
Rice Science ›› 2021, Vol. 28 ›› Issue (2): 166-177.DOI: 10.1016/j.rsci.2021.01.006
• Research Paper • Previous Articles Next Articles
Singh Priyanka1, Pokharia Chitra2, Shah Kavita2( )
)
Received:2020-04-11
Accepted:2020-08-12
Online:2021-03-28
Published:2021-03-28
Singh Priyanka, Pokharia Chitra, Shah Kavita. Exogenous Peroxidase Mitigates Cadmium Toxicity, Enhances Rhizobial Population and Lowers Root Knot Formation in Rice Seedlings[J]. Rice Science, 2021, 28(2): 166-177.
Add to citation manager EndNote|Ris|BibTeX
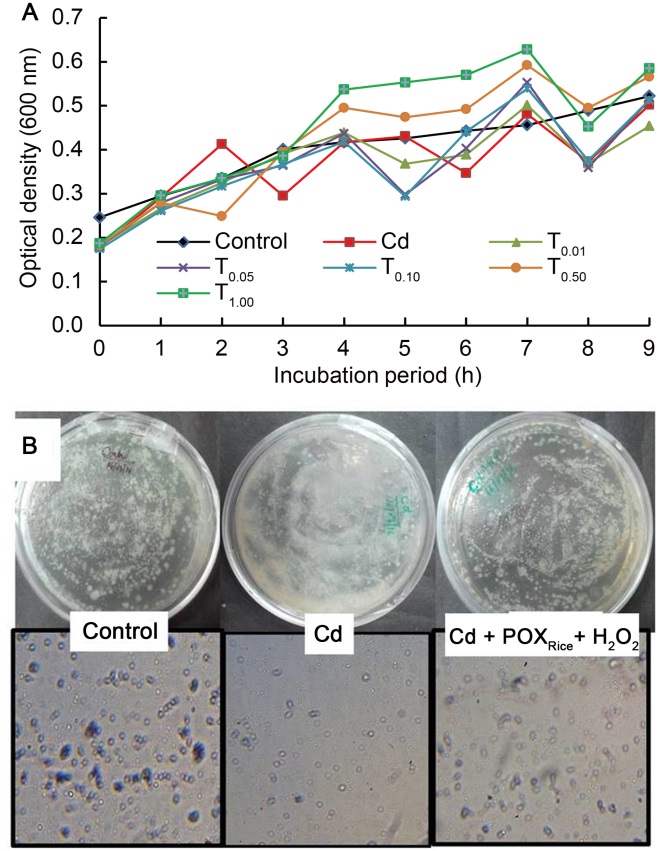
Fig. 1. Effects of exogenous peroxidase from rice (POXRice) + H2O2 on growth and population of Cd-exposed Rhizobium leguminosarum.A, Growth in Rhizobium population (OD600) with increasing incubation time pre-exposed to treatment (T) with 0.01, 0.05, 0.10, 0.50 and 1.00 mg/L Cd. B, Rhizobia grown on yeast extract mannitol agar media in the absence (control) and presence of Cd and/or exogenous POXRice + H2O2.
| Treatment | Cd | P | Fe | K | Mo | Mg | Mn |
|---|---|---|---|---|---|---|---|
| Control | 1.8 ± 0.1 a | 1 236.0 ± 7.8 a | 3 118.0 ± 10.4 c | 20 311.0 ± 35.5 b | 6.3 ± 0.2 c | 3 336.0 ± 31.0 b | 163.0 ± 4.0 b |
| Cd | 2.1 ± 0.1 b | 1 271.0 ± 12.6 b | 2 019.0 ± 10.2 a | 21 037.0 ± 32.7 c | 4.3 ± 0.2 a | 3 163.0 ± 15.5 a | 95.9 ± 4.2 a |
| Cd + POXRice + H2O2 | 2.0 ± 0.1 b | 1 322.0 ± 12.5 c | 2 513.0 ± 14.0 b | 18 525.0 ± 167.6 a | 5.2 ± 0.1 b | 3 201.0 ± 13.0 a | 161.0 ± 11.6 b |
Table 1. Effects of Cd-stress on accumulation of Cd and essential mineral nutrients in rice with or without exogenous peroxidase from rice (POXRice) + H2O2 treatment to 30-day-old rice plants.
| Treatment | Cd | P | Fe | K | Mo | Mg | Mn |
|---|---|---|---|---|---|---|---|
| Control | 1.8 ± 0.1 a | 1 236.0 ± 7.8 a | 3 118.0 ± 10.4 c | 20 311.0 ± 35.5 b | 6.3 ± 0.2 c | 3 336.0 ± 31.0 b | 163.0 ± 4.0 b |
| Cd | 2.1 ± 0.1 b | 1 271.0 ± 12.6 b | 2 019.0 ± 10.2 a | 21 037.0 ± 32.7 c | 4.3 ± 0.2 a | 3 163.0 ± 15.5 a | 95.9 ± 4.2 a |
| Cd + POXRice + H2O2 | 2.0 ± 0.1 b | 1 322.0 ± 12.5 c | 2 513.0 ± 14.0 b | 18 525.0 ± 167.6 a | 5.2 ± 0.1 b | 3 201.0 ± 13.0 a | 161.0 ± 11.6 b |
| Parameter | 15-day-old seedling | 30-day-old seedling | |||||
|---|---|---|---|---|---|---|---|
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | ||
| Fresh weight (g) | 0.22 ± 0.04 a | 0.27 ± 0.01 b | 0.24 ± 0.02 a | 0.85 ± 0.05a | 0.88 ± 0.07 a | 0.85 ± 0.05 a | |
| Dry weight (g) | 0.04 ± 0.00 a | 0.04 ± 0.00 a | 0.04 ± 0.01 a | 0.20 ± 0.02 a | 0.20 ± 0.04 a | 0.17 ± 0.01 a | |
| Shoot length (cm) | 14.70 ± 1.06 a | 16.00 ± 1.07 b | 15.00 ± 1.40 a | 23.00 ± 2.20 b | 20.60 ± 2.50 a | 21.30 ± 3.10 a | |
| Root length (cm) | 4.00 ± 0.08 a | 4.70 ± 0.80 a | 7.00 ± 1.10 b | 8.00 ± 1.40 a | 7.80 ± 1.80 a | 7.50 ± 1.20 a | |
| EL [mol/(L∙cm)] | 22.20 ± 3.02 a | 37.50 ± 5.20 b | 24.10 ± 2.70 a | 24.60 ± 3.10 a | 39.50 ± 5.30 c | 33.20 ± 4.60 b | |
| CC (mg/g) | 0.27 ± 0.06 b | 0.16 ± 0.04 a | 0.22 ± 0.09 b | 0.28 ± 0.05 b | 0.19 ± 0.05 a | 0.27 ± 0.02 b | |
| Cell viability (%) | 100.00 | 66.66 b | 97.00 a | 100.00 | 55.17 c | 96.00 a | |
| Parameter | 75-day-old seedling | 90-day-old seedling | |||||
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | ||
| Fresh weight (g) | 4.08 ± 0.33 b | 2.88 ± 0.12 a | 5.58 ± 0.64 c | 2.90 ± 0.30 a | 2.78 ± 0.20 a | 5.87 ± 0.90 c | |
| Dry weight (g) | 0.47 ± 0.02 a | 0.75 ± 0.05 b | 1.42 ± 0.04 c | 0.72 ± 0.06 a | 0.64 ± 0.04 a | 1.61 ± 0.07 b | |
| Shoot length (cm) | 35.00 ± 2.90 a | 32.00 ± 4.20 a | 39.00 ± 4.20 a | 42.20 ± 6.10 b | 37.40 ± 4.10 a | 39.00 ± 5.20 a | |
| Root length (cm) | 10.00 ± 2.80 b | 5.00 ± 1.10 a | 11.00 ± 2.20 b | 11.20 ± 2.70 b | 11.00 ± 2.20 b | 9.00 ± 2.10 a | |
| EL [mol/(L∙cm)] | 29.70 ± 2.50 b | 42.40 ± 5.30 c | 20.10 ± 3.70 a | 38.30 ± 3.90 b | 50.10 ± 5.70 c | 33.10 ± 4.20 a | |
| CC (mg/g) | 0.29 ± 0.05 a | 0.21 ± 0.09 a | 0.26 ± 0.08 a | 0.30 ± 0.01 b | 0.22 ± 0.04 a | 0.32 ± 0.03 b | |
| Cell viability (%) | 100.00 | 53.60 c | 92.85 a | 100.00 | 61.53 b | 88.46 a | |
Table 2. Effects of Cd-stress and simultaneous application of exogenous peroxidase from rice (POXRice) and H2O2 on growth parameters, electrolyte leakage (EL), cell viability and chlorophyll content (CC) in Cd-stressed rice plants.
| Parameter | 15-day-old seedling | 30-day-old seedling | |||||
|---|---|---|---|---|---|---|---|
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | ||
| Fresh weight (g) | 0.22 ± 0.04 a | 0.27 ± 0.01 b | 0.24 ± 0.02 a | 0.85 ± 0.05a | 0.88 ± 0.07 a | 0.85 ± 0.05 a | |
| Dry weight (g) | 0.04 ± 0.00 a | 0.04 ± 0.00 a | 0.04 ± 0.01 a | 0.20 ± 0.02 a | 0.20 ± 0.04 a | 0.17 ± 0.01 a | |
| Shoot length (cm) | 14.70 ± 1.06 a | 16.00 ± 1.07 b | 15.00 ± 1.40 a | 23.00 ± 2.20 b | 20.60 ± 2.50 a | 21.30 ± 3.10 a | |
| Root length (cm) | 4.00 ± 0.08 a | 4.70 ± 0.80 a | 7.00 ± 1.10 b | 8.00 ± 1.40 a | 7.80 ± 1.80 a | 7.50 ± 1.20 a | |
| EL [mol/(L∙cm)] | 22.20 ± 3.02 a | 37.50 ± 5.20 b | 24.10 ± 2.70 a | 24.60 ± 3.10 a | 39.50 ± 5.30 c | 33.20 ± 4.60 b | |
| CC (mg/g) | 0.27 ± 0.06 b | 0.16 ± 0.04 a | 0.22 ± 0.09 b | 0.28 ± 0.05 b | 0.19 ± 0.05 a | 0.27 ± 0.02 b | |
| Cell viability (%) | 100.00 | 66.66 b | 97.00 a | 100.00 | 55.17 c | 96.00 a | |
| Parameter | 75-day-old seedling | 90-day-old seedling | |||||
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | ||
| Fresh weight (g) | 4.08 ± 0.33 b | 2.88 ± 0.12 a | 5.58 ± 0.64 c | 2.90 ± 0.30 a | 2.78 ± 0.20 a | 5.87 ± 0.90 c | |
| Dry weight (g) | 0.47 ± 0.02 a | 0.75 ± 0.05 b | 1.42 ± 0.04 c | 0.72 ± 0.06 a | 0.64 ± 0.04 a | 1.61 ± 0.07 b | |
| Shoot length (cm) | 35.00 ± 2.90 a | 32.00 ± 4.20 a | 39.00 ± 4.20 a | 42.20 ± 6.10 b | 37.40 ± 4.10 a | 39.00 ± 5.20 a | |
| Root length (cm) | 10.00 ± 2.80 b | 5.00 ± 1.10 a | 11.00 ± 2.20 b | 11.20 ± 2.70 b | 11.00 ± 2.20 b | 9.00 ± 2.10 a | |
| EL [mol/(L∙cm)] | 29.70 ± 2.50 b | 42.40 ± 5.30 c | 20.10 ± 3.70 a | 38.30 ± 3.90 b | 50.10 ± 5.70 c | 33.10 ± 4.20 a | |
| CC (mg/g) | 0.29 ± 0.05 a | 0.21 ± 0.09 a | 0.26 ± 0.08 a | 0.30 ± 0.01 b | 0.22 ± 0.04 a | 0.32 ± 0.03 b | |
| Cell viability (%) | 100.00 | 53.60 c | 92.85 a | 100.00 | 61.53 b | 88.46 a | |
| Parameter | Tissue | 15-day-old seedling | 30-day-old seedling | |||||
|---|---|---|---|---|---|---|---|---|
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | |||
| MDA content (nmol/g) | Shoot | 0.03 ± 0.01 a | 0.04 ± 0.02 a | 0.02 ± 0.02 a | 0.03 ± 0.01 a | 0.05 ± 0.02 a | 0.03 ± 0.04 a | |
| Root | 0.02 ± 0.01 b | 0.04 ± 0.01 c | 0.01 ± 0.01 a | 0.02 ± 0.01 ab | 0.04 ± 0.03 b | 0.02 ± 0.02 a | ||
| H2O2 content (µmol/g) | Shoot | 4.10 ± 0.04 a | 4.67 ± 0.06 b | 4.02 ± 0.03 a | 4.90 ± 0.06 a | 5.12 ± 0.08 a | 4.10 ± 0.05 a | |
| Root | 3.13 ± 0.91 a | 4.12 ± 0.53 b | 3.87 ± 0.45 b | 3.30 ± 0.26 a | 4.90 ± 0.59 b | 3.80 ± 0.78 a | ||
| O2·‒ content (nmol/g) | Shoot | 0.08 ± 0.01 a | 0.17 ± 0.06 b | 0.05 ± 0.08 a | 0.08 ± 0.05 a | 0.19 ± 0.03 a | 0.06 ± 0.03 a | |
| Root | 0.08 ± 0.01 b | 0.15 ± 0.02 c | 0.04 ± 0.01 a | 0.08 ± 0.02 a | 0.16 ± 0.05 a | 0.07 ± 0.01 a | ||
| Parameter | Tissue | 75-day-old seedling | 90-day-old seedling | |||||
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | |||
| MDA content (nmol/g) | Shoot | 0.04 ± 0.01 ab | 0.06 ± 0.02 b | 0.03 ± 0.01 a | 0.04 ± 0.01 a | 0.05 ± 0.02 a | 0.04 ± 0.01 a | |
| Root | 0.02 ± 0.01 a | 0.05 ± 0.00 b | 0.03 ± 0.01 ab | 0.03 ± 0.01 a | 0.04 ± 0.01 a | 0.03 ± 0.01 a | ||
| H2O2 content (µmol/g) | Shoot | 5.00 ± 0.10 b | 5.90 ± 0.30 c | 4.60 ± 0.23 a | 5.20 ± 0.30 a | 5.67 ± 0.50 a | 5.10 ± 0.60 a | |
| Root | 4.40 ± 0.47 ab | 5.30 ± 0.39 b | 4.10 ± 0.94 a | 5.10 ± 0.94 a | 5.40 ± 0.67 a | 4.00 ± 0.75 a | ||
| O2·‒ content (nmol/g) | Shoot | 0.09 ± 0.05 a | 0.20 ± 0.02 a | 0.08 ± 0.01 a | 0.09 ± 0.07 a | 0.23 ± 0.04 a | 0.09 ± 0.01 a | |
| Root | 0.09 ± 0.02 a | 0.19 ± 0.05 a | 0.08 ± 0.03 a | 0.09 ± 0.03 a | 0.21 ± 0.02 a | 0.09 ± 0.02 a | ||
Table 3. Effects of Cd-stress and simultaneous application of exogenous peroxidase from rice (POXRice) and H2O2 on malondialdehyde (MDA), H2O2 and oxidative (O2·?) damage levels in shoots and roots of Cd-stressed rice plants.
| Parameter | Tissue | 15-day-old seedling | 30-day-old seedling | |||||
|---|---|---|---|---|---|---|---|---|
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | |||
| MDA content (nmol/g) | Shoot | 0.03 ± 0.01 a | 0.04 ± 0.02 a | 0.02 ± 0.02 a | 0.03 ± 0.01 a | 0.05 ± 0.02 a | 0.03 ± 0.04 a | |
| Root | 0.02 ± 0.01 b | 0.04 ± 0.01 c | 0.01 ± 0.01 a | 0.02 ± 0.01 ab | 0.04 ± 0.03 b | 0.02 ± 0.02 a | ||
| H2O2 content (µmol/g) | Shoot | 4.10 ± 0.04 a | 4.67 ± 0.06 b | 4.02 ± 0.03 a | 4.90 ± 0.06 a | 5.12 ± 0.08 a | 4.10 ± 0.05 a | |
| Root | 3.13 ± 0.91 a | 4.12 ± 0.53 b | 3.87 ± 0.45 b | 3.30 ± 0.26 a | 4.90 ± 0.59 b | 3.80 ± 0.78 a | ||
| O2·‒ content (nmol/g) | Shoot | 0.08 ± 0.01 a | 0.17 ± 0.06 b | 0.05 ± 0.08 a | 0.08 ± 0.05 a | 0.19 ± 0.03 a | 0.06 ± 0.03 a | |
| Root | 0.08 ± 0.01 b | 0.15 ± 0.02 c | 0.04 ± 0.01 a | 0.08 ± 0.02 a | 0.16 ± 0.05 a | 0.07 ± 0.01 a | ||
| Parameter | Tissue | 75-day-old seedling | 90-day-old seedling | |||||
| Control | Cd | Cd + POXRice + H2O2 | Control | Cd | Cd + POXRice + H2O2 | |||
| MDA content (nmol/g) | Shoot | 0.04 ± 0.01 ab | 0.06 ± 0.02 b | 0.03 ± 0.01 a | 0.04 ± 0.01 a | 0.05 ± 0.02 a | 0.04 ± 0.01 a | |
| Root | 0.02 ± 0.01 a | 0.05 ± 0.00 b | 0.03 ± 0.01 ab | 0.03 ± 0.01 a | 0.04 ± 0.01 a | 0.03 ± 0.01 a | ||
| H2O2 content (µmol/g) | Shoot | 5.00 ± 0.10 b | 5.90 ± 0.30 c | 4.60 ± 0.23 a | 5.20 ± 0.30 a | 5.67 ± 0.50 a | 5.10 ± 0.60 a | |
| Root | 4.40 ± 0.47 ab | 5.30 ± 0.39 b | 4.10 ± 0.94 a | 5.10 ± 0.94 a | 5.40 ± 0.67 a | 4.00 ± 0.75 a | ||
| O2·‒ content (nmol/g) | Shoot | 0.09 ± 0.05 a | 0.20 ± 0.02 a | 0.08 ± 0.01 a | 0.09 ± 0.07 a | 0.23 ± 0.04 a | 0.09 ± 0.01 a | |
| Root | 0.09 ± 0.02 a | 0.19 ± 0.05 a | 0.08 ± 0.03 a | 0.09 ± 0.03 a | 0.21 ± 0.02 a | 0.09 ± 0.02 a | ||
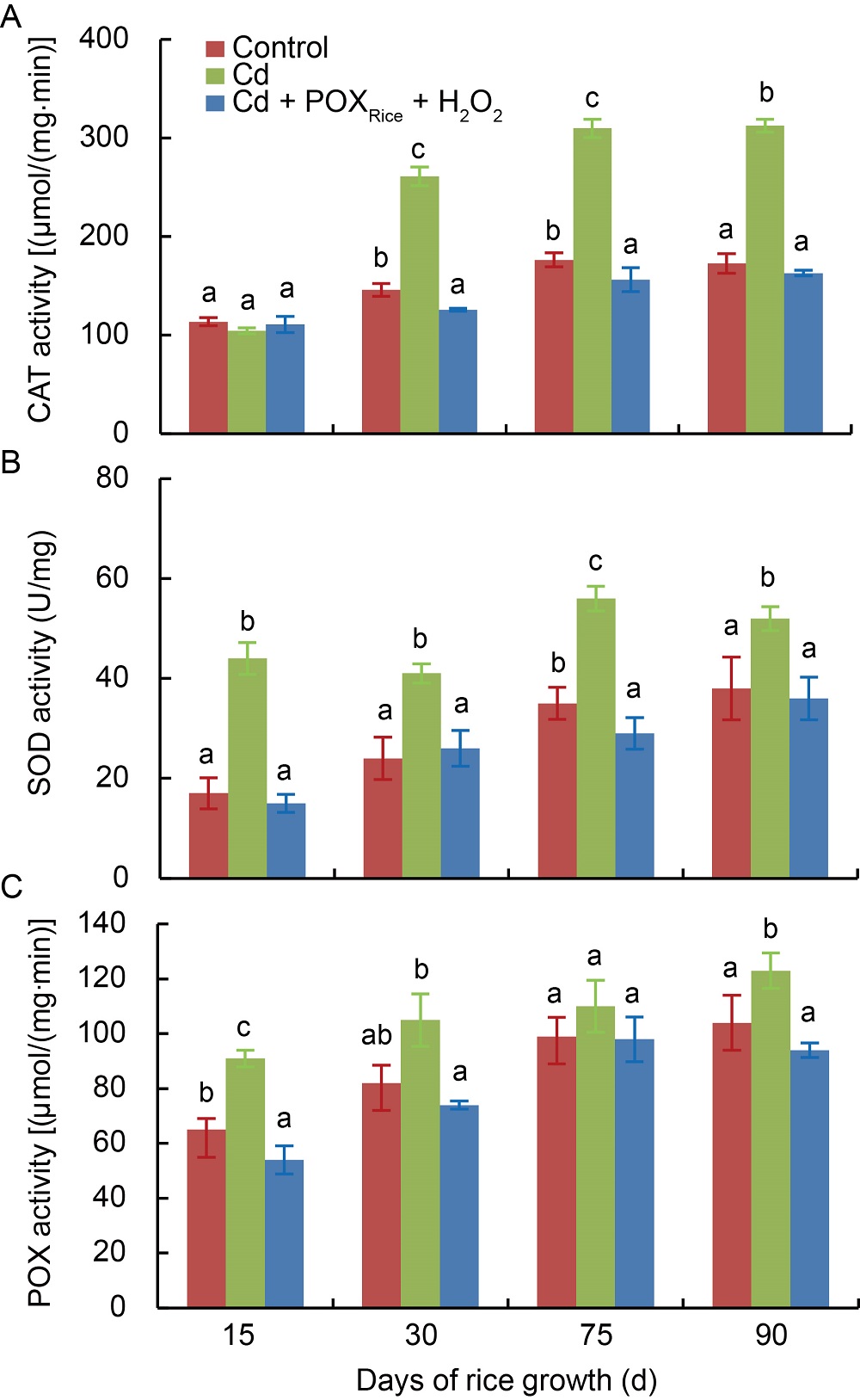
Fig. 2. Altered antioxidant enzyme activities.Activities of catalase (CAT) (A), superoxide dismutase (SOD) (B) and guaiacol peroxidase (POX) (C) in 1.00 mg/L Cd-stressed rice shoots in presence/absence of exogenous peroxidase from rice (POXRice) + H2O2 during 15?90 d of growth. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant difference among the treatments at the 0.05 level.
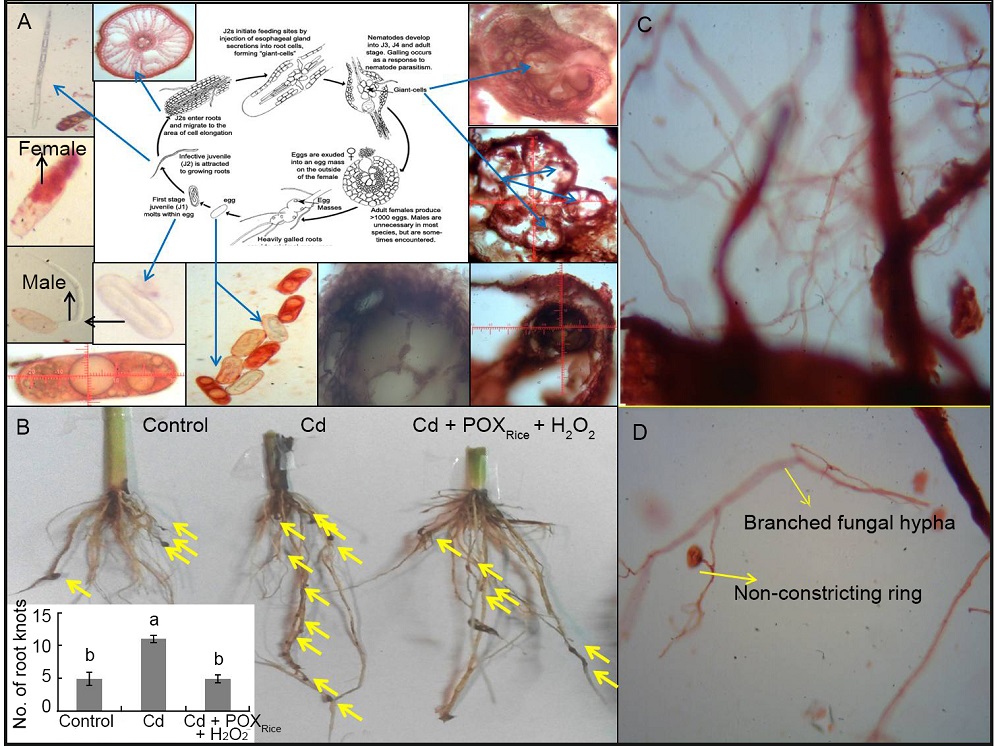
Fig. 3. Altered nematode (Meloidogyne spp.) infection in Cd-stressed rice plants.A, Life-stages of M. graminicola as seen in transverse section of a moderate- sized root knot under a compound microscope. B, Number of root-knot formation (inset graph) in control, Cd-exposure and its suppression in Cd + peroxidase from rice (POXRice) + H2O2. Yellow arrows show the locations of root knots. Values in the inset graph are Mean ± SD (n = 3). Different lower- case letters above the bars indicate significant difference among the treatments at the 0.05 level.C, Complex network of nematode- feeding fungal mycelia adhered to root knots.D, Presence of non-constriction rings in fungal mycelia indicating an increased protection to the Cd- stressed rice plants.
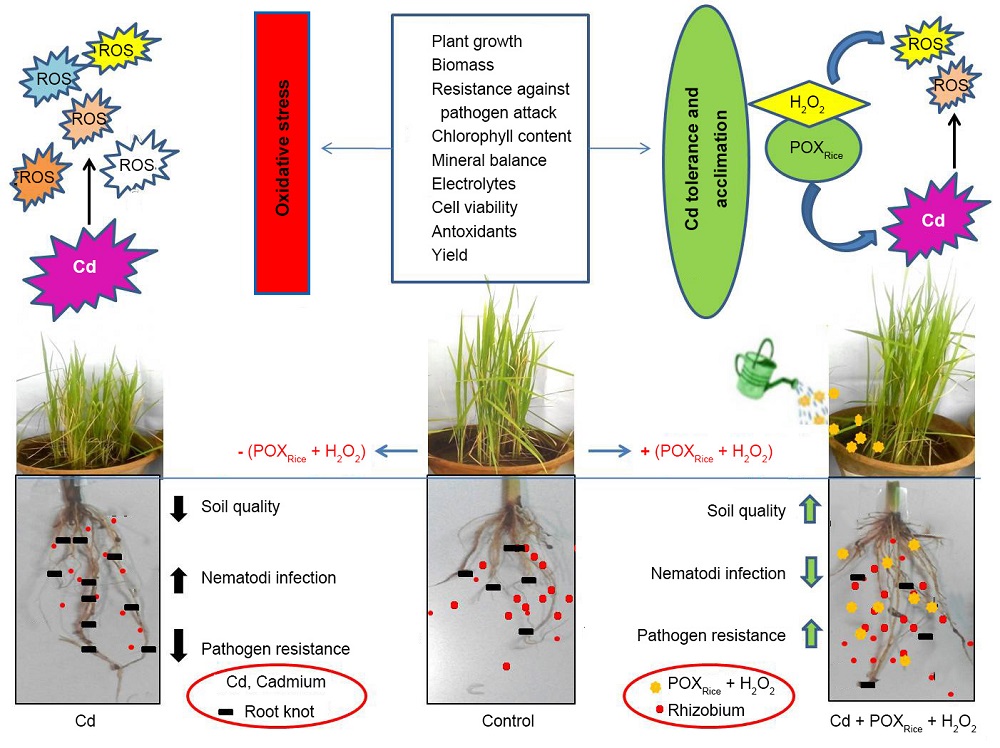
Fig. 4. Schematic representation showing altered biochemical parameters and nematode infection in Cd-stressed rice in presence/absence of exogenous POXRice + H2O2.
| [1] | Ali B, Gill R A, Yang S, Gill M B, Ali S, Rafiq M T, Zhou W.2014. Hydrogen sulfide alleviates cadmium-induced morphophysiological and ultrastructural changes in Brassica napus. Ecotoxicol Environ Saf, 110: 197-207. |
| [2] | Amtmann A, Troufflard S, Armengaud P.2008. The effect of potassium nutrition on pest and disease resistance in plants. Physiol Plant, 133: 682-691. |
| [3] | Baker C J, Mock N M.1994. An improved method for monitoring cell death in cell suspension and leaf disc assays using Evans blue.Plant Cell Tiss Organ Cult, 39: 7-12. |
| [4] | Bharwana S A, Ali S, Farooq M A, Ali B, Iqbal N, Abbas F, Ahmad M S A.2014. Hydrogen sulfide ameliorates lead-induced morphological, photosynthetic, oxidative damages and biochemical changes in cotton. Environ Sci Poll Res Int, 21: 717-731. |
| [5] | Bianucci E, Fabra A, Castro S.2011. Cadmium accumulation and tolerance in Brady Rhizobium spp. (peanut microsymbionts).Curr Microbiol, 62: 96-100. |
| [6] | Blokhina O, Virolainen E, Fagerstedt K V.2003. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann Bot, 91: 179-194. |
| [7] | Bolan N S, Makino T, Kunhikrishnan A, Kirkham M, Kim P J, Ishikawa S, Murakami M, Naidu R, Kirkham M B.2013. Cadmium contamination and its risk management in rice ecosystems. Adv Agron, 119: 183-273. |
| [8] | Cavalcanti F R, Oliveira J T, Martins-Miranda A S, Viégas R A, Silveira J A.2004. Superoxide dismutase, catalase and peroxidase activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. New Phytol, 163: 563-571. |
| [9] | Codex. 2006.Report of the 38th session of the Codex committee on food additives and contaminants. Hague, the Netherlands. 24-28 April, 2006. |
| [10] | DiToppi L S, Gabbrielli R.1999. Response to cadmium in higher plants. Environ Exp Bot, 41: 105-130. |
| [11] | Ellis M H, Dennis E S, Peacock W J.1999. Arabidopsis roots and shoots have different mechanisms for hypoxic stress tolerance. Plant Physiol, 119: 57-64. |
| [12] | Foyer C H, Lelandais M, Kunert K J.1994. Photooxidative stress in plants. Physiol Plant, 92: 696-717. |
| [13] | Gechev T S, Gadjev I, van Breusegem F, Inzé D, Dukiandjiev S, Toneva V, Minkov I.2002. Hydrogen peroxide protects tobacco from oxidative stress by inducing a set of antioxidant enzymes. Cell Mol Life Sci, 59: 708-714. |
| [14] | Gill S S, Tuteja N.2010. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem, 48: 909-930. |
| [15] | Hasanuzzaman M, Nahar K, Gill S S, Alharby H F, Razafindrabe B H, Fujita M.2017. Hydrogen peroxide pretreatment mitigates cadmium-induced oxidative stress in Brassica napus L.: An intrinsic study on antioxidant defense and glyoxalase systems.Front Plant Sci, 8: 115. |
| [16] | Kamnev A A, van der Lelie D.2000. Chemical and biological parameters as tools to evaluate and improve heavy metal phytoremediation. Biosci Rep, 20: 239-258. |
| [17] | Khare N, Goyary D, Singh N K, Shah P, Rathore M, Anandhan S, Sharma D, Arif M, Ahmed Z.2010. Transgenic tomato cv. Pusa Uphar expressing a bacterial mannitol-1-phosphate dehydrogenase gene confers abiotic stress tolerance.Plant Cell Tiss Organ Cult, 103: 267-277. |
| [18] | Kumar M, Shah K, Chand R.2014. Role of melanin in the biology of spot blotch pathogen of barley and its management. In: Hemantaranjan A. Advances in Plant Physiology. Vol. 15. Jodhpur, India: Scientific Publishers: 49-76. |
| [19] | Lamb C, Dixon R A.1997. The oxidative burst in plant disease resistance. Annu Rev Plant Biol, 48: 251-275. |
| [20] | Lavid N, Schwartz A, Yarden O, Tel-Or E.2001. The involvement of polyphenols and peroxidase activities in heavy-metal accumulation by epidermal glands of the waterlily (Nymphaeaceae). Planta, 212: 323-331. |
| [21] | Lei M, Tie B, Williams P N, Zheng Y, Huang Y.2011. Arsenic, cadmium, and lead pollution and uptake by rice (Oryza sativa L.) grown in greenhouse. J Soils Sediments, 11: 115-123. |
| [22] | Li H B, Zheng X W, Tao L X, Yang Y J, Gao L, Xiong J.2019. Aeration increases cadmium (Cd) retention by enhancing iron plaque formation and regulating pectin synthesis in the roots of rice (Oryza sativa) seedlings.Rice, 12: 28. |
| [23] | Li L, Wang Y Q, Shen W B.2012. Roles of hydrogen sulfide and nitric oxide in the alleviation of cadmium-induced oxidative damage in alfalfa seedling roots. Biometals, 25: 617-631. |
| [24] | Lowry O H, Rosenbrough R J, Farr A L, Randall R J.1951. Protein measurement with Folin-phenol reagent.J Biol Chem, 193: 265-275. |
| [25] | Mittler R, Blumwald E.2010. Genetic engineering for modern agriculture: Challenges and perspectives. Ann Rev Plant Biol, 61: 443-462. |
| [26] | Moskova I, Todorova D, Alexieva V, Ivanov S, Sergiev I.2009. Effect of exogenous hydrogen peroxide on enzymatic and nonenzymatic antioxidants in leaves of young pea plants treated with paraquat. Plant Growth Regul, 57: 193-202. |
| [27] | Mostofa M G, Rahman A, Ansary M M U, Watanabe A, Fujita M, Tran L S P.2015. Hydrogen sulfide modulates cadmium-induced physiological and biochemical responses to alleviate cadmium toxicity in rice.Sci Rep, 5: 14078. |
| [28] | Naeem A, Ghafoor A, Farooq M.2015. Suppression of cadmium concentration in wheat grains by silicon is related to its application rate and cadmium accumulating abilities of cultivars. J Sci Food Agric, 95: 2467-2472. |
| [29] | Nahakpam S, Shah K.2011. Expression of key antioxidant enzymes under combined effect of heat and cadmium toxicity in growing rice seedlings. Plant Growth Regul, 63: 23-35. |
| [30] | Nazar R, Iqbal N, Masood A, Khan M I R, Syeed S, Khan N A.2012. Cadmium toxicity in plants and role of mineral nutrients in its alleviation. Am J Plant Sci, 3: 1476-1489. |
| [31] | Noctor G, Foyer C H.1998. Ascorbate and glutathione: Keeping active oxygen under control. Ann Rev Plant Biol, 49: 249-279. |
| [32] | Popova N V, Plotnikov A N, Ziganshin R K, Deyev I E, Petrenko A G.2008. Analysis of proteins interacting with TRIP8b adapter. Biochemistry (Moscow), 73: 644-651. |
| [33] | Quan L J, Zhang B, Shi W W, Li H Y.2008. Hydrogen peroxide in plants: A versatile molecule of the reactive oxygen species network. J Integr Plant Biol, 50: 2-18. |
| [34] | Rai A C, Singh M, Shah K.2012. Effect of water withdrawal on formation of free radical, proline accumulation and activities of antioxidant enzymes in ZAT12-transformed transgenic tomato plants.Plant Physiol Biochem, 61: 108-114. |
| [35] | Roy S, Ihantola R, Hänninen O.1992. Peroxidase activity in lake macrophytes and its relation to pollution tolerance.Environ Exp Bot, 32: 457-464. |
| [36] | Salt D E, Blaylock M, Kumar N P, Dushenkov V, Ensley B D, Chet I, Raskin I.1995. Phytoremediation: A novel strategy for the removal of toxic metals from the environment using plants. Nat Biotechnol, 13: 468-474. |
| [37] | Sandalio L M, Dalurzo H C, Gomez M,Romero-Puertas M C, Del Rio L A. 2001. Cadmium induced changes in the growth and oxidative metabolism of pea plants. J Exp Bot, 52: 2115-2126. |
| [38] | Schuller D J, Ban N, van Huystee R B, McPherson A, Poulos T L.1996. The crystal structure of peanut peroxidase. Structure, 4: 311-321. |
| [39] | Shah K, Dubey R S.1998. A 18 kDa cadmium inducible protein complex: Its isolation and characterization from rice (Oryza sativa L.) seedlings. J Plant Physiol, 152: 448-454. |
| [40] | Shah K, Nongkynrih J M.2007. Metal hyperaccumulation and bioremediation. Biol Plant, 51: 618-634. |
| [41] | Shah K, Nahakpam S.2012. Heat exposure alters the expression of SOD, POD, APX and CAT isoenzymes and mitigates low cadmium toxicity in seedlings of sensitive and tolerant rice cultivars. Plant Physiol Biochem, 57: 106-113. |
| [42] | Shah K, Kumar R G, Verma S, Dubey R S.2001. Effect of cadmium on lipid peroxidation, superoxide anion generation and activities of antioxidant enzymes in growing rice seedlings. Plant Sci, 161: 1135-1144. |
| [43] | Shah K, Singh P, Nahakpam S.2013. Effect of cadmium uptake and heat stress on root ultrastructure, membrane damage and antioxidative response in rice seedlings.J Plant Biochem Biotechnol, 22: 103-112. |
| [44] | Singh I, Shah K.2015. Evidences for suppression of cadmium induced oxidative stress in presence of sulphosalicylic acid in rice seedlings. Plant Growth Regul, 76: 99-110. |
| [45] | Singh P, Prakash R, Shah K.2012. Effect of organic solvents on peroxidases from rice and horseradish: Prospects for enzyme- based applications. Talanta, 97: 204-210. |
| [46] | Smeets K, Ruytinx J, Semane B, van Belleghem F, Remans T, van Sanden S, Vangronsveld J, Cuypers A.2008. Cadmium-induced transcriptional and enzymatic alterations related to oxidative stress. Environ Exp Bot, 63: 1-8. |
| [47] | Sohag A A, Tahjib-Ul-Arif M, Brestic M, Afrin S, Sakil M A, Hossain M T, Hossain M A, Hossain M A,.2020. Exogenous salicylic acid and hydrogen peroxide attenuate drought stress in rice. Plant Soil Environ, 66: 7-13. |
| [48] | Sun J, Wang R G, Zhang X, Yu Y C, Zhao R, Li Z Y, Chen S L.2013. Hydrogen sulfide alleviates cadmium toxicity through regulations of cadmium transport across the plasma and vacuolar membranes in Populus euphratica cells. Plant Physiol Biochem, 65: 67-74. |
| [49] | Xu C M, Chen L P, Chen S, Chu G, Wang D Y, Zhang X F.2020. Rhizosphere aeration improves nitrogen transformation in soil, and nitrogen absorption and accumulation in rice plants. Rice Sci, 27(2): 162-174. |
| [50] | Yoshida K, Kaothien P, Matsui T, Kawaoka A, Shinmyo A.2003. Molecular biology and application of plant peroxidase genes.Appl Microbiol Biotechnol, 60: 665-670. |
| [51] | Zeigler R S, Barclay A.2008. The relevance of rice.Rice, 1: 3-10. |
| [52] | Zhang J P, Zhou X H, Chen L, Cheng Z G, Chu J Y, Li Y M.2016. Comparison of the abundance and community structure of ammonia oxidizing prokaryotes in rice rhizosphere under three different irrigation cultivation modes.World J Microbiol Biotechnol, 32: 85. |
| [53] | Zhu Y, Qian W Q, Hua J.2010. Temperature modulates plant defense responses through NB-LRR proteins.PLoS Pathog, 6: e1000844. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | XIA Xiaodong, ZHANG Xiaobo, WANG Zhonghao, CHENG Benyi, Sun Huifeng, XU Xia, GONG Junyi, YANG Shihua, WU Jianli, SHI Yongfeng, XU Rugen. Mapping and Functional Analysis of LE Gene in a Lethal Etiolated Rice Mutant at Seedling Stage [J]. Rice Science, 2023, 30(6): 13-. |
| [7] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [8] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [9] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [10] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [11] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [12] | Jiang Changjie, Liang Zhengwei, Xie Xianzhi. Priming for Saline-Alkaline Tolerance in Rice: Current Knowledge and Future Challenges [J]. Rice Science, 2023, 30(5): 417-425. |
| [13] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [14] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [15] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||