
Rice Science ›› 2021, Vol. 28 ›› Issue (1): 99-108.DOI: 10.1016/j.rsci.2020.11.011
• Research Papers • Previous Articles
Jan Mehmood, Shah Gulmeena, Yuqing Huang, Xuejiao Liu, Peng Zheng, Hao Du, Hao Chen( ), Jumin Tu
), Jumin Tu
Received:2020-02-14
Accepted:2020-08-08
Online:2021-01-28
Published:2021-01-28
About author:#These authors contributed equally to this study
Jan Mehmood, Shah Gulmeena, Yuqing Huang, Xuejiao Liu, Peng Zheng, Hao Du, Hao Chen, Jumin Tu. Development of Heat Tolerant Two-Line Hybrid Rice Restorer Line Carrying Dominant Locus of OsHTAS[J]. Rice Science, 2021, 28(1): 99-108.
Add to citation manager EndNote|Ris|BibTeX
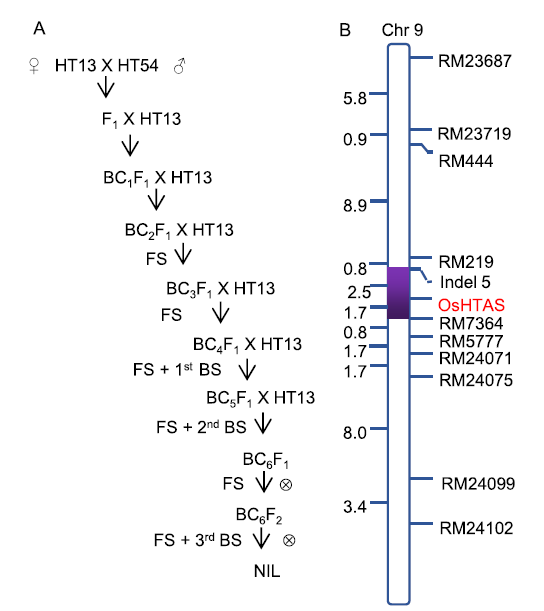
Fig. 1. Schematic diagram of marker-assised backcross breeding and graphical genotype of near isognic line MHT13.A, Flow chart of marker-assisted backcross breeding for developing near isogenic line with the dominant locus of OsHTAS. FS, Foreground selection; BS, Background selection.B, Graphical genotype of MHT13. The purple solid bar represents the foreground fragment containing OsHTAS introduced from HT54, while the hollow vertical bar represents the background genotype inherited from HT13. The background selection was carried out using 144 SSR markers. Chr, Chromosome.
| Line | Growth duration (d) | Plant height (cm) | No. of tillers per plant | Panicle length (cm) | No. of grains per panicle | 1000-grain weight (g) | Spikelet fertility (%) |
|---|---|---|---|---|---|---|---|
| HT54 | 96.0 ± 0.0 | 85.2 ± 1.7 | 8.0 ± 1.4 | 20.8 ± 1.4 | 119.6 ± 5.4 | 28.4 ± 0.9 | 89.8 ± 1.7 |
| HT13 | 126.0 ± 0.0 | 93.1 ± 2.5 | 7.8 ± 0.9 | 23.5 ± 0.8 | 115.1 ± 7.0 | 24.2 ± 0.6 | 93.4 ± 2.6 |
| MHT13 | 126.0 ± 0.0 | 92.8 ± 1.0 | 8.0 ± 1.2 | 23.8 ± 0.9 | 116.8 ± 9.7 | 24.3 ± 1.0 | 92.2 ± 2.9 |
Table 1 Agronomic performance of MHT13 under normal growth conditions (Hangzhou, 2017).
| Line | Growth duration (d) | Plant height (cm) | No. of tillers per plant | Panicle length (cm) | No. of grains per panicle | 1000-grain weight (g) | Spikelet fertility (%) |
|---|---|---|---|---|---|---|---|
| HT54 | 96.0 ± 0.0 | 85.2 ± 1.7 | 8.0 ± 1.4 | 20.8 ± 1.4 | 119.6 ± 5.4 | 28.4 ± 0.9 | 89.8 ± 1.7 |
| HT13 | 126.0 ± 0.0 | 93.1 ± 2.5 | 7.8 ± 0.9 | 23.5 ± 0.8 | 115.1 ± 7.0 | 24.2 ± 0.6 | 93.4 ± 2.6 |
| MHT13 | 126.0 ± 0.0 | 92.8 ± 1.0 | 8.0 ± 1.2 | 23.8 ± 0.9 | 116.8 ± 9.7 | 24.3 ± 1.0 | 92.2 ± 2.9 |
| Cross | Growth duration (d) | Plant height (cm) | No. of tillers per plant | Panicle length (cm) | No. of grains per panicle | 1000-grain weight (g) | Seed-setting rate (%) | Yield per plant (g) |
|---|---|---|---|---|---|---|---|---|
| 2017, Hangzhou, China | ||||||||
| 218S/HT13 | 134.2 ± 1.2 a | 127.5 ± 5.2 a | 14.6 ± 0.1 a | 26.2 ± 1.5 a | 236.8 ± 10.5 a | 27.5 ± 0.2 a | 84.8 ± 4.0 a | 58.83 ± 0.10 b |
| 218S/MHT13 | 134.0 ± 1.1 a | 127.8 ± 2.6 a | 15.5 ± 1.5 a | 27.6 ± 2.1 a | 235.3 ± 13.0 a | 27.0 ± 1.0 a | 87.4 ± 1.4 b | 65.90 ± 1.20 a |
| 208S/HT13 | 135.7 ± 1.3 a | 124.8 ± 0.9 a | 15.2 ± 0.5 a | 25.9 ± 2.5 a | 218.8 ± 5.4 a | 24.5 ± 3.1 a | 85.2 ± 2.0 a | 66.20 ± 0.57 b |
| 208S/MHT13 | 135.7 ± 1.2 a | 125.8 ± 1.9 a | 16.0 ± 1.0 a | 26.1 ± 0.5 a | 227.4 ± 3.5 b | 26.5 ± 0.5 a | 86.8 ± 5.0 a | 70.36 ± 0.28 a |
| 2018, Changxing, China | ||||||||
| 218S/HT13 | 134.3 ± 2.3 a | 127.0 ± 2.2 a | 13.8 ± 1.5 a | 27.1 ± 1.1 a | 243.1 ± 9.3 a | 26.2 ± 1.5 b | 88.0 ± 5.6 a | 64.77 ± 1.30 c |
| 218S/MHT13 | 134.2 ± 1.8 a | 127.0 ± 0.5 a | 15.5 ± 0.7 b | 26.4 ± 2.2 a | 244.4 ± 5.0 a | 27.4 ± 2.0 a | 89.5 ± 2.2 a | 72.03 ± 0.60 a |
| 228S/HT13 | 133.2 ± 2.1 a | 125.4 ± 1.5 a | 15.3 ± 0.7 a | 25.5 ± 3.4 a | 221.2 ± 12.1 a | 27.9 ± 2.2 a | 73.3 ± 0.6 a | 61.52 ± 0.85 a |
| 228S/MHT13 | 133.4 ± 1.6 a | 130.2 ± 3.4 b | 14.7 ± 0.3 a | 26.1 ± 2.0 a | 220.4 ± 2.1 a | 28.4 ± 0.3 a | 75.7 ± 3.3 a | 63.77 ± 0.43 a |
Table 2 Agronomic performance of MHT13-derivative hybrids under field conditions in 2017 and 2018, respectively.
| Cross | Growth duration (d) | Plant height (cm) | No. of tillers per plant | Panicle length (cm) | No. of grains per panicle | 1000-grain weight (g) | Seed-setting rate (%) | Yield per plant (g) |
|---|---|---|---|---|---|---|---|---|
| 2017, Hangzhou, China | ||||||||
| 218S/HT13 | 134.2 ± 1.2 a | 127.5 ± 5.2 a | 14.6 ± 0.1 a | 26.2 ± 1.5 a | 236.8 ± 10.5 a | 27.5 ± 0.2 a | 84.8 ± 4.0 a | 58.83 ± 0.10 b |
| 218S/MHT13 | 134.0 ± 1.1 a | 127.8 ± 2.6 a | 15.5 ± 1.5 a | 27.6 ± 2.1 a | 235.3 ± 13.0 a | 27.0 ± 1.0 a | 87.4 ± 1.4 b | 65.90 ± 1.20 a |
| 208S/HT13 | 135.7 ± 1.3 a | 124.8 ± 0.9 a | 15.2 ± 0.5 a | 25.9 ± 2.5 a | 218.8 ± 5.4 a | 24.5 ± 3.1 a | 85.2 ± 2.0 a | 66.20 ± 0.57 b |
| 208S/MHT13 | 135.7 ± 1.2 a | 125.8 ± 1.9 a | 16.0 ± 1.0 a | 26.1 ± 0.5 a | 227.4 ± 3.5 b | 26.5 ± 0.5 a | 86.8 ± 5.0 a | 70.36 ± 0.28 a |
| 2018, Changxing, China | ||||||||
| 218S/HT13 | 134.3 ± 2.3 a | 127.0 ± 2.2 a | 13.8 ± 1.5 a | 27.1 ± 1.1 a | 243.1 ± 9.3 a | 26.2 ± 1.5 b | 88.0 ± 5.6 a | 64.77 ± 1.30 c |
| 218S/MHT13 | 134.2 ± 1.8 a | 127.0 ± 0.5 a | 15.5 ± 0.7 b | 26.4 ± 2.2 a | 244.4 ± 5.0 a | 27.4 ± 2.0 a | 89.5 ± 2.2 a | 72.03 ± 0.60 a |
| 228S/HT13 | 133.2 ± 2.1 a | 125.4 ± 1.5 a | 15.3 ± 0.7 a | 25.5 ± 3.4 a | 221.2 ± 12.1 a | 27.9 ± 2.2 a | 73.3 ± 0.6 a | 61.52 ± 0.85 a |
| 228S/MHT13 | 133.4 ± 1.6 a | 130.2 ± 3.4 b | 14.7 ± 0.3 a | 26.1 ± 2.0 a | 220.4 ± 2.1 a | 28.4 ± 0.3 a | 75.7 ± 3.3 a | 63.77 ± 0.43 a |
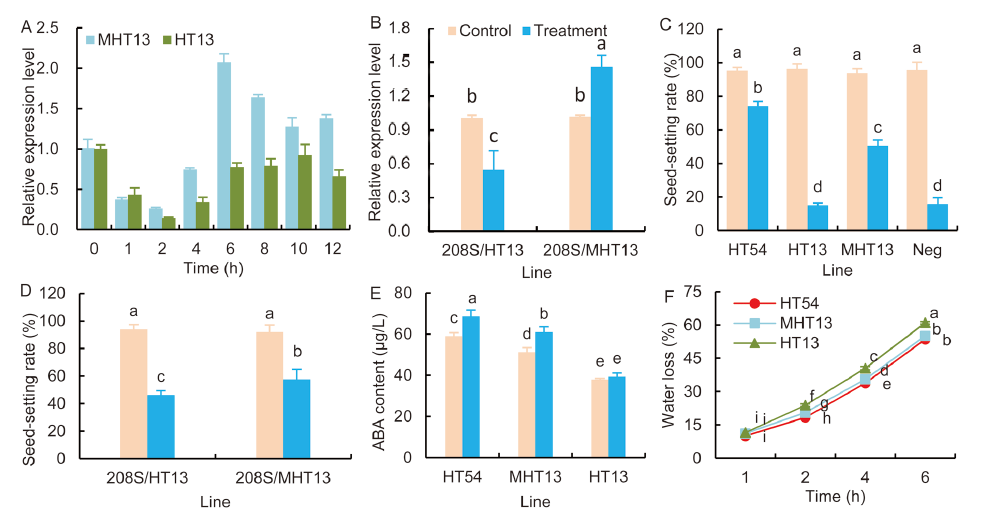
Fig. 2. Dynamic expression, phenotype and heat tolerance mechanism of OsHTAS in MHT13 and its derivative hybrids.A, Dynamic expression features of OsHTAS under 48 ºC high temperature.B, Expression changes of OsHTAS in hybrids of 208S/HT13 and 208S/MHT13 before and after 48 ºC high temperature treatment.C, Changes of seed-setting rate of heat resistant and sensitive parents and their continuously-backcrossing obtained MHT13 and negative materials before and after 38 ºC high temperature treatment.D, Changes of seed-setting rate of heat-sensitive HT13 and its improved MHT13-formulated hybrids before and after 38 ºC high temperature treatment.E, ABA content of MHT13 and its parental lines before and after 48 ºC high temperature treatment.F, Changes of water loss before and after 48 ºC high temperature treatment.High temperature (48 ºC) treatment lasting 79 h was carried out at the seedling stage, and heat stress response was then scored and photographed after end of treatment and recovery for 10 d. High temperature (38 ºC) treatment was carried out on the day of flowering, the treatment lasted 6 h and the seed-setting rate was counted after maturity. Error bars indicate SE based on three biological replicates. Significant differences between lines were determined by the Duncan’s multiple range test at the 0.05 level.

Fig. 3. High temperature tolerance of MHT13 at seedling stage. Neg represents the negative line obtained by marker-assisted breeding. The treatment used was heat stress at 48 ºC for 79 h.
| Index | BRR (%) | HMRR (%) | GL (mm) | CD (%) | TD (Rank) | ASV (Rank) | GC (mm) | AC (%) | MRR (%) | GLWR | CGR (%) | PC (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grade 3 standard | ≥ 77.0 | ≥ 52.0 | > 6.5, 5.6‒6.5 or < 5.6 a | ≤ 5.0 | ≤ 2 | ≥ 5.0 | ≥ 50 | 13.0-22.0 | - | - | - | - |
| Test result | 80.6 | 64.3 | 6.7 | 3.3 | 2 | 7.0 | 58 | 15.0 | 72.3 | 3.3 | 13 | 8.36 |
| Single judgment | 2 | 1 | Long grain | 3 | 2 | 1 | 3 | 1 | - | - | - | - |
Table 3 Evaluation results of grain quality of MHT13 according to quality standard of edible indica rice issued by the Ministry of Agriculture of China.
| Index | BRR (%) | HMRR (%) | GL (mm) | CD (%) | TD (Rank) | ASV (Rank) | GC (mm) | AC (%) | MRR (%) | GLWR | CGR (%) | PC (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grade 3 standard | ≥ 77.0 | ≥ 52.0 | > 6.5, 5.6‒6.5 or < 5.6 a | ≤ 5.0 | ≤ 2 | ≥ 5.0 | ≥ 50 | 13.0-22.0 | - | - | - | - |
| Test result | 80.6 | 64.3 | 6.7 | 3.3 | 2 | 7.0 | 58 | 15.0 | 72.3 | 3.3 | 13 | 8.36 |
| Single judgment | 2 | 1 | Long grain | 3 | 2 | 1 | 3 | 1 | - | - | - | - |
| [1] | Aghamolki M T K, Yusop M K, Oad F C, Zakikhani H, Jaafar H Z, Kharidah S, Musa M H. 2014. Heat stress effects on yield parameters of selected rice cultivars at reproductive growth stages. J Food Agric Environ, 12: 741-746. |
| [2] | Alberio C, Aguirrezábal L A, Izquierdo N G, Reid R, Zuil S, Zambelli A. 2018. Effect of genetic background on the stability of sunflower fatty acid composition in different high oleic mutations. J Food Agric Environ, 98(11): 4074-4084. |
| [3] | Cao Z B, Li Y, Zeng B H, Mao L H, Cai Y H, Wu X F, Yuan L F. 2020. QTL mapping for heat tolerance of chalky grain rate of Oryza glaberrima Steud. Chin J Rice Sci, 34(2): 135-142. (in Chinese with English abstract) |
| [4] | Challinor A J, Watson J, Lobell D B, Howden S M, Smith D R, Chhetri N. 2014. A meta-analysis of crop yield under climate change and adaptation. Nat Clim Change, 4: 287-291. |
| [5] | Cheabu S, Panichawong N, Rattanametta P, Wasuri B, Kasemsap P, Arikit S, Vanavichit A, Malumpong C. 2019. Screening for spikelet fertility and validation of heat tolerance in a large rice mutant population. Rice Sci, 26(4): 229-238. |
| [6] | Cheng L R, Wang J M, Uzokwe V, Meng L J, Wang Y, Sun Y, Zhu L H, Xu J L, Li Z K. 2012. Genetic analysis of cold tolerance at seedling stage and heat tolerance at anthesis in rice (Oryza sativa L.). J Integr Agric, 11(3): 359-367. |
| [7] | Collard B C, Mackill D J. 2008. Marker-assisted selection: An approach for precision plant breeding in the twenty-first century. Philos Trans Royal Soc B Biol Sci, 363: 557-572. |
| [8] | Dellaporta S L, Wood J, Hicks J B. 1983. A plant DNA minipreparation: Version II. Plant Mol Biol Rep, 1: 19-21. |
| [9] | Driedonks N, Rieu I, Vriezen W H. 2016. Breeding for plant heat tolerance at vegetative and reproductive stages. Plant Reprod, 29: 67-79. |
| [10] | El-Esawi M A, Alayafi A A. 2019. Overexpression of rice Rab7 gene improves drought and heat tolerance and increases grain yield in rice(Oryza sativa L.). Genes, 10(1): 56. |
| [11] | Fahad S, Hussain S, Saud S, Hassan S, Tanveer M, Ihsan M Z, Shah A N, Ullah A, Nasrullah, Khan F, Ullah S, Alharby H F, Nasim W, Wu C, Huang J L. 2016. A combined application of biochar and phosphorus alleviates heat-induced adversities on physiological, agronomical and quality attributes of rice. Plant Physiol Biochem, 103: 191-198. |
| [12] | Fang Y J, Liao K F, Du H, Xu Y, Song H Z, Li X H, Xiong L Z. 2015. A stress-responsive NAC transcription factor SNAC3 confers heat and drought tolerance through modulation of reactive oxygen species in rice. J Exp Bot, 66(21): 6803-6817. |
| [13] | Feng H Y, Jiang H L, Wang Meng, Tang X R, Duan M Y, Pan S G, Tian H, Wang S L, Mo Z W. 2019. Morphophysiological responses of different scented rice varieties to high temperature at seedling stage. Chin J Rice Sci, 33(1): 68-74. (in Chinese with English abstract) |
| [14] | Figueiredo N, Carranca C, Trindade H, Pereira J, Goufo P, Coutinho J, Marques P, Maricato R, de Varennes A. 2015. Elevated carbon dioxide and temperature effects on rice yield, leaf greenness, and phenological stages duration. Paddy Water Environ, 13: 313-324. |
| [15] | Gallois J L, Moury B, German-Retana S. 2018. Role of the genetic background in resistance to plant viruses. Int J Mol Sci, 19(10): 2856. |
| [16] | Jagadish S V K, Cairns J, Lafitte R, Wheeler T R, Price A H, Craufurd P Q. 2010. Genetic analysis of heat tolerance at anthesis in rice. Crop Sci, 50(5): 1633-1641. |
| [17] | Kilasi N L, Singh J, Vallejos C E, Ye C R, Jagadish S V K, Kusolwa P, Rathinasabapathi B. 2018. Heat stress tolerance in rice (Oryza sativa L.): Identification of quantitative trait loci and candidate genes for seedling growth under heat stress. Front Plant Sci, 9: 1578. |
| [18] | Kunimitsu Y, Iizumi T, Yokozawa M. 2014. Is long-term climate change beneficial or harmful for rice total factor productivity in Japan: Evidence from a panel data analysis. Paddy Water Environ, 12: 213-225. |
| [19] | Lei D Y, Tan L B, Liu F X, Chen L Y, Sun C Q. 2013. Identification of heat-sensitive QTL derived from common wild rice (Oryza rufipogon Griff.). Plant Sci, 201/202: 121-127. |
| [20] | Lesk C, Rowhani P, Ramankutty N. 2016. Influence of extreme weather disasters on global crop production. Nature, 529: 84-87. |
| [21] | Li X M, Chao D Y, Wu Y, Huang X H, Chen K, Cui L G, Su L, Ye W W, Chen H, Chen H C, Dong N Q, Guo T, Shi M, Feng Q, Zhang P, Han B, Shan J X, Gao J P, Lin H X. 2015. Natural alleles of a proteasome α2 subunit gene contribute to thermo- tolerance and adaptation of African rice. Nat Genet, 47: 827-833. |
| [22] | Li X, Lawas L M F, Malo R, Glaubitz U, Erban A, Mauleon R, Heuer S, Zuther E, Kopka J, Hincha D K, Jagadish K S V. 2015. Metabolic and transcriptomic signatures of rice floral organs reveal sugar starvation as a factor in reproductive failure under heat and drought stress. Plant Cell Environ, 38(10): 2171-2192. |
| [23] | Liu J P, Sun X J, Xu F Y, Zhang Y J, Zhang Q, Miao R, Zhang J H, Liang J S, Xu W F. 2018. Suppression of OsMDHAR4 enhances heat tolerance by mediating H2O2-induced stomatal closure in rice plants. Rice, 11: 38. |
| [24] | Liu J P, Zhang C C, Wei C C, Liu X, Wang M G, Yu F F, Xie Q, Tu J M. 2016. The RING finger ubiquitin E3 ligase OsHTAS enhances heat tolerance by promoting H2O2-induced stomatal closure in rice. Plant Physiol, 170(1): 429-443. |
| [25] | Liu X H, Lyu Y S, Yang W P, Yang Z T, Lu S J, Liu J X. 2019. A membrane-associated NAC transcription factor OsNTL3 is involved in thermo tolerance in rice. Plant Biotechnol J, 18(5): 1317-1329. |
| [26] | Livak K J, Schmittgen T D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods, 25(4): 402-408. |
| [27] | Ministry of Agriculture of the People’s Republic of China. 2013. Cooking Rice Variety Quality. Agricultural Industry Standard of the People’s Republic of China. NY/T 593-2013. |
| [28] | Mishra N, Srivastava A P, Esmaeili N, Hu W J, Shen G X. 2018. Overexpression of the rice gene OsSIZ1 in Arabidopsis improves drought-, heat-, and salt-tolerance simultaneously. PLoS One, 13(8): e0201716. |
| [29] | Nakurte I, Keisa A, Rostoks N. 2012. Development and validation of a reversed-phase liquid chromatography method for the simultaneous determination of indole-3-acetic acid, indole-3- pyruvic acid, and abscisic acid in barley (Hordeum vulgare L.). J Anal Methods Chem, 2012: 103575. |
| [30] | Panaud O, Chen X, McCouch S. 1996. Development of microsatellite markers and characterization of simple sequence length polymorphism (SSLP) in rice (Oryza sativa L.). Mol Gen Genet, 252(5): 597-607. |
| [31] | Chen Q Q, Yu S B, Li C H. 2008. Identification of QTLs for heat tolerance at flowering stage in rice. Sci Agric Sin, 41(2): 315-321. |
| [32] | Sato H, Todaka D, Kudo M, Mizoi J, Kidokoro S, Zhao Y, Shinozaki K, Yamaguchi-Shinozaki K. 2016. The Arabidopsis transcriptional regulator DPB 3-1 enhances heat stress tolerance without growth retardation in rice. Plant Biotechnol J, 14(8): 1756-1767. |
| [33] | Shandil R K, Chakrabarti S K, Singh B P, Sharma S, Sundaresha S, Kaushik S K, Bhatt A K, Sharma N N. 2017. Genotypic background of the recipient plant is crucial for conferring RB gene mediated late blight resistance in potato. BMC Genet, 18(1): 22. |
| [34] | Shanmugavadivel P S, Sv A M, Prakash C, Mk R, Tiwari R, Mohapatra T, Singh N K. 2017. High resolution mapping of QTLs for heat tolerance in rice using a 5K SNP array. Rice, 10: 28. |
| [35] | Shen H, Zhong X B, Zhao F F, Wang Y M, Yan B X, Li Q, Chen G Y, Mao B Z, Wang J J, Li Y S, Xiao G Y, He Y K, Xiao H, Li J M, He Z H. 2015. Overexpression of receptor-like kinase ERECTA improves thermotolerance in rice and tomato. Nat Biotechnol, 33: 996-1003. |
| [36] | Shi W J, Yin X Y, Struik P C, Solis C, Xie F M, Schmidt R C, Huang M, Zou Y B, Ye C R, Jagadish S V K. 2017. High day- and night-time temperatures affect grain growth dynamics in contrasting rice genotypes. J Exp Bot, 68(18): 5233-5245. |
| [37] | Wei H, Liu J P, Wang Y, Huang N R, Zhang X B, Wang L C, Zhang J W, Tu J M, Zhong X H. 2013. A dominant major locus in chromosome 9 of rice (Oryza sativa L.) confers tolerance to 48 ºC high temperature at seedling stage. J Hered, 104(2): 287-294. |
| [38] | Xu Y Y, Ramanathan V, Victor D G. 2018. Global warming will happen faster than we think. Nature, 564: 30-32. |
| [39] | Ye C R, Argayoso M A, Redoña E D, Sierra S N, Laza M A, Dilla C J, Mo Y J, Thomson M J, Chin J H, Delaviña C B, Diaz G Q, Hernandez J E. 2012. Mapping QTL for heat tolerance at flowering stage in rice using SNP markers. Plant Breeding, 131(1): 33-41. |
| [40] | Ye C R, Tenorio F A, Argayoso M A, Laza M A, Koh H J, Redona E D, Jagadish K S V, Gregorio G B. 2015a. Identifying and confirming quantitative trait loci associated with heat tolerance at flowering stage in different rice populations. BMC Genet, 16: 41. |
| [41] | Ye C R, Tenorio F A, Redoña E D, Morales-Cortezano P S, Cabrega G A, Jagadish K S V, Gregorio G B. 2015b. Fine- mapping and validating qHTSF4.1 to increase spikelet fertility under heat stress at flowering in rice. Theor Appl Genet, 128: 1507-1517. |
| [42] | Zhang C C, Yuan W Y, Zhang Q F. 2012. RPL1, a gene involved in epigenetic processes regulates phenotypic plasticity in rice. Mol Plant, 5(2): 482-493. |
| [43] | Zhang Y E, Xu W Y, Li Z H, Deng X W, Wu W H, Xue Y B. 2008. F-box protein DOR functions as a novel inhibitory factor for abscisic acid-induced stomatal closure under drought stress in Arabidopsis. Plant Physiol, 148(4): 2121-2133. |
| [44] | Zhao C, Liu B, Piao S L, Wang X H, Lobell D B, Huang Y, Huang M T, Yao Y T, Bassu S, Ciais P, Durand J L, Elliott J, Ewert F, Janssens I A, Li T, Lin E, Liu Q, Martre P, Muller C, Peng S S, Penuelas J, Ruane A C, Wallach D, Wang T, Wu D H, Liu Z, Zhu Y, Zhu Z C, Asseng S. 2017. Temperature increase reduces global yields of major crops in four independent estimates. Proc Natl Acad Sci USA, 114: 9326-9331. |
| [45] | Zhao L, Lei J G, Huang Y J, Zhu S, Chen H P, Huang R L, Peng Z Q, Tu Q H, Shen X H, Yan S. 2016. Mapping quantitative trait loci for heat tolerance at anthesis in rice using chromosomal segment substitution lines. Breeding Sci, 66(3): 358-366. |
| [46] | Zhao Z G, Jiang L, Xiao Y H, Zhang W W, Zhai H Q, Wan J M. 2006. Identification of QTLs for heat tolerance at the booting stage in rice (Oryza sativa L.) Acta Agron Sin, 32(5): 640-644. |
| [47] | Zhou Y, Cao Y L, Huang Y, Xie W B, Xu C G, Li X H, Wang S P. 2009. Multiple gene loci affecting genetic background-controlled disease resistance conferred by R gene Xa3/Xa26 in rice. Theor Appl Genet, 120(1): 127-138. |
| [48] | Zhu S, Huang R L, Wai H P, Xiong H L, Shen X H, He H H, Yan S. 2017. Mapping quantitative trait loci for heat tolerance at the booting stage using chromosomal segment substitution lines in rice. Physiol Mol Biol Plants, 23: 817-825. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [13] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| [14] | Lu Xuedan, Li Fan, Xiao Yunhua, Wang Feng, Zhang Guilian, Deng Huabing, Tang Wenbang. Grain Shape Genes: Shaping the Future of Rice Breeding [J]. Rice Science, 2023, 30(5): 379-404. |
| [15] | Zhang Guomei, Li Han, Liu Shanshan, Zhou Xuming, Lu Mingyang, Tang Liang, Sun Lihua. Water Extract of Rice False Smut Balls Activates Nrf2/HO-1 and Apoptosis Pathways, Causing Liver Injury [J]. Rice Science, 2023, 30(5): 473-485. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||