
Rice Science ›› 2023, Vol. 30 ›› Issue (1): 15-35.DOI: 10.1016/j.rsci.2022.08.002
• Review • Previous Articles Next Articles
M. Iqbal R. Khan1( ), Sarika Kumari1, Faroza Nazir1, Risheek Rahul Khanna1, Ravi Gupta2, Himanshu Chhillar1
), Sarika Kumari1, Faroza Nazir1, Risheek Rahul Khanna1, Ravi Gupta2, Himanshu Chhillar1
Received:2022-05-04
Accepted:2022-08-04
Online:2023-01-28
Published:2022-11-11
Contact:
M. Iqbal R. Khan
About author:First author contact:This is an open access article under the CC BY-NC-ND license (
Peer review under responsibility of China National Rice Research Institute
M. Iqbal R. Khan, Sarika Kumari, Faroza Nazir, Risheek Rahul Khanna, Ravi Gupta, Himanshu Chhillar. Defensive Role of Plant Hormones in Advancing Abiotic Stress-Resistant Rice Plants[J]. Rice Science, 2023, 30(1): 15-35.
Add to citation manager EndNote|Ris|BibTeX
| Plant hormone | Abiotic stress and its condition | Morphological, physiological, and molecular effects | Reference |
| Auxin | Drought stress SMC between 7.3% ± 0.5% and 6.7% ± 0.2% Heat stress (40.3 ºC ± 0.36 ºC and 37.7 ºC± 0.35 ºC) | Improve pollen viability, spikelet fertility, grain number per panicle, grain weight per panicle, 1000-grain weight, and grain yield per panicle; enhance chlorophyll content, delay senescence, and maintain greenness; lower lipid peroxidation and ROS accumulation while improving membrane stability; improve the expression levels of auxin signalling genes (TIR1, AFB2, SPL, TFL2 and HAF) | Sharma et al, |
| As stress (NaAsO2, 150 µmol/L) Se stress (Na2SeO4, 20 µmol/L) | I. Auxin (As and Se stress): Enhance root and shoot growths II. Auxin + Se (As stress): Improve chlorophyll content, proline and cysteine; lower protein content inhibition and DNA damage; reduce lipid peroxidation | Pandey and Gupta, | |
| Gibberellic acid (GA) | Drought stress (PEG6000, 15%) | Improve germination vigor, shoot length, root length, α-amylase activity, and soluble sugar content; induce the expression levels of α-amylase and expansion-related genes | Li J Z et al, 2019 |
| Salt stress (NaCl: 0, 50, 100, 150 and 200 mmol/L) | Enhance contents of proline, soluble sugar and protein as well as inorganic nutrients (K, Mg and Ca); increase panicle number, panicle length, grain yield per plant, 1000-grain weight, filled grain number per panicle, and harvest index | Misratia et al, | |
| Cold stress (10 ºC to 20 ºC) | Improve cold resistance and increase yield traits; promote seedling emergence and vigor | Chen et al, | |
| Abscisic acid (ABA) | Drought stress (PEG6000, 15%) | Enhance net photosynthesis, stomatal conductance, and transpiration rate; improve expression levels of chloroplast genes (OsPsbD1 and OsPsbD2); up-regulate expression levels of ABA biosynthesis genes (OsNCED2, OsNCED3, OsNCED4 and OsNCED5) | Teng et al, |
| Cold stress at 5 ºC | Improve the survival ratio | Lee et al, | |
| Salicylic acid (SA) | Salt stress (NaCl: 0, 100, 200, 300 and 400 mmol/L) | Enhance germination rate, root length, shoot length and plant height; improve the number of grains per panicle, number of filled grains per panicle, 1000-grain weight, the number of panicles per plant, and yield; increase contents of carbohydrate and protein along with minerals (Ca, P and K) while decreasing Na and Cl contents as well as Na+ and Cl- accumulation; enhance endogenous SA content | Jini and Joseph, |
| Cd stress (CdCl2, 50 µmol/L) | Improve root growth; reduce ROS level, and membrane damage; enhance SOD, POD and CAT activities as well as GSH, and AsA contents; improve non-protein thiols’ concentration | Guo et al, | |
| Pb stress (0.05, 0.15 and 0.25 mmol/L) | Increase seedling shoot, and root length; improve chlorophyll content; reduce peroxide levels; alter SOD and APX activities | Chen et al, | |
| Heat stress (40 ºC for 10 d) | Improve pollen viability, and seed setting; lower ROS generation, and MDA content in anthers; differently alter SOD, POD, CAT and APX activities in anthers | Zhang et al, | |
| Brassinosteroid (BR) | Salt stress (NaCl: 0, 75, 100 and 125 mmol/L) | Enhance shoot length, root length, root number, fresh weight and dry weight; improve total chlorophyll, protein, and proline contents; lower lipid peroxidation while enhance activities of enzymatic antioxidants (SOD, APX, CAT, GR, GPX, DHAR and MDHAR); improve expression levels of antioxidant enzyme genes | Sharma I et al, |
| Auxin precursor | Cd stress (30 mg/kg) | Enhance plant height and yield; increase the numbers of tillers, panicles and grain weight; decrease Cd content in grains | Farooq et al, |
| Methyl jasmonate | Cd stress [Cd(NO3)2, 50 µmol/L] | Improve growth of root and shoot, and chlorophyll content; lower electrolyte leakage, lipid peroxidation, and ROS generation; decrease Cd uptake and accumulation; enhance GSH content, and activities of CAT, GR, POD and SOD | Singh and Shah, |
| As stress (NaAsO2, 25 µmol/L) | Improve root growth, and seedling biomass; decrease As accumulation; enhance photosynthetic rates, and chlorophyll content; lower membrane damage, electrolyte leakage, and lipid peroxidation; alter gene expression levels of JA signalling genes (OsCOI1 and OsJAZ3), JA responsive transport factor (OsMYC2), as well as As uptake, translocation and detoxification genes (OsLsi1, OsLsi2, OsNIP1;1, OsNIP3;1, OsLsi6, OsINT5, OsNRAMP1, OsPCS2 and OsABCC2) | Verma et al, | |
| BR and BR mimic | Heat stress (47 ºC for 2 h) | Improve shoot and root biomass; enhance photosynthetic pigment content, RWC and sugar content; improve stomatal conductivity and CO2 assimilation; reduce ROS generation and membrane damage | Thussagunpanit et al, |
| Brassinolide | Cold stress for 15 ºC | Promote cell elongation | Fujii and Saka, |
| As, Arsenic; AsA, Ascorbate; APX, Ascorbate peroxidase; Ca, Calcium; CAT, Catalase; Cd, Cadmium; DHAR, Dehydroascorbate reductase; GR, Glutathione reductase; GPX, Glutathione peroxidase; GSH, Glutathione; IAA, Indole-3-acetic acid; MDHAR, Monodehydroascorbate reductase; MDA, Malondialdehyde; Mg, Magnesium; P, Phosphorous; Pb, Plumbum; PEG, Polyethylene glycol; POX, Peroxidase; ROS; Reactive oxygen species; Se, Selenium; SMC, Soil moisture content; SOD, Superoxide dismutase; POD, Peroxidase; RWC, Relative water content. | |||
Table 1. Exogenous application of various plant hormones and their impacts on abiotic stress amelioration in rice.
| Plant hormone | Abiotic stress and its condition | Morphological, physiological, and molecular effects | Reference |
| Auxin | Drought stress SMC between 7.3% ± 0.5% and 6.7% ± 0.2% Heat stress (40.3 ºC ± 0.36 ºC and 37.7 ºC± 0.35 ºC) | Improve pollen viability, spikelet fertility, grain number per panicle, grain weight per panicle, 1000-grain weight, and grain yield per panicle; enhance chlorophyll content, delay senescence, and maintain greenness; lower lipid peroxidation and ROS accumulation while improving membrane stability; improve the expression levels of auxin signalling genes (TIR1, AFB2, SPL, TFL2 and HAF) | Sharma et al, |
| As stress (NaAsO2, 150 µmol/L) Se stress (Na2SeO4, 20 µmol/L) | I. Auxin (As and Se stress): Enhance root and shoot growths II. Auxin + Se (As stress): Improve chlorophyll content, proline and cysteine; lower protein content inhibition and DNA damage; reduce lipid peroxidation | Pandey and Gupta, | |
| Gibberellic acid (GA) | Drought stress (PEG6000, 15%) | Improve germination vigor, shoot length, root length, α-amylase activity, and soluble sugar content; induce the expression levels of α-amylase and expansion-related genes | Li J Z et al, 2019 |
| Salt stress (NaCl: 0, 50, 100, 150 and 200 mmol/L) | Enhance contents of proline, soluble sugar and protein as well as inorganic nutrients (K, Mg and Ca); increase panicle number, panicle length, grain yield per plant, 1000-grain weight, filled grain number per panicle, and harvest index | Misratia et al, | |
| Cold stress (10 ºC to 20 ºC) | Improve cold resistance and increase yield traits; promote seedling emergence and vigor | Chen et al, | |
| Abscisic acid (ABA) | Drought stress (PEG6000, 15%) | Enhance net photosynthesis, stomatal conductance, and transpiration rate; improve expression levels of chloroplast genes (OsPsbD1 and OsPsbD2); up-regulate expression levels of ABA biosynthesis genes (OsNCED2, OsNCED3, OsNCED4 and OsNCED5) | Teng et al, |
| Cold stress at 5 ºC | Improve the survival ratio | Lee et al, | |
| Salicylic acid (SA) | Salt stress (NaCl: 0, 100, 200, 300 and 400 mmol/L) | Enhance germination rate, root length, shoot length and plant height; improve the number of grains per panicle, number of filled grains per panicle, 1000-grain weight, the number of panicles per plant, and yield; increase contents of carbohydrate and protein along with minerals (Ca, P and K) while decreasing Na and Cl contents as well as Na+ and Cl- accumulation; enhance endogenous SA content | Jini and Joseph, |
| Cd stress (CdCl2, 50 µmol/L) | Improve root growth; reduce ROS level, and membrane damage; enhance SOD, POD and CAT activities as well as GSH, and AsA contents; improve non-protein thiols’ concentration | Guo et al, | |
| Pb stress (0.05, 0.15 and 0.25 mmol/L) | Increase seedling shoot, and root length; improve chlorophyll content; reduce peroxide levels; alter SOD and APX activities | Chen et al, | |
| Heat stress (40 ºC for 10 d) | Improve pollen viability, and seed setting; lower ROS generation, and MDA content in anthers; differently alter SOD, POD, CAT and APX activities in anthers | Zhang et al, | |
| Brassinosteroid (BR) | Salt stress (NaCl: 0, 75, 100 and 125 mmol/L) | Enhance shoot length, root length, root number, fresh weight and dry weight; improve total chlorophyll, protein, and proline contents; lower lipid peroxidation while enhance activities of enzymatic antioxidants (SOD, APX, CAT, GR, GPX, DHAR and MDHAR); improve expression levels of antioxidant enzyme genes | Sharma I et al, |
| Auxin precursor | Cd stress (30 mg/kg) | Enhance plant height and yield; increase the numbers of tillers, panicles and grain weight; decrease Cd content in grains | Farooq et al, |
| Methyl jasmonate | Cd stress [Cd(NO3)2, 50 µmol/L] | Improve growth of root and shoot, and chlorophyll content; lower electrolyte leakage, lipid peroxidation, and ROS generation; decrease Cd uptake and accumulation; enhance GSH content, and activities of CAT, GR, POD and SOD | Singh and Shah, |
| As stress (NaAsO2, 25 µmol/L) | Improve root growth, and seedling biomass; decrease As accumulation; enhance photosynthetic rates, and chlorophyll content; lower membrane damage, electrolyte leakage, and lipid peroxidation; alter gene expression levels of JA signalling genes (OsCOI1 and OsJAZ3), JA responsive transport factor (OsMYC2), as well as As uptake, translocation and detoxification genes (OsLsi1, OsLsi2, OsNIP1;1, OsNIP3;1, OsLsi6, OsINT5, OsNRAMP1, OsPCS2 and OsABCC2) | Verma et al, | |
| BR and BR mimic | Heat stress (47 ºC for 2 h) | Improve shoot and root biomass; enhance photosynthetic pigment content, RWC and sugar content; improve stomatal conductivity and CO2 assimilation; reduce ROS generation and membrane damage | Thussagunpanit et al, |
| Brassinolide | Cold stress for 15 ºC | Promote cell elongation | Fujii and Saka, |
| As, Arsenic; AsA, Ascorbate; APX, Ascorbate peroxidase; Ca, Calcium; CAT, Catalase; Cd, Cadmium; DHAR, Dehydroascorbate reductase; GR, Glutathione reductase; GPX, Glutathione peroxidase; GSH, Glutathione; IAA, Indole-3-acetic acid; MDHAR, Monodehydroascorbate reductase; MDA, Malondialdehyde; Mg, Magnesium; P, Phosphorous; Pb, Plumbum; PEG, Polyethylene glycol; POX, Peroxidase; ROS; Reactive oxygen species; Se, Selenium; SMC, Soil moisture content; SOD, Superoxide dismutase; POD, Peroxidase; RWC, Relative water content. | |||
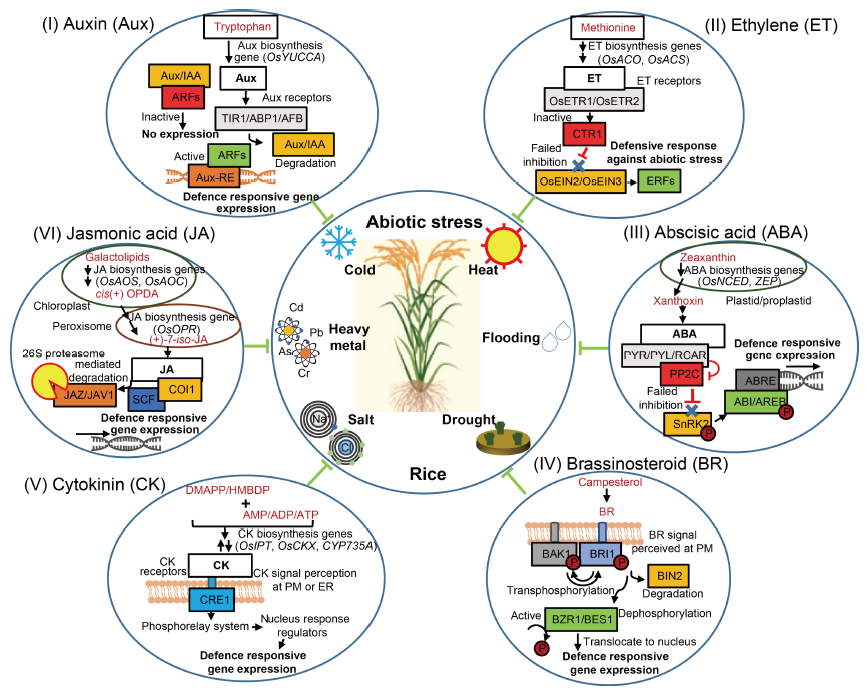
Fig. 1. Plant hormone involvement in amelioration of abiotic stress in rice. Green lines with inhibitory heads signify repression mediated by hormonal action. Red lines with inhibitory heads indicate inhibition/repression. Black lines with arrowheads indicate activation/course of signalling. Blue cross indicates failure of inhibition. Aux/IAA, Auxin/indole-3-acetic acid; ARF, Auxin response factor; ABP1, Auxin-binding protein 1; AFB, Auxin signalling F-box protein; Aux-RE, Auxin response DNA element; ACS, 1-aminocyclopropane-1-carboxylic acid (AC) synthase; ACO, AC oxidase; AOS, Allene oxide synthase; AOC, Allene oxide cyclase; ADP, Adenosine diphosphate; AMP, Adenosine monophosphate; ATP, Adenosine triphosphate; AREB, bZIP transcription factors; As, Arsenic; ABRE, Abscisic acid (ABA) response element; ABI, ABA insensitive; BZR1, Brassinazole resistant 1; BRI1, Brassinosteroid insensitive 1; BAK1, BRI1-associated receptor kinase 1; BES1, BRI1-EMS-suppressor 1; CTR1, Constitutive triple response 1; CYP, Cytochrome P450 monooxygenase; CKX, Cytokinin oxidase/dehydrogenase; Cr, Chromium; CRE1, Cyclic AMP response element 1; Cd, Cadmium; DMAPP, Dimethylallyl pyrophosphate; ER, Endoplasmic reticulum; ETR, Ethylene receptor; EIN, Ethylene-insensitive; ERF, Ethylene response factor; HMBPP, Hydroxymethylbutenyl pyrophosphate; IPT, Isopentenyl transferase; JAZ, Jasmonate-ZIM domain proteins; JAV1, Jasmonate-associated VQ domain protein 1; NCED, 9-cis-epoxycarotenoid dioxygenase; OPDA, 12-oxophytodienoic acid; OPR, OPDA reductase; Pb, Lead; PM, Plasma membrane; PYR/PYL/RCAR, ABA receptors; PP2C, 2C-type protein phosphatase; SnRK2, SNF1-related kinase 2; SCF-COI1, Ubiquitin-ligase complex; TIR1, Transport inhibitor response 1; YUCCA, A flavin monooxygenase gene; ZEP, Zeaxanthin epoxidase.
| Plant hormone | Transgenic line/ gene-targeted | Abiotic stress | Response of plant | Reference |
| Auxin | OsIAA6 (Aux/IAA gene) | Drought | Upon over-expression, transgenic lines show reduced leaf rolling, improved recovery upon rehydration, better chlorophyll fluorescence, and lower transcript levels of DIP1 (molecular dehydration marker). Knock-down mutants exhibit irregular tiller outgrowth | Jung et al, |
| OsPIN3t (auxin efflux carrier gene) | Drought | Over-expression seedlings show improved shoot and root growth, better tillering, greater seed setting and adventitious root formation; improve the expression levels of drought-responsive genes (OsDREB2A and OsAP37); result in improved recovery upon rehydration. RNAi knock-down seedlings exhibit stunted growth and lower seed setting, tillering and yield per plant | Zhang et al, | |
| OsMYB-R1 (TF) | Cr6+ and drought | Over-expression lines show greater auxin accumulation and enhanced lateral root formation, leading to enhanced enzymatic antioxidant defence (CAT, APX, SOD and GPX); show improved revival efficiency from drought stress conditions. Loss-of-function mutants exhibit greater stress sensitivity showing severe wilting in drought and ineffective recovery patterns. Knock-down lines experience greater membrane damage and electrolyte leakage | Tiwari et al, | |
| Auxin and ABA | OsGH3-2 | Cold | Over-expression lines show delayed wilting, improve recovery rates after removal of stress conditions, lower membrane damage, and better oxidative stress tolerance | Du et al, |
| OsMADS25 | Salt | Over-expression lines show greater primary root length, lateral root density, and root cell elongation; show improved chlorophyll content as well as lower lipid peroxidation, membrane damage, and ROS accumulation in roots, shoots, leaves and bracts; exhibit significantly reduced water loss upon ABA application; result in better enzymatic antioxidant (CAT, APX, GPX and GR) activities in roots exposed to H2O2 as well as improved expression of ROS scavenger genes (OsCATB and OsGST4); show enhanced transcript levels of ABA-dependent stress-responsive genes (OsZIP23, OsZIP46, OsTRAB1, OsLEA3, OsABI5, OsP5CS1 and OsP5CR); result in enhanced transcript levels of auxin biosynthesis and signalling genes (OsYUC4, OsARF1 and OsARF16). Knock-down lines result in lowering of primary root elongation and root cell elongation; show enhanced membrane damage, chlorosis and leaf wilting as well as hampered ROS homeostasis in roots, shoots, leaves and bracts; hamper the root enzymatic antioxidant activity after H2O2 exposure and expression of ROS scavenger genes | Xu N et al, | |
| ABA | JERF1 (an ERF) | Drought | Over-expression lines lower wilting and yellowing while improved leaf production, fresh weight, tillering and recovery upon rehydration as well as greater root length; result in improved proline content and leaf RWC while lower water loss; exhibit greater expression of stress-responsive genes (OsP5CS, OsSPDS2, OsCDPK13 and OsLTP1); enhance expression of ABA synthesis genes (OsABA2 and Os03g0810800), and up-regulate ABA synthesis causing improved ABA content | Zhang et al, |
| SAPK6 and OsbZIP46 (Co-over- expressed) | Heat, cold and drought | Co-over-expression lines result in improved biomass, grain number, spikelet number, average panicle number, and yield per plant under drought conditions; show lesser water loss rate, drought symptoms such as leaf rolling, improved survival under heat and cold stress conditions | Chang et al, | |
| CK | OsCKX2 | Salt | Knock-down lines result in enhanced endogenous CK content; show improved growth, photosynthetic pigment content, net photosynthetic rate, stomatal conductance, intercellular CO2 concentration, RWC and PSII efficiency while reduce electrolyte leakage; show greater spikelet number per panicle, panicle branching, grain number per panicle, grain weight and overall panicle number | Joshi et al, |
| ET | OsARD1 | Submergence, drought and salt | Over-expression lines result in greater endogenous ethylene content, coleoptile elongation upon germination under submergence accompanying anoxia; exhibit longer shoot under submergence and greater induction of Ahd1 and Sub1C genes; show delayed symptoms of drought and improved green color retention lines; expose to salt stress show longer shoot length, improved maintenance of leaf structure, green color retention, and delayed withering; show greater trichome, and stomatal density as well as improved water holding capacity; exhibit enhanced expression of ethylene biosynthesis genes (OsACS2 and OsACS4), ERFs (AP37 and AP59), TFs (OsbZIP23) and stress-responsive genes (OsNCED4, OsNCED5, OsPP108 and OsSalT) | Liang et al, |
| ABA, Abcisic acid; APX, Ascorbate peroxidase; Aux/IAA, Auxin/indole-3-acetic acid; CAT, Catalase; Cr, Chromium; CK, Cytokinin; ET, Ethylene; ERF, Ethylene response factor; GR, Glutathione reductase; GPX, Glutathione peroxidase; GSH, Glutathione; K, Potassium; POX, Peroxidase; ROS, Reactive oxygen species; RNAi, RNA interference; RWC, Relative water content; SOD, Superoxide dismutase; TF, Transcription factor. | ||||
Table 2. Representative studies of rice genes targeted at plant hormone for abiotic stress mitigation.
| Plant hormone | Transgenic line/ gene-targeted | Abiotic stress | Response of plant | Reference |
| Auxin | OsIAA6 (Aux/IAA gene) | Drought | Upon over-expression, transgenic lines show reduced leaf rolling, improved recovery upon rehydration, better chlorophyll fluorescence, and lower transcript levels of DIP1 (molecular dehydration marker). Knock-down mutants exhibit irregular tiller outgrowth | Jung et al, |
| OsPIN3t (auxin efflux carrier gene) | Drought | Over-expression seedlings show improved shoot and root growth, better tillering, greater seed setting and adventitious root formation; improve the expression levels of drought-responsive genes (OsDREB2A and OsAP37); result in improved recovery upon rehydration. RNAi knock-down seedlings exhibit stunted growth and lower seed setting, tillering and yield per plant | Zhang et al, | |
| OsMYB-R1 (TF) | Cr6+ and drought | Over-expression lines show greater auxin accumulation and enhanced lateral root formation, leading to enhanced enzymatic antioxidant defence (CAT, APX, SOD and GPX); show improved revival efficiency from drought stress conditions. Loss-of-function mutants exhibit greater stress sensitivity showing severe wilting in drought and ineffective recovery patterns. Knock-down lines experience greater membrane damage and electrolyte leakage | Tiwari et al, | |
| Auxin and ABA | OsGH3-2 | Cold | Over-expression lines show delayed wilting, improve recovery rates after removal of stress conditions, lower membrane damage, and better oxidative stress tolerance | Du et al, |
| OsMADS25 | Salt | Over-expression lines show greater primary root length, lateral root density, and root cell elongation; show improved chlorophyll content as well as lower lipid peroxidation, membrane damage, and ROS accumulation in roots, shoots, leaves and bracts; exhibit significantly reduced water loss upon ABA application; result in better enzymatic antioxidant (CAT, APX, GPX and GR) activities in roots exposed to H2O2 as well as improved expression of ROS scavenger genes (OsCATB and OsGST4); show enhanced transcript levels of ABA-dependent stress-responsive genes (OsZIP23, OsZIP46, OsTRAB1, OsLEA3, OsABI5, OsP5CS1 and OsP5CR); result in enhanced transcript levels of auxin biosynthesis and signalling genes (OsYUC4, OsARF1 and OsARF16). Knock-down lines result in lowering of primary root elongation and root cell elongation; show enhanced membrane damage, chlorosis and leaf wilting as well as hampered ROS homeostasis in roots, shoots, leaves and bracts; hamper the root enzymatic antioxidant activity after H2O2 exposure and expression of ROS scavenger genes | Xu N et al, | |
| ABA | JERF1 (an ERF) | Drought | Over-expression lines lower wilting and yellowing while improved leaf production, fresh weight, tillering and recovery upon rehydration as well as greater root length; result in improved proline content and leaf RWC while lower water loss; exhibit greater expression of stress-responsive genes (OsP5CS, OsSPDS2, OsCDPK13 and OsLTP1); enhance expression of ABA synthesis genes (OsABA2 and Os03g0810800), and up-regulate ABA synthesis causing improved ABA content | Zhang et al, |
| SAPK6 and OsbZIP46 (Co-over- expressed) | Heat, cold and drought | Co-over-expression lines result in improved biomass, grain number, spikelet number, average panicle number, and yield per plant under drought conditions; show lesser water loss rate, drought symptoms such as leaf rolling, improved survival under heat and cold stress conditions | Chang et al, | |
| CK | OsCKX2 | Salt | Knock-down lines result in enhanced endogenous CK content; show improved growth, photosynthetic pigment content, net photosynthetic rate, stomatal conductance, intercellular CO2 concentration, RWC and PSII efficiency while reduce electrolyte leakage; show greater spikelet number per panicle, panicle branching, grain number per panicle, grain weight and overall panicle number | Joshi et al, |
| ET | OsARD1 | Submergence, drought and salt | Over-expression lines result in greater endogenous ethylene content, coleoptile elongation upon germination under submergence accompanying anoxia; exhibit longer shoot under submergence and greater induction of Ahd1 and Sub1C genes; show delayed symptoms of drought and improved green color retention lines; expose to salt stress show longer shoot length, improved maintenance of leaf structure, green color retention, and delayed withering; show greater trichome, and stomatal density as well as improved water holding capacity; exhibit enhanced expression of ethylene biosynthesis genes (OsACS2 and OsACS4), ERFs (AP37 and AP59), TFs (OsbZIP23) and stress-responsive genes (OsNCED4, OsNCED5, OsPP108 and OsSalT) | Liang et al, |
| ABA, Abcisic acid; APX, Ascorbate peroxidase; Aux/IAA, Auxin/indole-3-acetic acid; CAT, Catalase; Cr, Chromium; CK, Cytokinin; ET, Ethylene; ERF, Ethylene response factor; GR, Glutathione reductase; GPX, Glutathione peroxidase; GSH, Glutathione; K, Potassium; POX, Peroxidase; ROS, Reactive oxygen species; RNAi, RNA interference; RWC, Relative water content; SOD, Superoxide dismutase; TF, Transcription factor. | ||||
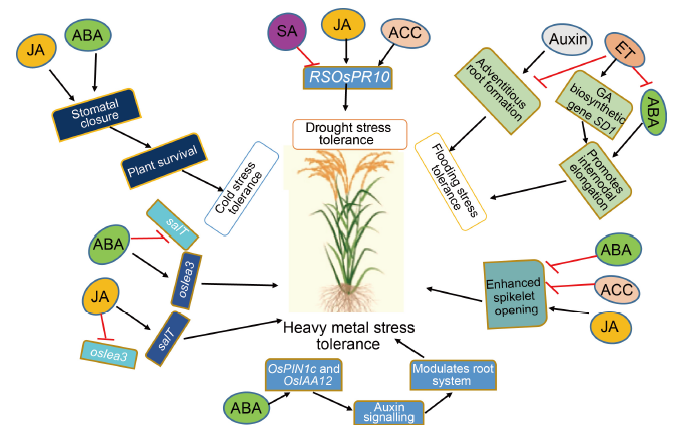
Fig. 2. Plant hormones modulate signaling mechanism in plant system. Red lines with inhibitory heads indicate inhibition/repression; Black lines with arrowheads indicate activation/course of signalling. ABA, Abscisic acid; ET, Ethylene; ACC, Aminocyclopropane-1-carboxylic acid; JA, Jasmonic acid; GA, Gibberellic; SA, Salicylic acid.
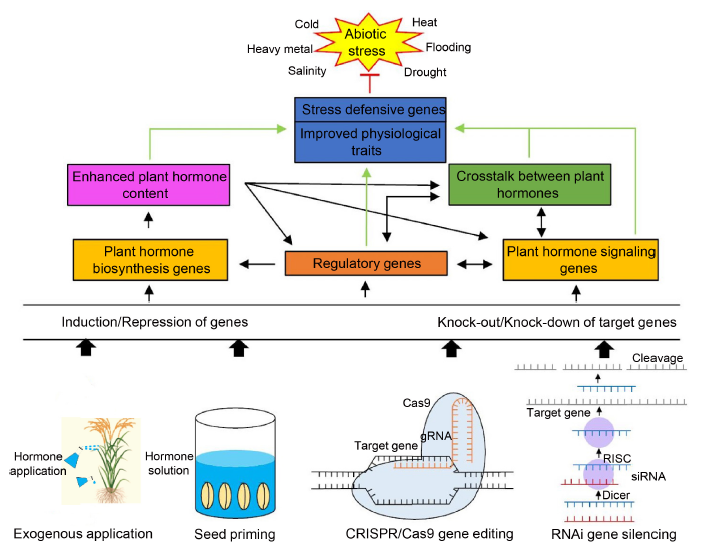
Fig. 3. Approaches for improving abiotic stress tolerance in rice by targeted regulation of plant hormone genes. gRNA, guide RNA; RISC, RNA-induced silencing complex; RNAi, RNA interference; siRNA, Small interfering RNA.
| [1] | Abiri R, Shaharuddin N A, Maziah M, Yusof Z N B, Atabaki N, Sahebi M, Valdiani A, Kalhori N, Azizi P, Hanafi M M. 2017. Role of ethylene and the APETALA 2/ethylene response factor superfamily in rice under various abiotic and biotic stress conditions. Environ Exp Bot, 134: 33-44. |
| [2] | Agrawal G K, Rakwal R, Iwahashi H. 2002. Isolation of novel rice (Oryza sativa L.) multiple stress responsive MAP kinase gene, OsMSRMK2, whose mRNA accumulates rapidly in response to environmental cues. Biochem Biophys Res Commun, 294(5): 1009-1016. |
| [3] | Agrawal G K, Tamogami S, Iwahashi H, Agrawal V P, Rakwal R. 2003. Transient regulation of jasmonic acid-inducible rice MAP kinase gene (OsBWMK1) by diverse biotic and abiotic stresses. Plant Physiol Biochem, 41(4): 355-361. |
| [4] | Ahammed G J, Li X, Zhou J, Zhou Y H, Yu J Q. 2016. Role of hormones in plant adaptation to heat stress. In: Ahammed G J, Yu J Q. Plant Hormones under Challenging Environment Factors. Dordrecht, the Netherlands: Springer: 1-21. |
| [5] | Ahmed T, Noman M, Rizwan M, Ali S, Shahid M S, Li B. 2021. Recent progress on the heavy metals ameliorating potential of engineered nanomaterials in rice paddy: A comprehensive outlook on global food safety with nanotoxicitiy issues. Crit Rev Food Sci Nutr, S23: 1-15. |
| [6] | Akasha A, Ashraf M, Shereen A, Mahboob W, Faisal S. 2019. Heat tolerance screening studies and evaluating salicylic acid efficacy against high temperature in rice (Oryza sativa L.) genotypes. J Plant Biochem Physiol, 7: 235. |
| [7] | Ali L G, Nulit R, Ibrahim M H, Yien C Y S. 2021. Efficacy of KNO3, SiO2 and SA priming for improving emergence, seedling growth and antioxidant enzymes of rice (Oryza sativa), under drought. Sci Rep, 11(1): 3864. |
| [8] | Azuma T, Hirano T, Deki Y, Uchida N, Yasuda T, Yamaguchi T. 1995. Involvement of the decrease in levels of abscisic acid in the internodal elongation of submerged floating rice. J Plant Physiol, 146(3): 323-328. |
| [9] | Basit F, Chen M, Ahmed T, Shahid M, Noman M, Liu J X, An J Y, Hashem A, Fahad Al-Arjani A B, Alqarawi A A, Alsayed M F S, Fathi Abd Allah E, Hu J, Guan Y J. 2021. Seed priming with brassinosteroids alleviates chromium stress in rice cultivars via improving ROS metabolism and antioxidant defense response at biochemical and molecular levels. Antioxidants, 10(7): 1089. |
| [10] | Basit F, Liu J X, An J Y, Chen M, He C, Zhu X B, Li Z, Hu J, Guan Y J. 2022. Seed priming with brassinosteroids alleviates aluminum toxicity in rice via improving antioxidant defense system and suppressing aluminum uptake. Environ Sci Pollut Res, 29: 10183-10197. |
| [11] |
Bücker-Neto L, Paiva A L S, Machado R D, Arenhart R A, Margis- Pinheiro M. 2017. Interactions between plant hormones and heavy metals responses. Genet Mol Biol, 40: 373-386.
PMID |
| [12] | Cai S L, Jiang G B, Ye N H, Chu Z Z, Xu X Z, Zhang J H, Zhu G H. 2015. A key ABA catabolic gene, OsABA8ox3, is involved in drought stress resistance in rice. PLoS One, 10(2): e0116646. |
| [13] |
Chang Y, Nguyen B H, Xie Y J, Xiao B Z, Tang N, Zhu W L, Mou T M, Xiong L Z. 2017. Co-overexpression of the constitutively active form of OsbZIP46 and ABA-activated protein kinase SAPK6 improves drought and temperature stress resistance in rice. Front Plant Sci, 8: 1102.
PMID |
| [14] | Chen C W, Yang Y W, Lur H S, Tsai Y G, Chang M C. 2006. A novel function of abscisic acid in the regulation of rice (Oryza sativa L.) root growth and development. Plant Cell Physiol, 47(1): 1-13. |
| [15] | Chen D, Gunawardena T A, Naidu B P, Fukai S, Basnayake J. 2005. Seed treatment with gibberellic acid and glycinebetaine improves seedling emergence and seedling vigour of rice under low temperature. Seed Sci Technol, 33(2): 471-479. |
| [16] | Chen J, Zhu C, Li L P, Sun Z Y, Pan X B. 2007. Effects of exogenous salicylic acid on growth and H2O2-metabolizing enzymes in rice seedlings under lead stress. J Environ Sci, 19(1): 44-49. |
| [17] | Chen J, Fei K Q, Zhang W Y, Wang Z Q, Zhang J H, Yang J C. 2021. Brassinosteroids mediate the effect of high temperature during anthesis on the pistil activity of photo-thermosensitive genetic male-sterile rice lines. Crop J, 9(1): 109-119. |
| [18] | Chen L, Wang Q, Tang M Y, Zhang X L, Pan Y H, Yang X H, Gao G Q, Lv R H, Tao W, Jiang L G, Liang T F. 2021. QTL mapping and identification of candidate genes for heat tolerance at the flowering stage in rice. Front Genet, 11: 621871. |
| [19] | Chunthaburee S, Sanitchon J, Pattanagul W, Theerakulpisut P. 2014. Alleviation of salt stress in seedlings of black glutinous rice by seed priming with spermidine and gibberellic acid. Not Bot Horti Agrobo Cluj-Na, 42(2): 405-413. |
| [20] |
de Wit M, Lorrain S, Fankhauser C. 2014. Auxin-mediated plant architectural changes in response to shade and high temperature. Physiol Plant, 151(1): 13-24.
PMID |
| [21] | Dong N N, Yin W C, Liu D P, Zhang X X, Yu Z K, Huang W, Liu J H, Yang Y Z, Meng W J, Niu M, Tong H N. 2020. Regulation of brassinosteroid signalling and salt resistance by SERK2 and potential utilization for crop improvement in rice. Front Plant Sci, 11: 621859. |
| [22] |
Du H, Liu H B, Xiong L Z. 2013. Endogenous auxin and jasmonic acid levels are differentially modulated by abiotic stresses in rice. Front Plant Sci, 4: 397.
PMID |
| [23] | Du H, Wu N, Cui F, You L, Li X H, Xiong L Z. 2014. A homolog of ETHYLENE OVERPRODUCER, OsETOL1, differentially modulates drought and submergence tolerance in rice. Plant J, 78(5): 834-849. |
| [24] | Eyidogan F, Oz M T, Yucel M, Oktem H A. 2012. Signal transduction of phytohormones under abiotic stresses. In: Khan N A, Nazar R, Iqbal N, Anjum N A. Phytohormones and Abiotic Stress Tolerance in Plants. Berlin, Germany: Springer: 1-48. |
| [25] | FAO(Food and Agricultural Organisation of the United Nations). 2022. World: Rice, Paddy Production Quantity[2021-12-24]. https://knoema.com/atlas/World/topics/Agriculture/Crops-Production-Quantity-tonnes/Rice-paddy-production. |
| [26] | Farooq H, Asghar H N, Khan M Y, Saleem M, Zahir Z A. 2015. Auxin-mediated growth of rice in cadmium-contaminated soil. Turk J Agric For, 39: 272-276. |
| [27] | Farooq M, Basra S M A, Wahid A, Ahmad N, Saleem B A. 2009a. Improving the drought tolerance in rice (Oryza sativa L.) by exogenous application of salicylic acid. J Agron Crop Sci, 195(4): 237-246. |
| [28] | Farooq M, Wahid A, Basra S M A,Islam-ud-Din. 2009b. Improving water relations and gas exchange with brassinosteroids in rice under drought stress. J Agron Crop Sci, 195(4): 262-269. |
| [29] |
Feng B H, Zhang C X, Chen T T, Zhang X F, Tao L X, Fu G F. 2018. Salicylic acid reverses pollen abortion of rice caused by heat stress. BMC Plant Biol, 18(1): 245.
PMID |
| [30] |
Franklin K A, Lee S H, Patel D, Kumar S V, Spartz A K, Gu C, Ye S Q, Yu P, Breen G, Cohen J D, Wigge P A, Gray W M. 2011. Phytochrome-interacting factor 4 (PIF4) regulates auxin biosynthesis at high temperature. Proc Natl Acad Sci USA, 108: 20231-20235.
PMID |
| [31] | Fujii S, Saka H. 2001. The promotive effect of brassinolide on Lamina joint-cell elongation, germination and seedling growth under low-temperature stress in rice (Oryza sativa L.). Plant Prod Sci, 4(3): 210-214. |
| [32] | Fukao T, Bailey-Serres J. 2008. Submergence tolerance conferred by Sub1A is mediated by SLR1 and SLRL1 restriction of gibberellin responses in rice. Proc Natl Acad Sci USA, 105: 16814-16819. |
| [33] |
Garg R, Tyagi A K, Jain M. 2012. Microarray analysis reveals overlapping and specific transcriptional responses to different plant hormones in rice. Plant Signal Behav, 7(8): 951-956.
PMID |
| [34] | Gautam H, Fatma M, Sehar Z, Iqbal N, Albaqami M, Khan N A. 2022. Exogenously-sourced ethylene positively modulates photosynthesis, carbohydrate metabolism, and antioxidant defense to enhance heat tolerance in rice. Int J Mol Sci, 23(3): 1031. |
| [35] | Ghate T, Soneji K, Barvkar V, Ramakrishnan P, Prusty D, Islam S R, Manna S K, Srivastava A K. 2022. Thiourea mediated ROS- metabolites reprogramming restores root system architecture under arsenic stress in rice. J Hazard Mater, 435: 129020. |
| [36] |
Guo B, Liang Y C, Zhu Y G, Zhao F J. 2007. Role of salicylic acid in alleviating oxidative damage in rice roots (Oryza sativa) subjected to cadmium stress. Environ Pollut, 147(3): 743-749.
PMID |
| [37] | Guo B, Liang Y C, Zhu Y G. 2009. Does salicylic acid regulate antioxidant defense system, cell death, cadmium uptake and partitioning to acquire cadmium tolerance in rice? J Plant Physiol, 166(1): 20-31. |
| [38] | Guo J C, Duan R J, Hu X W, Li K M, Fu S P. 2010. Isopentenyl transferase gene (ipt) downstream transcriptionally fused with gene expression improves the growth of transgenic plants. Transgenic Res, 19(2): 197-209. |
| [39] | Guo Y, Huang R, Duan L, Wang J. 2017. The APETALA2/ethylene- responsive factor transcription factor OsDERF2 negatively modulates drought stress in rice by repressing abscisic acid responsive genes. J Agric Sci, 155(6): 966-977. |
| [40] |
Hashimoto M, Kisseleva L, Sawa S, Furukawa T, Komatsu S, Koshiba T. 2004. A novel rice PR10 protein, RSOsPR10, specifically induced in roots by biotic and abiotic stresses, possibly via the jasmonic acid signalling pathway. Plant Cell Physiol, 45(5): 550-559.
PMID |
| [41] |
Hoang X L T, Nhi D N H, Thu N B A, Thao N P, Tran L S P. 2017. Transcription factors and their roles in signal transduction in plants under abiotic stresses. Curr Genomics, 18(6): 483-497.
PMID |
| [42] |
Hoffmann-Benning S, Kende H. 1992. On the role of abscisic acid and gibberellin in the regulation of growth in rice. Plant Physiol, 99(3): 1156-1161.
PMID |
| [43] | Horváth E, Szalai G, Janda T. 2007. Induction of abiotic stress tolerance by salicylic acid signaling. J Plant Growth Regul, 26(3): 290-300. |
| [44] | Hosain M T, Poly K, Rahman M M, Munshi M, Rahman M S. 2020. Drought stress response of rice yield (Oryza sativa L.) and role of exogenous salicylic acid. Int J Biosci, 6: 222-230. |
| [45] | Hsu Y T, Kao C H. 2003. Role of abscisic acid in cadmium tolerance of rice (Oryza sativa L.) seedlings. Plant Cell Environ, 26(6): 867-874. |
| [46] | Hu Y, Wu Q Y, Peng Z, Sprague S A, Wang W, Park J, Akhunov E, Jagadish K S V, Nakata P A, Cheng N H, Hirschi K D, White F F, Park S. 2017. Silencing of OsGRXS17 in rice improves drought stress tolerance by modulating ROS accumulation and stomatal closure. Sci Rep, 7: 15950. |
| [47] | Huang T L, Nguyen Q T T, Fu S F, Lin C Y, Chen Y C, Huang H J. 2012. Transcriptomic changes and signalling pathways induced by arsenic stress in rice roots. Plant Mol Biol, 80(6): 587-608. |
| [48] |
Hussain S, Khan F, Hussain H A, Nie L X. 2016. Physiological and biochemical mechanisms of seed priming-induced chilling tolerance in rice cultivars. Front Plant Sci, 7: 116.
PMID |
| [49] | Hussain S, Khaliq A, Ali B, Hussain H A, Qadir T, Hussain S,. 2019. Temperature extremes:Impact on rice growth and development. In: Hasanuzzaman M, Hakeem K, Nahar K, Alharby H. Plant Abiotic Stress Tolerance. Cham, Cambodia: Springer: 153-171. |
| [50] | Jadamba C, Kang K, Paek N C, Lee S I, Yoo S C. 2020. Overexpression of rice Expansin7 (Osexpa7) confers enhanced tolerance to salt stress in rice. Int J Mol Sci, 21(2): 454. |
| [51] | Jagadish S V K, Craufurd P Q, Wheeler T R. 2008. Phenotyping parents of mapping populations of rice for heat tolerance during anthesis. Crop Sci, 48(3): 1140-1146. |
| [52] | Jing C, Cheng Z H U, Li L P, Sun Z Y, Pan X B. 2007. Effects of exogenous salicylic acid on growth and H2O2-metabolizing enzymes in rice seedlings under lead stress. J Environ Sci, 19(1): 44-49. |
| [53] | Jini D, Joseph B. 2017. Physiological mechanism of salicylic acid for alleviation of salt stress in rice. Rice Sci, 24(2): 97-108. |
| [54] | Joshi R, Sahoo K K, Tripathi A K, Kumar R, Gupta B K, Pareek A, Singla-Pareek S L. 2018. Knockdown of an inflorescence meristem-specific cytokinin oxidase-OsCKX2 in rice reduces yield penalty under salinity stress condition. Plant Cell Environ, 41(5): 936-946. |
| [55] |
Jung H, Lee D K, Choi Y D, Kim J K. 2015. OsIAA6, a member of the rice Aux/IAA gene family, is involved in drought tolerance and tiller outgrowth. Plant Sci, 236: 304-312.
PMID |
| [56] | Kang D J, Seo Y J, Lee J D, Ishii R, Kim K U, Shin D H, Park S K, Jang S W, Lee I J. 2005. Jasmonic acid differentially affects growth, ion uptake and abscisic acid concentration in salt-tolerant and salt-sensitive rice cultivars. J Agron Crop Sci, 191(4): 273-282. |
| [57] | Khan M I R, Fatma M, Per T S, Anjum N A, Khan N A. 2015a. Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front Plant Sci, 6: 462. |
| [58] | Khan M I R, Nazir F, Asgher M, Per T S, Khan N A. 2015b. Selenium and sulfur influence ethylene formation and alleviate cadmium-induced oxidative stress by improving proline and glutathione production in wheat. J Plant Physiol, 173: 9-18. |
| [59] | Khan M I R, Trivellini A, Chhillar H, Chopra P, Ferrante A, Khan N A, Ismail A M. 2020. The significance and functions of ethylene in flooding stress tolerance in plants. Environ Exp Bot, 179: 104188. |
| [60] | Khan M I R, Palakolanu S R, Chopra P, Rajurkar A B, Gupta R, Iqbal N, Maheshwari C. 2021. Improving drought tolerance in rice: Ensuring food security through multi-dimensional approaches. Physiol Plant, 172(2): 645-668. |
| [61] | Khan M I R, Reddy P S, Gupta R. 2022. Advancements in Developing Abiotic Stress-Resilient Plants: Basic Mechanisms to Trait Improvements. Boca Raton, USA: CRC Press: 394. |
| [62] | Khan M S, Akther T, Mubarak Ali D, Hemalatha S. 2019. An investigation on the role of salicylic acid alleviate the saline stress in rice crop (Oryza sativa (L)). Biocatal Agric Biotechnol, 18: 101027. |
| [63] |
Kim E H, Kim Y S, Park S H, Koo Y J, Choi Y D, Chung Y Y, Lee I J, Kim J K. 2009. Methyl jasmonate reduces grain yield by mediating stress signals to alter spikelet development in rice. Plant Physiol, 149(4): 1751-1760.
PMID |
| [64] | Koh S, Lee S C, Kim M K, Koh J H, Lee S, An G, Choe S, Kim S R. 2007. T-DNA tagged knockout mutation of rice OsGSK1, an orthologue of Arabidopsis BIN2 with enhanced tolerance to various abiotic stresses. Plant Mol Biol, 65(4): 453-466. |
| [65] |
Kudo M, Kidokoro S, Yoshida T, Mizoi J, Todaka D, Fernie A R, Shinozaki K, Yamaguchi-Shinozaki K. 2017. Double overexpression of DREB and PIF transcription factors improves drought stress tolerance and cell elongation in transgenic plants. Plant Biotechnol J, 15(4): 458-471.
PMID |
| [66] | Kuroha T, Nagai K, Gamuyao R, Wang D R, Furuta T, Nakamori M, Kitaoka T, Adachi K, Minami A, Mori Y, Mashiguchi K, Seto Y, Yamaguchi S, Kojima M, Sakakibara H, Wu J Z, Ebana K, Mitsuda N, Ohme-Takagi M, Yanagisawa S, Yamasaki M, Yokoyama R, Nishitani K, Mochizuki T, Tamiya G, McCouch S R, Ashikari M. 2018. Ethylene-gibberellin signalling underlies adaptation of rice to periodic flooding. Science, 361: 181-186. |
| [67] |
Larkindale J, Knight M R. 2002. Protection against heat stress-induced oxidative damage in Arabidopsis involves calcium, abscisic acid, ethylene, and salicylic acid. Plant Physiol, 128(2): 682-695.
PMID |
| [68] |
Larkindale J, Hall J D, Knight M R, Vierling E. 2005. Heat stress phenotypes of Arabidopsis mutants implicate multiple signalling pathways in the acquisition of thermotolerance. Plant Physiol, 138(2): 882-897.
PMID |
| [69] | Lee T M, Lur H S, Chu C. 1997. Role of abscisic acid in chilling tolerance of rice (Oryza sativa L.) seedlings: II. Modulation of free polyamine levels. Plant Sci, 126(1): 1-10. |
| [70] | Li J J, Yang J, Zhu B H, Xie G S. 2019. Overexpressing OsFBN1 enhances plastoglobule formation, reduces grain-filling percent and jasmonate levels under heat stress in rice. Plant Sci, 285: 230-238. |
| [71] | Li J Z, Li M Q, Han Y C, Sun H Z, Du Y X, Zhao Q Z. 2019. The crucial role of gibberellic acid on germination of drought- resistant upland rice. Biol Plant, 63: 529-535. |
| [72] | Li K, Kamiya T, Fujiwara T. 2015. Differential roles of PIN1 and PIN2 in root meristem maintenance under low-B conditions in Arabidopsis thaliana. Plant Cell Physiol, 56(6): 1205-1214. |
| [73] | Liang S, Xiong W, Yin C, Xie X, Jin Y.J, Zhang S, Yang B, Ye G, Chen S, Luan W J. 2019. Overexpression of OsARD1 improves submergence, drought, and salt tolerances of seedling through the enhancement of ethylene synthesis in rice. Front Plant Sci, 10: 1088. |
| [74] | Lin C Y, Trinh N N, Lin C W, Huang H J. 2013. Transcriptome analysis of phytohormone, transporters and signalling pathways in response to vanadium stress in rice roots. Plant Physiol Biochem, 66: 98-104. |
| [75] | Liu X X, Wang X Y, Yin L N, Deng X P, Wang S W. 2018. Exogenous application of gibberellic acid participates in up-regulation of lipid biosynthesis under salt stress in rice. Theor Exp Plant Physiol, 30(4): 335-345. |
| [76] |
Lorbiecke R, Sauter M. 1999. Adventitious root growth and cell-cycle induction in deepwater rice. Plant Physiol, 119(1): 21-30.
PMID |
| [77] | Mahmud S, Sharmin S, Chowdhury B L D, Hossain M A, Bhuiyan M J H. 2016. Mitigation of salt stress in rice plant at germination stage by using methyl jasmonate. Asian J Med Biol Res, 2(1): 74-81. |
| [78] | Mega R, Meguro-Maoka A, Endo A, Shimosaka E, Murayama S, Nambara E, Seo M, Kanno Y, Abrams S R, Sato Y. 2015. Sustained low abscisic acid levels increase seedling vigor under cold stress in rice (Oryza sativa L.). Sci Rep, 5: 13819. |
| [79] |
Minami A, Yano K, Gamuyao R, Nagai K, Kuroha T, Ayano M, Nakamori M, Koike M, Kondo Y, Niimi Y, Kuwata K, Suzuki T, Higashiyama T, Takebayashi Y, Kojima M, Sakakibara H, Toyoda A, Fujiyama A, Kurata N, Ashikari M, Reuscher S. 2018. Time-course transcriptomics analysis reveals key responses of submerged deepwater rice to flooding. Plant Physiol, 176(4): 3081-3102.
PMID |
| [80] | Misratia K M, Islam M R, Ismail M R, Oad F C, Hanafi M M, Puteh A. 2015. Interactive effects of gibberellic acid (GA3) and salt stress on growth biochemical parameters and ion accumulation of two rice (Oryza sativa L.) varieties differing in salt tolerance. J Food Agric Environ, 13(1): 66-70. |
| [81] | Mohabbati F, Moradi F, Paknejad F, Vazan S, Behneya D H S, Irandoost H P. 2012. Effect of foliar application of auxin, absisic acid and cytokinin hormones on grain yield and yield components of three rice (Oryza sativa L.) genotypes under low temperature stress conditions. Iran J Crop Sci, 14(1): 58-71. |
| [82] |
Moons A, Prinsen E, Bauw G, van Montagu M. 1997. Antagonistic effects of abscisic acid and jasmonates on salt stress-inducible transcripts in rice roots. Plant Cell, 9(12): 2243-2259.
PMID |
| [83] |
Mousavi S R, Niknejad Y, Fallah H, Tari D B. 2020. Methyl jasmonate alleviates arsenic toxicity in rice. Plant Cell Rep, 39(8): 1041-1060.
PMID |
| [84] | Mukamuhirwa A, Persson Hovmalm H, Bolinsson H, Ortiz R, Nyamangyoku O, Johansson E. 2019. Concurrent drought and temperature stress in rice: A possible result of the predicted climate change: Effects on yield attributes, eating characteristics, and health promoting compounds. Int J Environ Res Public Health, 16(6): 1043. |
| [85] | Najeeb S, Ali J, Mahender A, Pang Y L, Zilhas J, Murugaiyan V, Vemireddy L R, Li Z. 2020. Identification of main-effect quantitative trait loci (QTLs) for low-temperature stress tolerance germination- and early seedling vigor-related traits in rice (Oryza sativa L.). Mol Breed, 40(1): 10. |
| [86] | Neang S, de Ocampo M, Egdane J A, Platten J D, Ismail A M, Seki M, Suzuki Y, Skoulding N S, Kano-Nakata M, Yamauchi A, Mitsuya S. 2020. A GWAS approach to find SNPs associated with salt removal in rice leaf sheath. Ann Bot, 126(7): 1193-1202. |
| [87] | Nishiyama R, Watanabe Y, Fujita Y, Le D T, Kojima M, Werner T, Vankova R, Yamaguchi-Shinozaki K, Shinozaki K, Kakimoto T, Sakakibara H, Schmülling T, Tran L S P. 2011. Analysis of cytokinin mutants and regulation of cytokinin metabolic genes reveals important regulatory roles of cytokinins in drought, salt and abscisic acid responses, and abscisic acid biosynthesis. Plant Cell, 23(6): 2169-2183. |
| [88] | Nolan T M, Vukašinović N, Liu D R, Russinova E, Yin Y H. 2020. Brassinosteroids: Multidimensional regulators of plant growth, development, and stress responses. Plant Cell, 32(2): 295-318. |
| [89] |
O’Brien J A, Benková E. 2013. Cytokinin cross-talking during biotic and abiotic stress responses. Front Plant Sci, 4: 451.
PMID |
| [90] | Ogata T, Ishizaki T, Fujita M, Fujita Y. 2020. CRISPR/Cas9- targeted mutagenesis of OsERA1 confers enhanced responses to abscisic acid and drought stress and increased primary root growth under nonstressed conditions in rice. PLoS One, 15(12): e0243376. |
| [91] | Pandey C, Gupta M. 2015. Selenium and auxin mitigates arsenic stress in rice (Oryza sativa L.) by combining the role of stress indicators, modulators and genotoxicity assay. J Hazard Mater, 287: 384-391. |
| [92] |
Pei Z M, Ghassemian M, Kwak C M, McCourt P, Schroeder J I. 1998. Role of farnesyltransferase in ABA regulation of guard cell anion channels and plant water loss. Science, 282: 287-290.
PMID |
| [93] |
Peleg Z, Blumwald E. 2011. Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol, 14(3): 290-295.
PMID |
| [94] | Per T S, Khan M I R, Anjum N A, Masood A, Hussain S J, Khan N A. 2018. Jasmonates in plants under abiotic stresses: Crosstalk with other phytohormones matters. Environ Exp Bot, 145: 104-120. |
| [95] | Pospíšilová J. 2003. Participation of phytohormones in the stomatal regulation of gas exchange during water stress. Biol Plant, 46(4): 491-506. |
| [96] | Rafiq C M, Raza Q, Riaz A, Hanif M, Saeed W, Iqbal S, Awan T H, Ali S S, Sabar M. 2021. Salicylic acid improves rice seed germination under induced drought stress. J Innov Sci, 7(1): 152-160. |
| [97] | Ramachandran M, Arulbalachandran D, Dilipan E, Ramya S. 2021. Comparative analysis of abscisic acid recovery on two varieties of rice (Oryza sativa L.) under drought condition. Biocatal Agric Biotechnol, 33: 102006. |
| [98] |
Ramireddy E, Hosseini S A, Eggert K, Gillandt S, Gnad H, von Wirén N, Schmülling T. 2018. Root engineering in barley: Increasing cytokinin degradation produces a larger root system, mineral enrichment in the shoot and improved drought tolerance. Plant Physiol, 177(3): 1078-1095.
PMID |
| [99] |
Raza A, Charagh S, Zahid Z, Mubarik M S, Javed R, Siddiqui M H, Hasanuzzaman M. 2021. Jasmonic acid: A key frontier in conferring abiotic stress tolerance in plants. Plant Cell Rep, 40(8): 1513-1541.
PMID |
| [100] | Ronzan M, Piacentini D, Fattorini L, Caboni E, Eiche E, Ziegler J, Hause B, Riemann M, Betti C, Altamura M M, Falasca G. 2019. Auxin-jasmonate crosstalk in Oryza sativa L. root system formation after cadmium and/or arsenic exposure. Environ Exp Bot, 165: 59-69. |
| [101] |
RoyChoudhury A, Roy C, Sengupta D N. 2007. Transgenic tobacco plants overexpressing the heterologous lea gene Rab16A from rice during high salt and water deficit display enhanced tolerance to salinity stress. Plant Cell Rep, 26(10): 1839-1859.
PMID |
| [102] |
RoyChoudhury A, Paul S, Basu S. 2013. Cross-talk between abscisic acid-dependent and abscisic acid-independent pathways during abiotic stress. Plant Cell Rep, 32(7): 985-1006.
PMID |
| [103] | Rucińska-Sobkowiak R. 2016. Water relations in plants subjected to heavy metal stresses. Acta Physiol Plant, 38(11): 1-13. |
| [104] | Saedipour S. 2016. Role of exogenous application of auxin on antioxidant enzyme activities in rice under salt stress. J Plant Physiol Breed, 6(2): 49-61. |
| [105] |
Saini S, Sharma I, Kaur N, Pati P K. 2013. Auxin: A master regulator in plant root development. Plant Cell Rep, 32(6): 741-757.
PMID |
| [106] | Saini S, Kaur N, Marothia D, Singh B, Singh V, Gantet P, Pati P K. 2021. Morphological analysis, protein profiling and expression analysis of auxin homeostasis genes of roots of two contrasting cultivars of rice provide inputs on mechanisms involved in rice adaptation towards salinity stress. Plants, 10(8): 1544. |
| [107] | Sharma I, Ching E, Saini S, Bhardwaj R, Pati P K. 2013. Exogenous application of brassinosteroid offers tolerance to salinity by altering stress responses in rice variety Pusa Basmati-1. Plant Physiol Biochem, 69: 17-26. |
| [108] |
Sharma I, Kaur N, Pati P K. 2017. Brassinosteroids: A promising option in deciphering remedial strategies for abiotic stress tolerance in rice. Front Plant Sci, 8: 2151.
PMID |
| [109] | Sharma L, Dalal M, Verma R K, Kumar S V V, Yadav S K, Pushkar S, Kushwaha S R, Bhowmik A, Chinnusamy V. 2018. Auxin protects spikelet fertility and grain yield under drought and heat stresses in rice. Environ Exp Bot, 150: 9-24. |
| [110] |
Sharma R, Priya P, Jain M. 2013. Modified expression of an auxin-responsive rice CC-type glutaredoxin gene affects multiple abiotic stress responses. Planta, 238(5): 871-884.
PMID |
| [111] | Shashibhushan D, Reddy A, Bhadru D, Pradeep T. 2021. Effect of gibberellic acid (GA3) on the yield attributing traits during a cold period in rice. Int J Environ Clim Change, 11: 34-38. |
| [112] | Shatpathy P, Kar M, Dwibedi S K, Dash A. 2018. Seed priming with salicylic acid improves germination and seedling growth of rice (Oryza sativa L.) under PEG-6000 induced water stress. Int J Curr Microbiol App Sci, 7(10): 907-924. |
| [113] |
Sheteiwy M S, An J Y, Yin M Q, Jia X W, Guan Y J, He F, Hu J. 2019. Cold plasma treatment and exogenous salicylic acid priming enhances salinity tolerance of Oryza sativa seedlings. Protoplasma, 256(1): 79-99.
PMID |
| [114] | Shi B, Ni L, Zhang A Y, Cao J M, Zhang H, Qin T T, Tan M P, Zhang J H, Jiang M Y. 2012. OsDMI3 is a novel component of abscisic acid signalling in the induction of antioxidant defense in leaves of rice. Mol Plant, 5(6): 1359-1374. |
| [115] |
Singh I, Shah K. 2014. Exogenous application of methyl jasmonate lowers the effect of cadmium-induced oxidative injury in rice seedlings. Phytochemistry, 108: 57-66.
PMID |
| [116] | Sohag A A M, Tahjib-Ul-Arif M, Brestic M, Afrin S, Sakil M A, Hossain M T, Hossain M A, Hossain M A,. 2020. Exogenous salicylic acid and hydrogen peroxide attenuate drought stress in rice. Plant Soil Environ, 66: 7-13. |
| [117] |
Steffens B. 2014. The role of ethylene and ROS in salinity, heavy metal, and flooding responses in rice. Front Plant Sci, 5: 685.
PMID |
| [118] | Sun M Z, Shen Y, Yang J K, Cai X X, Li H Y, Zhu Y M, Jia B W, Sun X L. 2020. miR535 negatively regulates cold tolerance in rice. Mol Breed, 40: 14. |
| [119] | Sytar O, Kumari P, Yadav S, Brestic M, Rastogi A. 2019. Phytohormone priming: Regulator for heavy metal stress in plants. J Plant Growth Regul, 38(2): 739-752. |
| [120] | Takeuchi K, Hasegawa H, Gyohda A, Komatsu S, Okamoto T, Okada K, Terakawa T, Koshiba T. 2016. Overexpression of RSOsPR10, a root-specific rice PR10 gene, confers tolerance against drought stress in rice and drought and salt stresses in bentgrass. Plant Cell Tiss Organ Cult, 127(1): 35-46. |
| [121] | Tamaoki D, Seo S, Yamada S, Kano A, Miyamoto A, Shishido H, Miyoshi S, Taniguchi S, Akimitsu K, Gomi K. 2013. Jasmonic acid and salicylic acid activate a common defense system in rice. Plant Signal Behav, 8(6): e24260. |
| [122] | Teng K Q, Li J Z, Liu L, Han Y C, Du Y X, Zhang J, Sun H Z, Zhao Q Z. 2014. Exogenous ABA induces drought tolerance in upland rice: The role of chloroplast and ABA biosynthesis-related gene expression on photosystem II during PEG stress. Acta Physiol Plant, 36(8): 2219-2227. |
| [123] | Thao N P, Khan M I R, Thu N B A, Hoang X L T, Asgher M, Khan N A, Tran L S P. 2015. Role of ethylene and its cross talk with other signalling molecules in plant responses to heavy metal stress. Plant Physiol, 169(1): 73-84. |
| [124] | Thurtle-Schmidt D M, Lo T W. 2018. Molecular biology at the cutting edge: A review on CRISPR/CAS9 gene editing for undergraduates. Biochem Mol Biol Educ, 46(2): 195-205. |
| [125] | Thussagunpanit J, Jutamanee K, Kaveeta L, Chai-Arree W, Pankean P, Suksamrarn A. 2013. Effects of a brassinosteroid and an ecdysone analogue on pollen germination of rice under heat stress. J Pestic Sci, 38(3): 105-111. |
| [126] | Thussagunpanit J, Jutamanee K, Kaveeta L, Chai-arree W, Pankean P, Homvisasevongsa S, Suksamrarn A. 2015. Comparative effects of brassinosteroid and brassinosteroid mimic on improving photosynthesis, lipid peroxidation, and rice seed set under heat stress. J Plant Growth Regul, 34(2): 320-331. |
| [127] | Tian Y, Zhang H, Pan X, Chen X, Zhang Z, Lu X, Huang R. 2011. Overexpression of ethylene response factor TERF2 confers cold tolerance in rice seedlings. Transgen Res, 20(4): 857-866. |
| [128] | Tiwari P, Indoliya Y, Chauhan A S, Singh P, Singh P K, Singh P C, Srivastava S, Pande V, Chakrabarty D. 2020. Auxin-salicylic acid cross-talk ameliorates OsMYB-R1 mediated defense towards heavy metal, drought and fungal stress. J Hazard Mater, 399: 122811. |
| [129] | Trinh N N, Huang T L, Chi W C, Fu S F, Chen C C, Huang H J. 2014. Chromium stress response effect on signal transduction and expression of signalling genes in rice. Physiol Plant, 150(2): 205-224. |
| [130] | Umesh D K, Pal M. 2018. Differential role of jasmonic acid under drought and heat stress in rice (Oryza sativa). J Pharmacogn Phytochem, S1: 2626-2631. |
| [131] | Usman B, Nawaz G, Zhao N, Liao S Y, Liu Y G, Li R B. 2020. Precise editing of the OsPYL9 gene by RNA-guided Cas9 nuclease confers enhanced drought tolerance and grain yield in rice (Oryza sativa L.) by regulating circadian rhythm and abiotic stress responsive proteins. Int J Mol Sci, 21(21): 7854. |
| [132] | Verma G, Srivastava D, Narayan S, Shirke P A, Chakrabarty D. 2020. Exogenous application of methyl jasmonate alleviates arsenic toxicity by modulating its uptake and translocation in rice (Oryza sativa L.). Ecotoxicol Environ Saf, 201: 110735. |
| [133] | Wang B, Zhong Z H, Zhang H H, Wang X, Liu B L, Yang L J, Han X Y, Yu D S, Zheng X L, Wang C G, Song W Q, Chen C B, Zhang Y. 2019. Targeted mutagenesis of NAC transcription factor gene OsNAC041 leading to salt sensitivity in rice. Rice Sci, 26(2): 98-108. |
| [134] | Wang B, Zhong Z H, Wang X, Han X Y, Yu D S, Wang C G, Song W Q, Zheng X L, Chen C B, Zhang Y. 2020. Knockout of the OsNAC006 transcription factor causes drought and heat sensitivity in rice. Int J Mol Sci, 21(7): 2288. |
| [135] | Wang J, Islam F, Yang C, Long M J, Li L, Hu L Y, Gill R A, Wan G L, Zhou W J. 2019. Use of phytohormones in improving abiotic stress tolerance in rice. In: Hasanuzzaman M, Fujita M, Nahar K, Biswas J K. Advances in Rice Research for Abiotic Stress Tolerance. Amsterdam: Woodhead Publishing: 651-675. |
| [136] | Wani S H, Kumar V, Shriram V, Sah S K. 2016. Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop J, 4(3): 162-176. |
| [137] | Wen F P, Zhang Z H, Bai T, Xu Q, Pan Y H. 2010. Proteomics reveals the effects of gibberellic acid (GA3) on salt-stressed rice (Oryza sativa L.) shoots. Plant Sci, 178(2): 170-175. |
| [138] | Wu C, Tang S, Li G H, Wang S H, Fahad S, Ding Y F. 2019. Roles of phytohormone changes in the grain yield of rice plants exposed to heat: A review. PeerJ, 7: e7792. |
| [139] | Wu X Y, Cobbina S J, Mao G H, Xu H, Zhang Z, Yang L Q. 2016. A review of toxicity and mechanisms of individual and mixtures of heavy metals in the environment. Environ Sci Pollut Res Int, 23(9): 8244-8259. |
| [140] | Wu Y S, Yang C Y. 2019. Ethylene-mediated signalling confers thermotolerance and regulates transcript levels of heat shock factors in rice seedlings under heat stress. Bot Stud, 60(1): 23. |
| [141] | Xu F F, Ye L M, Fu S Q. 2010. Effects of salicylic acid on resistance to cold stress of rice seedlings. Guangdong Agric Sci, 37: 18-20. (in Chinese with English abstract) |
| [142] | Xu N, Chu Y L, Chen H L, Li X X, Wu Q, Jin L, Wang G X, Huang J L. 2018. Rice transcription factor OsMADS25 modulates root growth and confers salinity tolerance via the ABA-mediated regulatory pathway and ROS scavenging. PLoS Genet, 14(10): e1007662. |
| [143] | Xu X Z, Wang T, Wan W, Li S H, Zhu G H. 2018. ABA biosynthesis gene OsNCED3 confers drought stress tolerance in rice. Acta Agron Sin, 44(1): 24-31. (in Chinese with English abstract) |
| [144] | Xu Y X, Xiao M Z, Liu Y, Fu J L, He Y, Jiang D A. 2017. The small auxin-up RNA OsSAUR45 affects auxin synthesis and transport in rice. Plant Mol Biol, 94(1/2): 97-107. |
| [145] | Yang J C, Miao W Q, Chen J. 2021. Roles of jasmonates and brassinosteroids in rice responses to high temperature stress: A review. Crop J, 9(5): 977-985. |
| [146] | Yeh S Y, Chen H W, Ng C Y, Lin C Y, Tseng T H, Li W H, Ku M S B. 2015. Down-regulation of cytokinin oxidase 2 expression increases tiller number and improves rice yield. Rice, 8(1): 36. |
| [147] |
Yu L J, Luo Y F, Liao B, Xie L J, Chen L, Xiao S, Li J T, Hu S N, Shu W S. 2012. Comparative transcriptome analysis of transporters, phytohormone and lipid metabolism pathways in response to arsenic stress in rice (Oryza sativa). New Phytol, 195(1): 97-112.
PMID |
| [148] | Yue E K, Cao H, Liu B H. 2020. OsmiR535, a potential genetic editing target for drought and salinity stress tolerance in Oryza sativa. Plants, 9(10): 1337. |
| [149] | Zafar S A, Zaidi S S E A, Gaba Y, Singla-Pareek S L, Dhankher O P, Li X Y, Mansoor S, Pareek A. 2020. Engineering abiotic stress tolerance via CRISPR/Cas-mediated genome editing. J Exp Bot, 71(2): 470-479. |
| [150] | Zeng Y F, Wen J Y, Zhao W B, Wang Q, Huang W C. 2020. Rational improvement of rice yield and cold tolerance by editing the three genes OsPIN5b, GS3, and OsMYB30 with the CRISPR- Cas9 system. Front Plant Sci, 10: 1663. |
| [151] | Zhang C X, Feng B H, Chen T T, Zhang X F, Tao L X, Fu G F. 2017. Sugars, antioxidant enzymes and IAA mediate salicylic acid to prevent rice spikelet degeneration caused by heat stress. Plant Growth Regul, 83(2): 313-323. |
| [152] | Zhang Q, Li J J, Zhang W J, Yan S N, Wang R, Zhao J F, Li Y J, Qi Z G, Sun Z X, Zhu Z G. 2012. The putative auxin efflux carrier OsPIN3t is involved in the drought stress response and drought tolerance. Plant J, 72(5): 805-816. |
| [153] |
Zhang Y, Li J, Chen S J, Ma X S, Wei H B, Chen C, Gao N N, Zou Y Q, Kong D Y, Li T F, Liu Z C, Yu S W, Luo L J. 2020. An APETALA2/ethylene responsive factor, OsEBP89 knockout enhances adaptation to direct-seeding on wet land and tolerance to drought stress in rice. Mol Genet Genomics, 295(4): 941-956.
PMID |
| [154] | Zhang Z J, Li F, Li D J, Zhang H W, Huang R F. 2010. Expression of ethylene response factor JERF1 in rice improves tolerance to drought. Planta, 232(3): 765-774. |
| [155] | Zhao F Y, Wang K, Zhang S Y, Ren J, Liu T, Wang X. 2014. Crosstalk between ABA, auxin, MAPK signaling, and the cell cycle in cadmium-stressed rice seedlings. Acta Physiol Plant, 36(7): 1879-1892. |
| [156] | Zhou Y, Xiong Q, Yin C C, Ma B, Chen S Y, Zhang J S. 2020. Ethylene biosynthesis, signaling, and crosstalk with other hormones in rice. Small Methods, 4(8): 1900278. |
| [157] | Zou J, Liu A L, Chen X B, Zhou X Y, Gao G F, Wang W F, Zhang X W. 2009. Expression analysis of nine rice heat shock protein genes under abiotic stresses and ABA treatment. J Plant Physiol, 166(8): 851-861. |
| [1] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [2] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [3] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [4] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [5] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [6] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [7] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [8] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [9] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [10] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [11] | Jiang Changjie, Liang Zhengwei, Xie Xianzhi. Priming for Saline-Alkaline Tolerance in Rice: Current Knowledge and Future Challenges [J]. Rice Science, 2023, 30(5): 417-425. |
| [12] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [13] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [14] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| [15] | Lu Xuedan, Li Fan, Xiao Yunhua, Wang Feng, Zhang Guilian, Deng Huabing, Tang Wenbang. Grain Shape Genes: Shaping the Future of Rice Breeding [J]. Rice Science, 2023, 30(5): 379-404. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||