO3) stresses at 0, 6, 12 and 24 h. Ascorbate peroxidase 8 (APX8) and cysteine protease (OsCP1) gene were used as positive controls for saline and alkaline treatments, respectively. Rice elongation factor 1α (rEF1α) was used as an internal control.
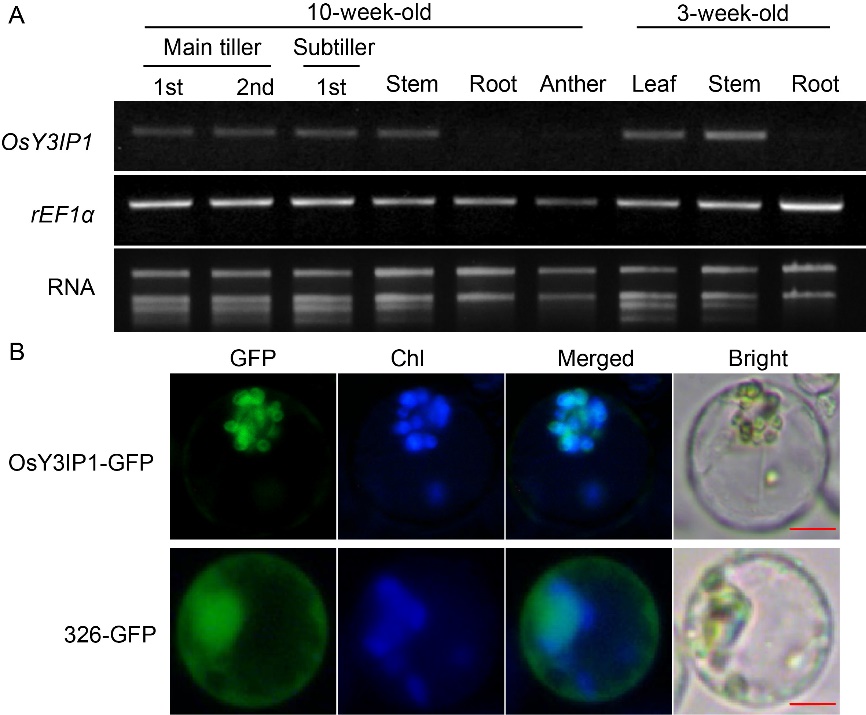
Fig. 4. Tissue-specific expression and subcellular localization of OsY3IP1. A, Expression analysis of OsY3IP1 gene in different organs. After total RNA was extracted from various tissues at 10- and 3-week-old Kitaake plants, RT-PCR was performed with specific primers for each gene. rEF1α was used as an internal control. B, Subcellular localization of OsY3IP1 in rice protoplasts. Ubi::OsY3IP1-GFP/pC1300 (OsY3IP1-GFP) (on upper panel) and 326-GFP (on lower panel) were introduced into rice protoplasts by polyethylene glycol (PEG)-mediated transformation, respectively. Chloroplasts (Chl) were visualized by their chlorophyll autofluorescence. Images were collected with an inverted fluorescence microscope Eclipse Ti (Nikon, Tokyo, Japan) fitted with an objective (400×). The filter sets were C-FL-C FITC (excitation 465 nm to 495 nm) and C-FL-C DAPI (excitation 361 nm to 389 nm) to detect GFP and the chlorophyll autofluorescence, respectively. GFP, Green fluorescent protein. Scale bars, 10 μm.
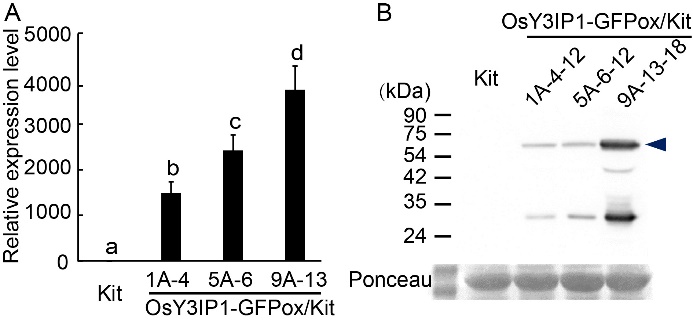
Fig. 5. Detection of overexpression of OsY3IP1-GFP. A, Expression levels of OsY3IP1 in OsY3IP1-GFPox/Kit transgenic lines T1 (progenies of 1A-4, 5A-6 and 9A-13). Expression level in Kitaake (Kit) was defined as 1. Data are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences at P < 0.05. B, Total protein was extracted from Kit and OsY3IP1-GFPox/Kit (1A-4-12, 5A-6-12 and 9A-13-18). Equal amounts (100 μg) of total protein were analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis and immunobloted with anti-GFP antibody. Equal total protein loading was demonstrated by Ponceau staining. OsY3IP1-GFP of approximately 60 kDa was marked with an arrowhead.
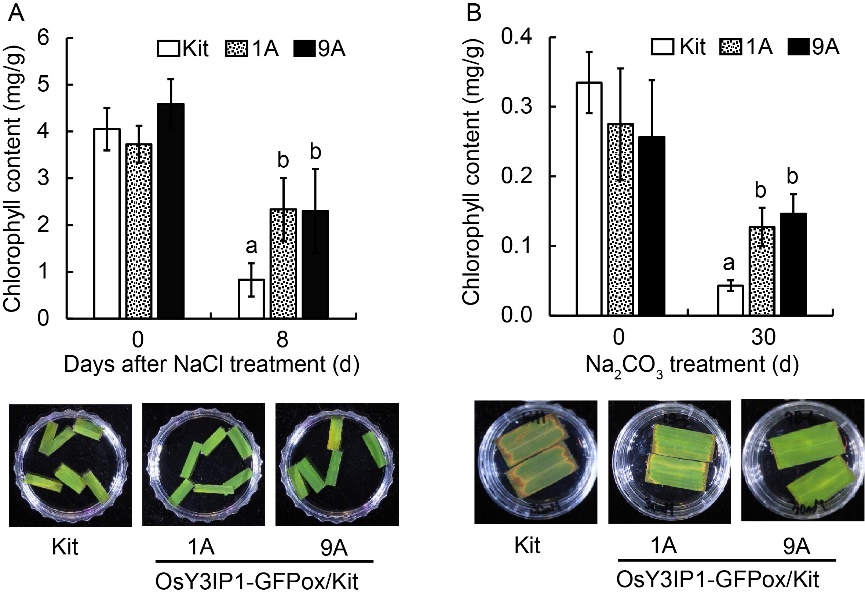
Fig. 6. Total chlorophyll contents of OsY3IP1-GFPox/Kit transgenic lines after saline (A) and alkaline (B) treatments. Quantification of chlorophyll content (upper) and leaf disks (lower) from eight-week- old Kitaake (Kit) and OsY3IP1-GFPox/Kit lines (1A and 9A) after 300 mmol/L NaCl treatment for 8 d and 30 mmol/L Na2CO3 treatment for 3 d. Error bars represent standard error of at least four leaves. Different lowercase letters above the bars indicate significant differences at P < 0.05.
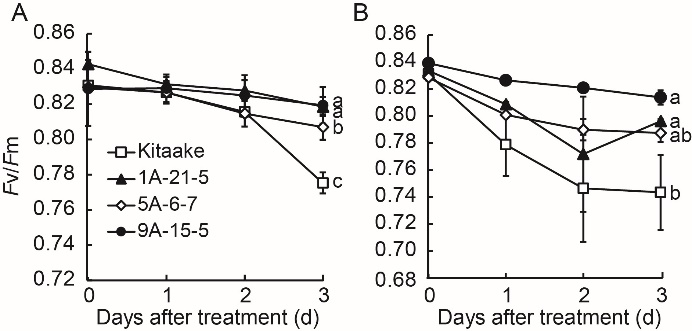
Fig. 7. Maximum quantum yield of PSII (Fv/Fm) in leaves of control (Kitaake) and OsY3IP1-GFPox/Kit T2 lines (1A-21-5, 5A-6-7 and 9A-15-5) under 300 mmol/L NaCl (A) and 30 mmol/L Na2CO3 (B). Error bars represent standard error of at least three leaves from each plant. Different lowercase letters indicate significant differences at P < 0.05.
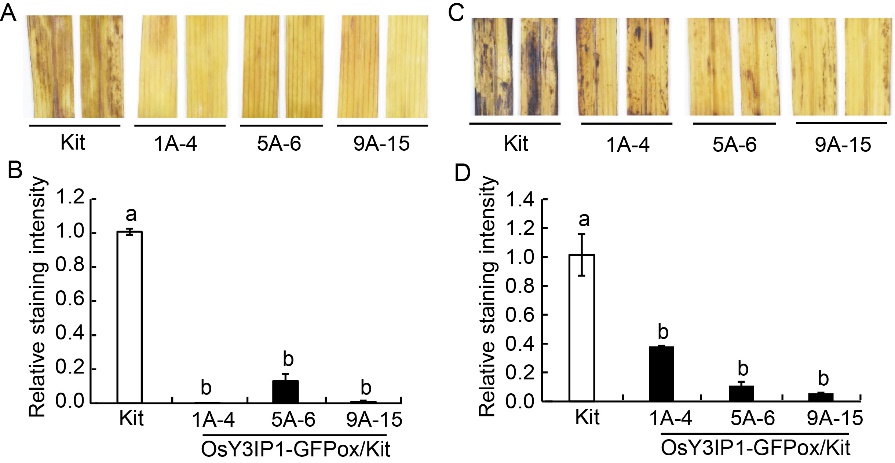
Fig. 8. Reduced accumulation of reactive oxygen species in OsY3IP1-GFPox/Kit transgenic plants (T1, 1A-4, 5A-6 and 9A-15) under saline (700 mmol/L NaCl) and alkaline (50 mmol/L Na2CO3) treatments for 3 d. A and C, Diaminobenzidine tetrahydrochloride (DAB) staining to detect H2O2 in the leaves of Kitaake (Kit) and OsY3IP1-GFPox/Kit T1 lines (1A-4, 5A-6 and 9A-15). Leaf segments were prepared from fully expanded leaf of 10-week-old Kitaake and OsY3IP1-GFPox/Kit. Two representative segments were photographed. B and D, Intensity of DAB staining in leaf segments was determined with the ImageJ software and displayed relative to the level of intensity in Kitaake, which are arbitrarily set at ‘1’. Error bars represent standard error of two leaves from each plant. Different lowercase letters above the bars indicate significant differences at P < 0.05.
| [1] | Albus C A, Ruf S, Schottler M A, Lein W, Kehr J, Bock R. 2010. Y3IP1, a nucleus-encoded thylakoid protein, cooperates with the plastid-encoded Ycf3 protein in photosystem I assembly of tobacco and Arabidopsis. Plant Cell, 22(8): 2838-2855. |
| [2] | Amunts A, Nelson N. 2009. Plant photosystem I design in the light of evolution. Structure, 17(5): 637-650. |
| [3] |
Baker N R. 2008. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu Rev Plant Biol, 59: 89-113.
PMID |
| [4] | Bart R, Chern M, Park C J, Bartley L, Ronald P C. 2006. A novel system for gene silencing using siRNAs in rice leaf and stem- derived protoplasts. Plant Methods, 2: 13. |
| [5] |
Baxter A, Mittler R, Suzuki N. 2014. ROS as key players in plant stress signalling. J Exp Bot, 65(5): 1229-1240.
PMID |
| [6] |
Boudreau E, Takahashi Y, Lemieux C, Turmel M, Rochaix J D. 1997. The chloroplast ycf3 and ycf4 open reading frames of Chlamydomonas reinhardtii are required for the accumulation of the photosystem I complex. EMBO J, 16(20): 6095-6104.
PMID |
| [7] | Chern M, Canlas P E, Fitzgerald H A, Ronald P C. 2005. Rice NRR, a negative regulator of disease resistance, interacts with Arabidopsis NPR1 and rice NH1. Plant J, 43(5): 623-635. |
| [8] | DalCorso G, Pesaresi P, Masiero S, Aseeva E, Schunemann D, Finazzi G, Joliot P, Barbato R, Leister D. 2008. A complex containing PGRL1 and PGR5 is involved in the switch between linear and cyclic electron flow in Arabidopsis. Cell, 132(2): 273-285. |
| [9] | Fan X D, Wang J Q, Yang N, Dong Y Y, Liu L, Wang F W, Wang N, Chen H, Liu W C, Sun Y P, Wu J Y, Li H Y. 2013. Gene expression profiling of soybean leaves and roots under salt, saline-alkali and drought stress by high-throughput Illumina sequencing. Gene, 512(2): 392-402. |
| [10] | Gerona M E B, Deocampo M P, Egdane J A, Ismail A M, Dionisio-Sese M L. 2019. Physiological responses of contrasting rice genotypes to salt stress at reproductive stage. Rice Sci, 26(4): 207-219. |
| [11] | Guo M X, Li S P, Tian S, Wang B, Zhao X S. 2017. Transcriptome analysis of genes involved in defense against alkaline stress in roots of wild jujube (Ziziphus acidojujuba). PLoS One, 12(10): e0185732. |
| [12] | He Y, Fu J L, Yu C L, Wang X M, Jiang Q S, Hong J, Lu K X, Xue G P, Yan C Q, James A, Xu L G, Chen J P, Jiang D A. 2015. Increasing cyclic electron flow is related to Na+ sequestration into vacuoles for salt tolerance in soybean. J Exp Bot, 66(21): 6877-6889. |
| [13] | Hong C Y, Hsu Y T, Tsai Y C, Kao C H. 2007. Expression of ASCORBATE PEROXIDASE 8 in roots of rice (Oryza sativa L.) seedlings in response to NaCl. J Exp Bot, 58(12): 3273-3283. |
| [14] | Horvath E M, Peter S O, Joet T, Rumeau D, Cournac L, Horvath G V, Kavanagh T A, Schafer C, Peltier G, Medgyesy P. 2000. Targeted inactivation of the plastid ndhB gene in tobacco results in an enhanced sensitivity of photosynthesis to moderate stomatal closure. Plant Physiol, 123(4): 1337-1350. |
| [15] | Ichikawa T, Nakazawa M, Kawashima M, Iizumi H, Kuroda H, Kondou Y, Tsuhara Y, Suzuki K, Ishikawa A, Seki M, Fujita M, Motohashi R, Nagata N, Takagi T, Shinozaki K, Matsui M. 2006. The FOX hunting system: An alternative gain-of-function gene hunting technique. Plant J, 48(6): 974-985. |
| [16] |
Inskeep W P, Bloom P R. 1985. Extinction coefficients of chlorophyll a and b in N,N-dimethylformamide and 80% acetone. Plant Physiol, 77(2): 483-485.
PMID |
| [17] | Jordan P, Fromme P, Witt H T, Klukas O, Saenger W, Krauss N. 2001. Three-dimensional structure of cyanobacterial photosystem I at 2.5 A resolution. Nature, 411: 909-917. |
| [18] | Kaur N, Sharma I, Kirat K, Pati P K. 2016. Detection of reactive oxygen species in Oryza sativa L. (rice). Bio-Protocol, 6(24): e2061. |
| [19] | Kondou Y, Higuchi M, Takahashi S, Sakurai T, Ichikawa T, Kuroda H, Yoshizumi T, Tsumoto Y, Horii Y, Kawashima M, Hasegawa Y, Kuriyama T, Matsui K, Kusano M, Albinsky D, Takahashi H, Nakamura Y, Suzuki M, Sakakibara H, Kojima M, Akiyama K, Kurotani A, Seki M, Fujita M, Enju A, Yokotani N, Saitou T, Ashidate K, Fujimoto N, Ishikawa Y, Mori Y, Nanba R, Takata K, Uno K, Sugano S, Natsuki J, Dubouzet J G, Maeda S, Ohtake M, Mori M, Oda K, Takatsuji H, Hirochika H, Matsui M. 2009. Systematic approaches to using the FOX hunting system to identify useful rice genes. Plant J, 57(5): 883-894. |
| [20] | Krech K, Ruf S, Masduki F F, Thiele W, Bednarczyk D, Albus C A, Tiller N, Hasse C, Schottler M A, Bock R. 2012. The plastid genome-encoded Ycf4 protein functions as a nonessential assembly factor for photosystem I in higher plants. Plant Physiol, 159(2): 579-591. |
| [21] | Kura-Hotta M, Satoh K, Katoh S. 1987. Relationship between photosynthesis and chlorophyll content during leaf senescence of rice seedlings. Plant Cell Physiol, 28(7): 1321-1329. |
| [22] |
Lee Y J, Kim D H, Kim Y W, Hwang I. 2001. Identification of a signal that distinguishes between the chloroplast outer envelope membrane and the endomembrane system in vivo. Plant Cell, 13(10): 2175-2190.
PMID |
| [23] | Li G T, Jain R, Chern M, Pham N T, Martin J A, Wei T, Schackwitz W S, Lipzen A M, Duong P Q, Jones K C, Jiang L R, Ruan D L, Bauer D, Peng Y, Barry K W, Schmutz J, Ronald P C. 2017. The sequences of 1504 mutants in the model rice variety Kitaake facilitate rapid functional genomic studies. Plant Cell, 29(6): 1218-1231. |
| [24] | Liu J, Wang Y Q, Li Q T. 2017. Analysis of differentially expressed genes and adaptive mechanisms of Prunus triloba Lindl. under alkaline stress. Hereditas, 154: 10. |
| [25] | Long T A, Okegawa Y, Shikanai T, Schmidt G W, Covert S F. 2008. Conserved role of PROTON GRADIENT REGULATION 5 in the regulation of PSI cyclic electron transport. Planta, 228(6): 907-918. |
| [26] | Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R. 2010. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ, 33(4): 453-467. |
| [27] |
Mittler R. 2002. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci, 7(9): 405-410.
PMID |
| [28] | Munne-Bosch S, Shikanai T, Asada K. 2005. Enhanced ferredoxin- dependent cyclic electron flow around photosystem I and α-tocopherol quinone accumulation in water-stressed ndhB-inactivated tobacco mutants. Planta, 222(3): 502-511. |
| [29] |
Naver H, Boudreau E, Rochaix J D. 2001. Functional studies of Ycf3: Its role in assembly of photosystem I and interactions with some of its subunits. Plant Cell, 13(12): 2731-2745.
PMID |
| [30] |
Nellaepalli S, Ozawa S I, Kuroda H, Takahashi Y. 2018. The photosystem I assembly apparatus consisting of Ycf3-Y3IP1 and Ycf4 modules. Nat Commun, 9(1): 2439.
PMID |
| [31] | Nellaepalli S, Kim R G, Grossman A R, Takahashi Y. 2021. Interplay of four auxiliary factors is required for the assembly of photosystem I reaction center subcomplex. Plant J, 106(4): 1075-1086. |
| [32] | Nelson N, Ben-Shem A. 2004. The complex architecture of oxygenic photosynthesis. Nat Rev Mol Cell Biol, 5(12): 971-982. |
| [33] | Nelson N, Junge W. 2015. Structure and energy transfer in photosystems of oxygenic photosynthesis. Annu Rev Biochem, 84: 659-683. |
| [34] |
Nishikawa Y, Yamamoto H, Okegawa Y, Wada S, Sato N, Taira Y, Sugimoto K, Makino A, Shikanai T. 2012. PGR5-dependent cyclic electron transport around PSI contributes to the redox homeostasis in chloroplasts rather than CO2 fixation and biomass production in rice. Plant Cell Physiol, 53(12): 2117-2126.
PMID |
| [35] | Park C J, Wei T, Sharma R, Ronald P C. 2017. Overexpression of rice auxilin-like protein, XB21, induces necrotic lesions, up-regulates endocytosis-related genes, and confers enhanced resistance to Xanthomonas oryzae pv. oryzae. Rice, 10(1): 27. |
| [36] | Peltier J B, Cai Y, Sun Q, Zabrouskov V, Giacomelli L, Rudella A, Ytterberg A J, Rutschow H, van Wijk K J. 2006. The oligomeric stromal proteome of Arabidopsis thaliana chloroplasts. Mol Cell Proteomics, 5(1): 114-133. |
| [37] | Qin X C, Suga M, Kuang T Y, Shen J R. 2015. Photosynthesis: Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science, 348: 989-995. |
| [38] |
Rohila J S, Chen M, Chen S, Chen J, Cerny R, Dardick C, Canlas P, Xu X, Gribskov M, Kanrar S, Zhu J K, Ronald P, Fromm M E. 2006. Protein-protein interactions of tandem affinity purification- tagged protein kinases in rice. Plant J, 46(1): 1-13.
PMID |
| [39] |
Ruf S, Kossel H, Bock R. 1997. Targeted inactivation of a tobacco intron-containing open reading frame reveals a novel chloroplast- encoded photosystem I-related gene. J Cell Biol, 139(1): 95-102.
PMID |
| [40] | Shafi A, Chauhan R, Gill T, Swarnkar M K, Sreenivasulu Y, Kumar S, Kumar N, Shankar R, Ahuja P S, Singh A K. 2015. Expression of SOD and APX genes positively regulates secondary cell wall biosynthesis and promotes plant growth and yield in Arabidopsis under salt stress. Plant Mol Biol, 87(6): 615-631. |
| [41] | Shikanai T. 2007. Cyclic electron transport around photosystem I: Genetic approaches. Annu Rev Plant Biol, 58: 199-217. |
| [42] | Sonoike K. 2011. Photoinhibition of photosystem I. Physiol Plant, 142(1): 56-64. |
| [43] |
Srivastava A K, Zhang C, Yates G, Bailey M, Brown A, Sadanandom A. 2016. SUMO is a critical regulator of salt stress responses in rice. Plant Physiol, 170(4): 2378-2391.
PMID |
| [44] | Sun J K, He L, Li T. 2019. Response of seedling growth and physiology of Sorghum bicolor (L.) moench to saline-alkali stress. PLoS One, 14(7): e0220340. |
| [45] | Suzuki N, Koussevitzky S, Mittler R, Miller G. 2012. ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ, 35(2): 259-270. |
| [46] | Takabayashi A, Endo T, Shikanai T, Sato F. 2002. Post-illumination reduction of the plastoquinone pool in chloroplast transformants in which chloroplastic NAD(P)H dehydrogenase was inactivated. Biosci Biotechnol Biochem, 66(10): 2107-2111. |
| [47] |
Takahashi S, Murata N. 2008. How do environmental stresses accelerate photoinhibition? Trends Plant Sci, 13(4): 178-182.
PMID |
| [48] | Wang D F, Jr Portis A R. 2007. A novel nucleus-encoded chloroplast protein, PIFI, is involved in NAD(P)H dehydrogenase complex- mediated chlororespiratory electron transport in Arabidopsis. Plant Physiol, 144(4): 1742-1752. |
| [49] | Wang P, Duan W, Takabayashi A, Endo T, Shikanai T, Ye J Y, Mi H L. 2006. Chloroplastic NAD(P)H dehydrogenase in tobacco leaves functions in alleviation of oxidative damage caused by temperature stress. Plant Physiol, 141(2): 465-474. |
| [50] | Xing W, Wang J G, Liu H L, Zou D T, Zhao H W. 2013. Influence of natural saline-alkali stress on chlorophyll content and chloroplast ultrastructure of two contrasting rice (Oryza sativa L. japonica) cultivars. Aust J Crop Sci, 7(2): 289-292. |
| [51] | Yamori W, Shikanai T. 2016. Physiological functions of cyclic electron transport around photosystem I in sustaining photosynthesis and plant growth. Annu Rev Plant Biol, 67: 81-106. |
| [52] | Yamori W, Sakata N, Suzuki Y, Shikanai T, Makino A. 2011. Cyclic electron flow around photosystem I via chloroplast NAD(P)H dehydrogenase (NDH) complex performs a significant physiological role during photosynthesis and plant growth at low temperature in rice. Plant J, 68(6): 966-976. |
| [53] | Yang H X, Liu J, Wen X G, Lu C M.2015. Molecular mechanism of photosystem I assembly in oxygenic organisms. Biochim Biophys Acta, 1847(9): 838-848. |
| [54] |
Yokotani N, Higuchi M, Kondou Y, Ichikawa T, Iwabuchi M, Hirochika H, Matsui M, Oda K. 2011. A novel chloroplast protein, CEST induces tolerance to multiple environmental stresses and reduces photooxidative damage in transgenic Arabidopsis. J Exp Bot, 62(2): 557-569.
PMID |
| [55] | Yu J Q, Wang J H, Sun C H, Zhang Q Y, Hu D G, Hao Y J. 2018. Ectopic expression of the apple nucleus-encoded thylakoid protein MdY3IP1 triggers early-flowering and enhanced salt-tolerance in Arabidopsis thaliana. BMC Plant Biol, 18(1): 18. |
| [56] | Zeng Z M, Xiong F J, Yu X H, Gong X P, Luo J T, Jiang Y D, Kuang H C, Gao B J, Niu X L, Liu Y S. 2016. Overexpression of a glyoxalase gene, OsGly I, improves abiotic stress tolerance and grain yield in rice (Oryza sativa L.). Plant Physiol Biochem, 109: 62-71. |
| [57] |
Zhang H, Liu X L, Zhang R X, Yuan H Y, Wang M M, Yang H Y, Ma H Y, Liu D, Jiang C J, Liang Z W. 2017. Root damage under alkaline stress is associated with reactive oxygen species accumulation in rice (Oryza sativa L.). Front Plant Sci, 8: 1580.
PMID |
| [58] | Zhang Z S, Li G, Gao H Y, Zhang L T, Yang C, Liu P, Meng Q W. 2012. Characterization of photosynthetic performance during senescence in stay-green and quick-leaf-senescence Zea mays L. inbred lines. PLoS One, 7(8): e42936. |
| [59] | Živčák M, Brestič M, Olšovská K, Slamka P. 2008. Performance index as a sensitive indicator of water stress in Triticum aestivum L. Plant Soil Environ, 4: 133-139. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | XIA Xiaodong, ZHANG Xiaobo, WANG Zhonghao, CHENG Benyi, Sun Huifeng, XU Xia, GONG Junyi, YANG Shihua, WU Jianli, SHI Yongfeng, XU Rugen. Mapping and Functional Analysis of LE Gene in a Lethal Etiolated Rice Mutant at Seedling Stage [J]. Rice Science, 2023, 30(6): 13-. |
| [7] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [8] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [9] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [10] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [11] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [12] | Jiang Changjie, Liang Zhengwei, Xie Xianzhi. Priming for Saline-Alkaline Tolerance in Rice: Current Knowledge and Future Challenges [J]. Rice Science, 2023, 30(5): 417-425. |
| [13] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [14] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [15] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||