
Rice Science ›› 2020, Vol. 27 ›› Issue (3): 201-214.DOI: 10.1016/j.rsci.2019.11.001
• Research Paper • Previous Articles Next Articles
Zhaowei Li1,2, Qian Zhao2, Fangmin Cheng2( )
)
Received:2019-05-24
Accepted:2019-11-20
Online:2020-05-28
Published:2020-01-17
Zhaowei Li, Qian Zhao, Fangmin Cheng. Sugar Starvation Enhances Leaf Senescence and Genes Involved in Sugar Signaling Pathways Regulate Early Leaf Senescence in Mutant Rice[J]. Rice Science, 2020, 27(3): 201-214.
Add to citation manager EndNote|Ris|BibTeX
| Gene name | Accession No. | Forward primer (5′-3′) | Reverse primer (5′-3′) |
|---|---|---|---|
| Actin-1 | X16280 | CAGCACATTCCAGCAGATGT | TAGGCCGGTTGAAAACTTTG |
| Hxk1 | AF372831 | GGGGTTGTTTTATGCATTGG | CTTCTGGGAGGTGGAAATCA |
| Hxk2 | AF372832 | GCAAGTCTCGATCCCAAGAG | CTTTCCCAGCAGTTCCAGAG |
| Frk1 | AF429948 | TGATGCGTTTGTTGGTGCTCT | CTTTCTTTGTGGCGGTGATTG |
| Frk2 | AF429947 | CATCAGCAAGGCAGCCAACT | CGAAGCAGCAACTAAATTAAATCG |
| CIN1 | AY342319 | TACACGGTGGGCATCTACAA | TCCAGCCATACCTTTCTTGG |
| CIN4 | AY578161 | CATGTTTGTGCCGGATACTG | TGCCATCACCATCTAACCAA |
| SuSy1 | AK100334 | TGACTGGTCTGGTTGAGCTG | ACAAAAGCACCCTTGGTGTC |
| SuSy2 | AK072074 | TCCTGGAGCTGACATGAGTG | AAGACGAGCCATGGAGAAGA |
| SuSy4 | AK102158 | AGCCAATGTGTTGGGCTTAC | GCATGATGTCCCTTTTGCTT |
| SUT1 | D87819 | GCCAAGGAAGTTCCATTCAA | TGAGGATCAGTTCCCTTTGG |
| SUT4 | AB091673 | GCATGCTGGCTACAGCAATA | AGCAGTCAGCTCAGCAGTCA |
| chFBP | AK062233 | TACGGCGAGTTCGTGCTGA | GGCTCCTTGAGGCTGTCCAT |
| cyFBP | AB007193 | CAGTGAATGAAGGGAATGCGA | CATTTGGGCTCTTCTTGTCTGC |
| SPS1 | NM_001051745 | GAGACTTCGGAGGACCTTGCA | TGGGAGTGTCTTGCTGTGCC |
| SPS2 | NM_001052643 | TGATAATGGGTAACCGTGAGGC | CAGAGTGCTTATGATGCTTGGGAT |
| SPS6 | NM_001064716 | CGTGGTAGAAAGAAGAGGGAGGA | CAGAGCGTGACAACGGAGTGA |
| SPS8 | NM_001068030 | GGAGATTCCTTGAGGGATGTTC | CCATTTATTGCCTGTGGTAGCC |
Supplemental Table 1. Primers used in quantitative real-time PCR.
| Gene name | Accession No. | Forward primer (5′-3′) | Reverse primer (5′-3′) |
|---|---|---|---|
| Actin-1 | X16280 | CAGCACATTCCAGCAGATGT | TAGGCCGGTTGAAAACTTTG |
| Hxk1 | AF372831 | GGGGTTGTTTTATGCATTGG | CTTCTGGGAGGTGGAAATCA |
| Hxk2 | AF372832 | GCAAGTCTCGATCCCAAGAG | CTTTCCCAGCAGTTCCAGAG |
| Frk1 | AF429948 | TGATGCGTTTGTTGGTGCTCT | CTTTCTTTGTGGCGGTGATTG |
| Frk2 | AF429947 | CATCAGCAAGGCAGCCAACT | CGAAGCAGCAACTAAATTAAATCG |
| CIN1 | AY342319 | TACACGGTGGGCATCTACAA | TCCAGCCATACCTTTCTTGG |
| CIN4 | AY578161 | CATGTTTGTGCCGGATACTG | TGCCATCACCATCTAACCAA |
| SuSy1 | AK100334 | TGACTGGTCTGGTTGAGCTG | ACAAAAGCACCCTTGGTGTC |
| SuSy2 | AK072074 | TCCTGGAGCTGACATGAGTG | AAGACGAGCCATGGAGAAGA |
| SuSy4 | AK102158 | AGCCAATGTGTTGGGCTTAC | GCATGATGTCCCTTTTGCTT |
| SUT1 | D87819 | GCCAAGGAAGTTCCATTCAA | TGAGGATCAGTTCCCTTTGG |
| SUT4 | AB091673 | GCATGCTGGCTACAGCAATA | AGCAGTCAGCTCAGCAGTCA |
| chFBP | AK062233 | TACGGCGAGTTCGTGCTGA | GGCTCCTTGAGGCTGTCCAT |
| cyFBP | AB007193 | CAGTGAATGAAGGGAATGCGA | CATTTGGGCTCTTCTTGTCTGC |
| SPS1 | NM_001051745 | GAGACTTCGGAGGACCTTGCA | TGGGAGTGTCTTGCTGTGCC |
| SPS2 | NM_001052643 | TGATAATGGGTAACCGTGAGGC | CAGAGTGCTTATGATGCTTGGGAT |
| SPS6 | NM_001064716 | CGTGGTAGAAAGAAGAGGGAGGA | CAGAGCGTGACAACGGAGTGA |
| SPS8 | NM_001068030 | GGAGATTCCTTGAGGGATGTTC | CCATTTATTGCCTGTGGTAGCC |
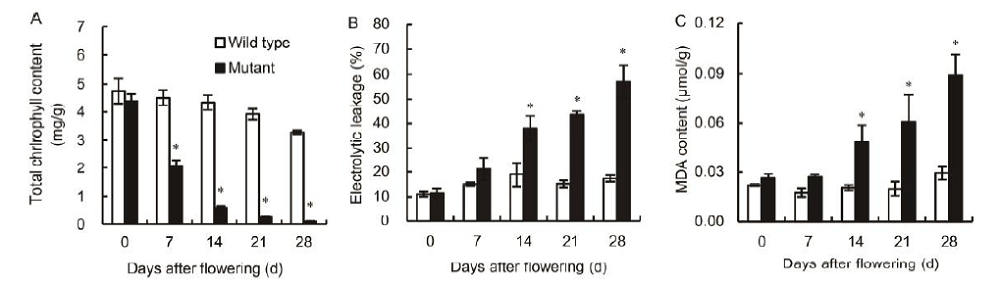
Fig. 1. Genotypic-dependent differences in total chlorophyll content (A), electrolytic leakage (B) and malondialdehyde (MDA) content (C) in flag leaves of wild type and mutant genotypes during grain-filling stage. Vertical bars represent standard errors (n = 3). Asterisks represent significant differences between the mutant and the wild type at the 0.05 level.
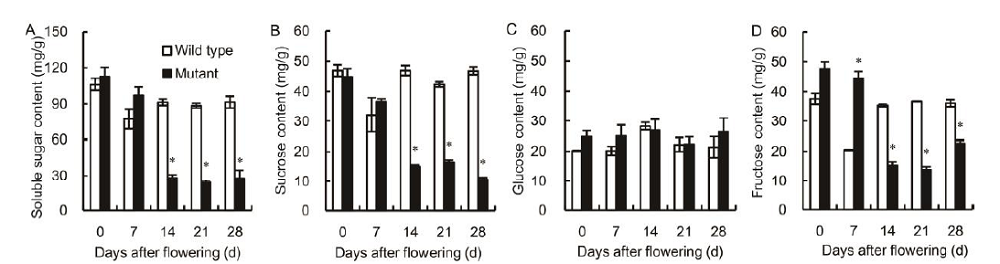
Fig. 2. Genotypic-dependent differences in soluble sugar (A), sucrose (B), glucose (C) and fructose (D) contents in flag leaves of wild type and mutant genotypes during grain-filling stage. Vertical bars represent standard errors (n = 3). Asterisks represent significant differences between the mutant and the wild type at the 0.05 level.
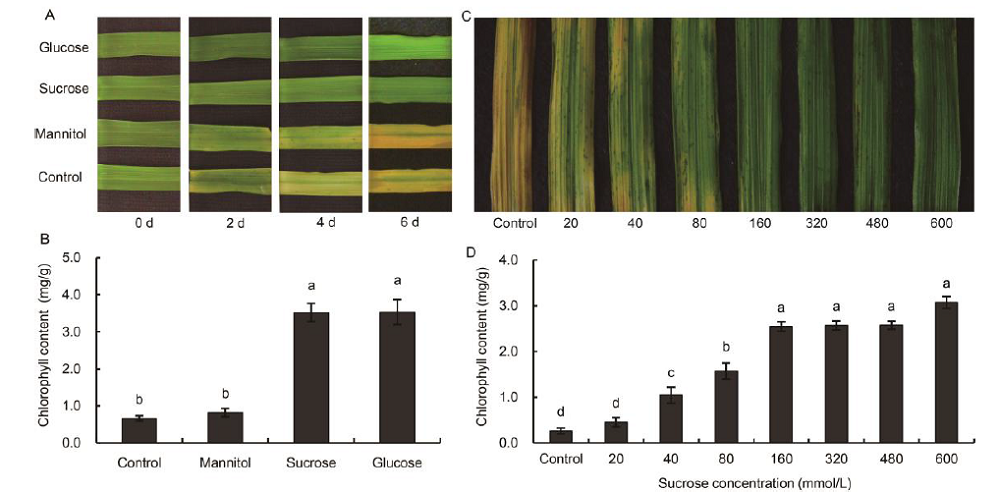
Fig. 3. Temporal analyses of the darkness-induced senescence of detached leaf segments incubated in 175 mmol/L glucose and sucrose, mannitol (osmosis control), hoagland nutrient solutions (control), and a gradient series of sucrose solutions for 6 d. A, Morphological changes exhibited by detached leaf segments after 0, 2, 4 and 6 d of incubation. B, Chlorophyll contents of detached leaf segments after 6 d of incubation in glucose, sucrose, mannitol and control. C, Morphological changes exhibited by detached leaf segments incubated in 20, 40, 80, 160, 320, 480 and 600 mmol/L sucrose solutions. D, Chlorophyll contents of detached leaf segments incubated in 20, 40, 80, 160, 320, 480 and 600 mmol/L sucrose solutions. Vertical bars represent standard errors (n = 3). Values with the same lowercase letters are not significantly different between different sugar treatments at the 0.05 level.
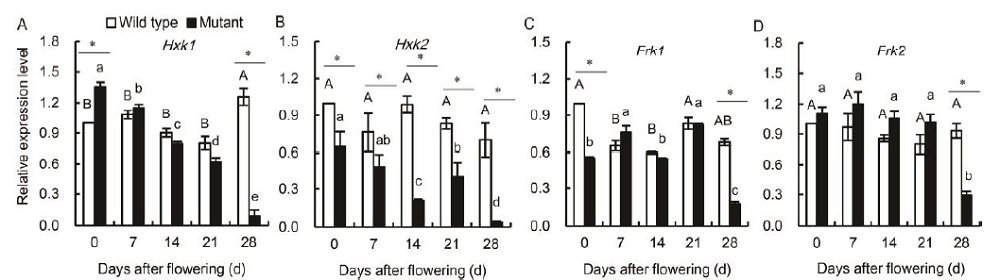
Fig. 4. Genotype-specific differences in the expression patterns of Hxk1 (A), Hxk2 (B), Frk1 (C) and Frk2 (D) in flag leaves of wild type and mutant genotypes during grain-filling stage. Vertical bars represent standard errors (n = 3). Asterisks represent significant differences between the wild type and the mutant at the 0.05 level. Values with the same uppercase and lowercase letters are not significantly different among wild type and mutants at the 0.05 level, respectively.
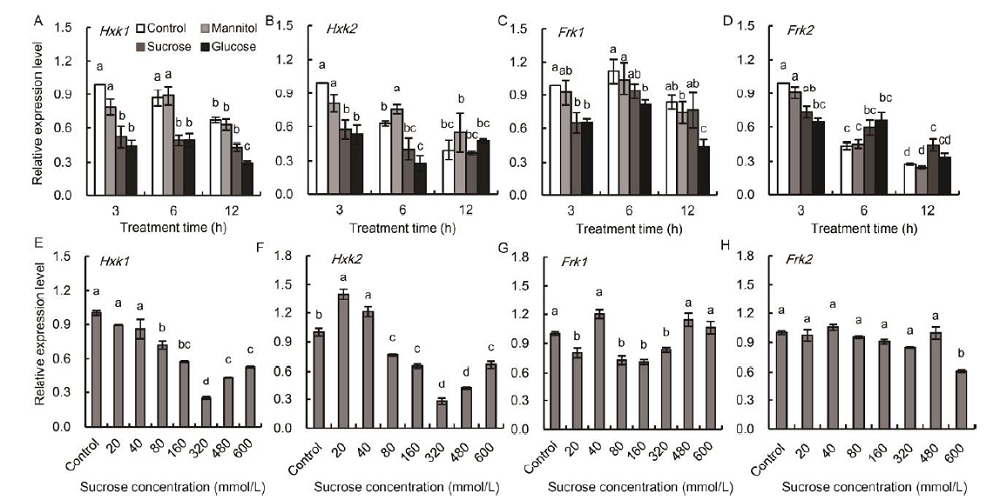
Fig. 5. Temporal analyses of Hxk1 (A), Hxk2 (B), Frk1 (C) and Frk2 (D) in detached wild type leaf segments incubated in 175 mmol/L sucrose, glucose and mannitol (osmosis control), and transcriptional analyses of Hxk1 (E), Hxk2 (F), Frk1 (G) and Frk2 (H) in the detached wild type leaf segments incubated in a gradient series of sucrose concentrations. For temporal analyses, the leaf segments were collected after 3, 6 and 12 h incubation, respectively, and treatment with Hoagland nutrient solution was set as the control. For transcriptional analyses, the leaf segments were collected after 6 h incubation. Vertical bars represent standard errors (n = 3). Values with the same lowercase letters are not significantly different between different sugar treatments at the 0.05 level.
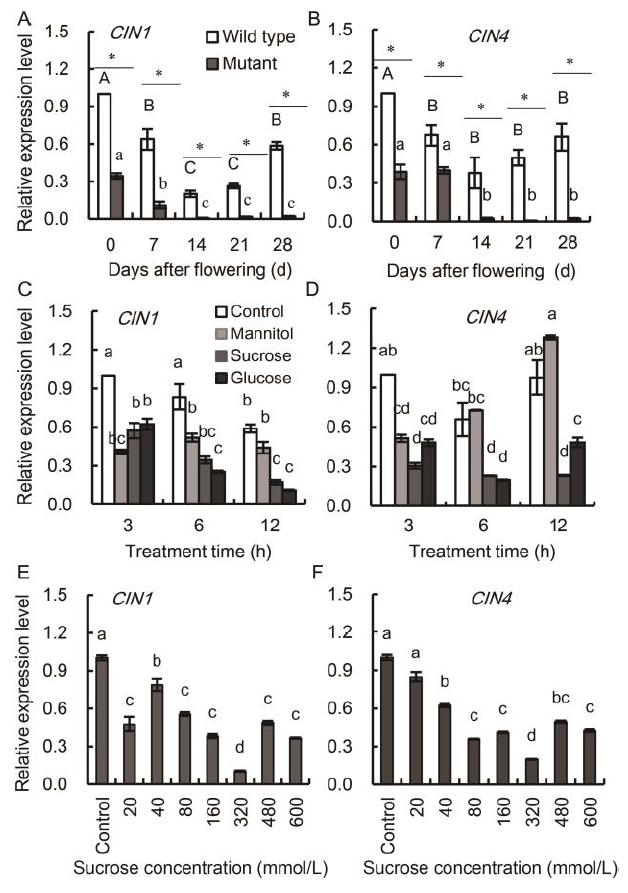
Fig. 6. Relative expression levels of CIN1 and CIN4 genes in the mutant and wild type leaves under different treatments.A and B, Genotypic-dependent differences in the expression patterns of CIN1 (A) and CIN4 (B) in the flag leaves of wild type and mutant genotypes during the grain-filling stage. Asterisks represent significant differences between the wild type and the mutant at the 0.05 level. Values with the same uppercase and lowercase letters are not significantly different among the wild type and the mutants at the 0.05 level, respectively. C and D, Temporal analyses of CIN1 (C) and CIN4 (D) in detached wild type leaf segments incubated in 175 mmol/L sucrose, glucose and mannitol (osmosis control). Leaf segments were collected after 3, 6 and 12 h incubation, respectively. Values with the same lowercase letters are not significantly different among different sugar treatments at the 0.05 level. E and F, Transcriptional analyses of CIN1 (E) and CIN4 (F) in the detached wild type leaf segments incubated in a gradient series of sucrose concentrations. Leaf segments were collected after 6 h incubation. Treatment with Hoagland nutrient solution was set as the control. Values with the same lowercase letters are not significantly different among different sugar concentration treatments at the 0.05 level.Vertical bars represent standard errors (n = 3).
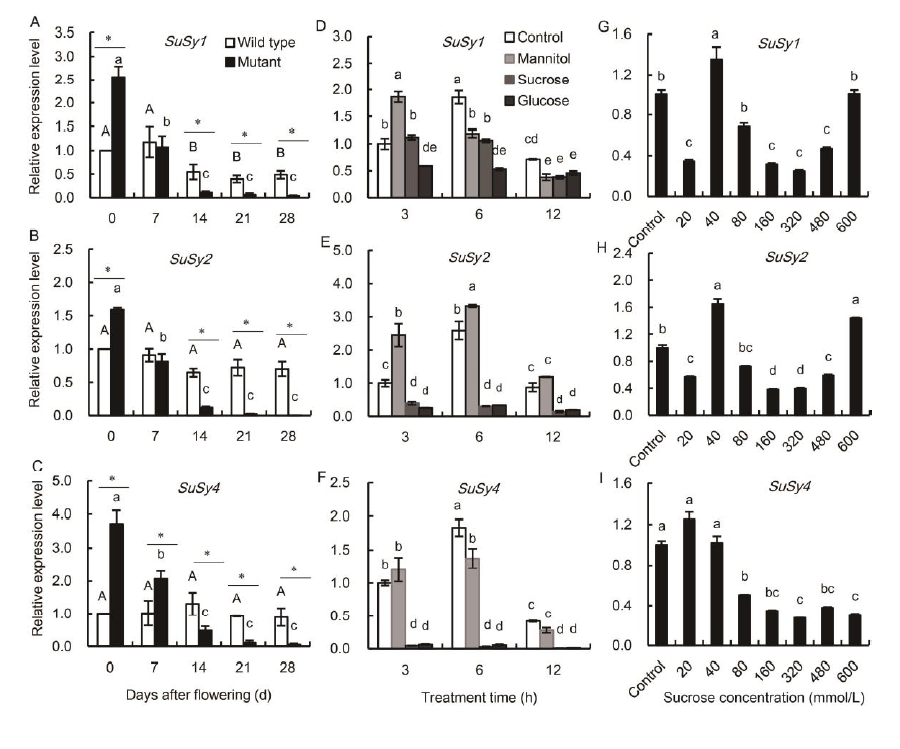
Fig. 7. Relative expression levels of SuSy1, SuSy2 and SuSy4 genes in the mutant and wild type leaves under different treatments.A?C, Genotypic-dependent differences in the expression patterns of SuSy1 (A), SuSy2 (B) and SuSy4 (C) in the flag leaves of wild type and mutant genotypes during the grain-filling stage. Asterisks represent significant differences between the wild type and the mutant at the 0.05 level. Values with the same uppercase and lowercase letters are not significantly different among the wild type and the mutants at the 0.05 level, respectively. D?F, Temporal analyses of SuSy1 (D), SuSy2 (E) and SuSy4 (F) in detached wild type leaf segments incubated in 175 mmol/L sucrose, glucose and mannitol (osmosis control). Leaf segments were collected after 3, 6 and 12 h incubation, respectively. Values with the same lowercase letters are not significantly different among different sugar treatments at the 0.05 level. G?I, The transcriptional analyses of SuSy1 (G), SuSy2 (H) and SuSy4 (I) in the detached wild-type leaf segments incubated in a gradient series of sucrose concentrations. Leaf segments were collected after 6 h incubation. Treatment with Hoagland nutrient solution was set as the control. Values with the same lowercase letters are not significantly different among different sugar concentration treatments at the 0.05 level. Vertical bars represent standard errors (n = 3).
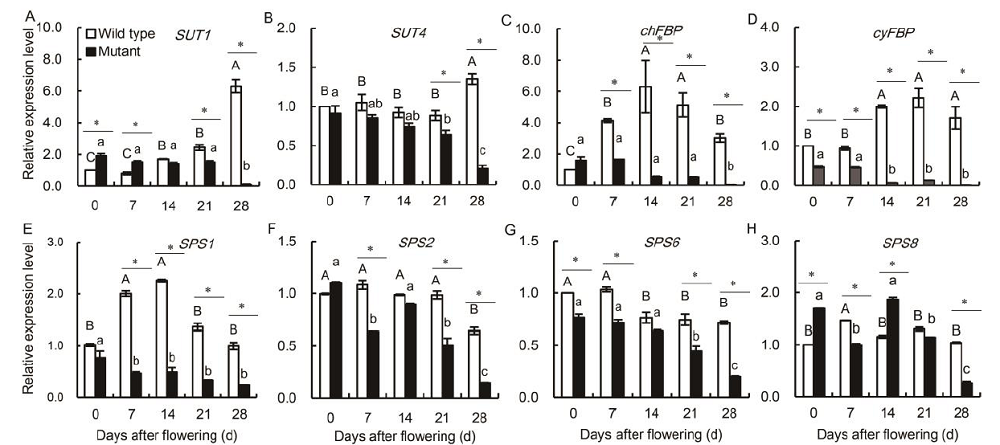
Fig. 8. Genotypic-dependent differences in the expression patterns of SUT1 (A) , SUT4 (B), chFBP (C), cyFBP (D), SPS1 (E), SPS2 (F), SPS6 (G) and SPS8 (H) in the flag leaves of wild type and mutant genotypes during the grain-filling stage.Vertical bars represent standard errors (n = 3). Asterisks represent significant differences between the wild type and the mutant at the 0.05 level. Values with the same uppercase and lowercase letters are not significantly different among wild type and mutants at the 0.05 level.
| [1] | Aoki N, Hirose T, Scofield G N, Whitfeld P R, Furbank R T.2003. The sucrose transporter gene family in rice.Plant Cell Physiol, 44(3): 223-232. |
| [2] | Balibrea Lara M E, Gonzalez Garcia M C, Fatima T, Ehness R, Lee T K, Proels R, Tanner W, Roitsch T.2004. Extracellular invertase is an essential component of cytokinin-mediated delay of senescence.Plant Cell, 16(5): 1276-1287. |
| [3] | Biswal B, Pandey J K.2018. Loss of photosynthesis signals a metabolic reprogramming to sustain sugar homeostasis during senescence of green leaves: Role of cell wall hydrolases.Photosynthetica, 56(1): 404-410. |
| [4] | Brouquisse R, Gaudillere J P, Raymond P.1998. Induction of a carbon-starvation-related proteolysis in whole maize plants submitted to light/dark cycle and to extended darkness.Plant Physiol, 117(4): 1281-1291. |
| [5] | Daie J.1993. Cytosolic fructose-1,6-bisphosphatase: A key enzyme in the sucrose biosynthetic pathway.Photosynth Res, 38(1): 5-14. |
| [6] | Davies H V, Shepherd L V T, Burrell M M, Carrari F, Urbanczyk- Wochniak E, Leisse A, Hancock R D, Taylor M, Viola R, Ross H, McRae D, Willmitzer L, Fernie A R.2005. Modulation of fructokinase activity of potato (Solanum tuberosum) results in substantial shifts in tuber metabolism. Plant Cell Physiol, 46(7): 1103-1115. |
| [7] | Dian W M, Jiang H W, Chen Q S, Liu F Y, Wu P.2003. Cloning and characterization of the granule-bound starch synthase II gene in rice: Gene expression is regulated by the nitrogen level, sugar and circadian rhythm.Planta, 218(2): 261-268. |
| [8] | Distelfeld A, Avni R, Fischer A M.2014. Senescence, nutrient remobilization, and yield in wheat and barley.J Exp Bot, 65(14): 3783-3798. |
| [9] | Essmann J, Schmitz-Thom I, Schön H, Sonnewald S, Weis E, Scharte J.2008. RNA interference-mediated repression of cell wall invertase impairs defense in source leaves of tobacco.Plant Physiol, 147(3): 1288-1299. |
| [10] | Feller U, Fischer A.1994. Nitrogen metabolism in senescing leaves.Crit Rev Plant Sci, 13(3): 241-273. |
| [11] | Gibson S I.2005. Control of plant development and gene expression by sugar signaling.Curr Opin Plant Biol, 8(1): 93-102. |
| [12] | Granot D.2007. Role of tomato hexose kinases.Funct Plant Biol, 34: 567-570. |
| [13] | Hanson J, Smeekens S.2009. Sugar perception and signaling: An update.Curr Opin Plant Biol, 12(5): 562-567. |
| [14] | Hensel L L, Grbić V, Baumgarten D A, Bleecker A B.1993. Developmental and age-related processes that influence the longevity and senescence of photosynthetic tissues inArabidopsis. Plant Cell, 5(5): 553-564. |
| [15] | International Rice Genome Sequencing Project (IRGSP). 2005. The map-based sequence of the rice genome.Nature, 436: 793-800. |
| [16] | Jang J C, León P, Zhou L, Sheen J.1997. Hexokinase as a sugar sensor in higher plants.Plant Cell, 9(1): 5-19. |
| [17] | Jin Y, Ni D A, Ruan Y L.2009. Posttranslational elevation of cell wall invertase activity by silencing its inhibitor in tomato delays leaf senescence and increases seed weight and fruit hexose level.Plant Cell, 21(7): 2072-2089. |
| [18] | Jongebloed U, Szederkényi J, Hartig K, Schobert C, Komor E.2004. Sequence of morphological and physiological events during natural ageing of a castor bean leaf: Sieve tube occlusions and carbohydrate back-up precede chlorophyll degradation.Physiol Plant, 120(2): 338-346. |
| [19] | Koch K.2004. Sucrose metabolism: Regulatory mechanisms and pivotal roles in sugar sensing and plant development.Curr Opin Plant Biol, 7(3): 235-246. |
| [20] | Lam E, Kato N, Lawton M.2001. Programmed cell death, mitochondria and the plant hypersensitive response.Nature, 411: 848-853. |
| [21] | Li Z W, Wang F B, Lei B T, Cao Z Z, Pan G, Cheng F M.2014. Genotypic-dependent alteration in transcriptional expression of various CAT isoenzyme genes inesl mutant rice and its relation to H2O2-induced leaf senescence. Plant Growth Regul, 73(3): 237-248. |
| [22] | Li Z W, Wang F B, Lin W W, Zhao Q, Liu J C, Cheng F M.2017. Carbon reserve and remobilization in leaf sheaths during the grain-filling stage in response to leaf early senescence.Acta Physiol Plant, 39: 10. |
| [23] | Lichtenthaler H K.1987. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes.Methods Enzymol, 148: 350-382. |
| [24] | Lim P O, Kim H J, Nam H G.2007. Leaf senescence.Annu Rev Plant Biol, 58: 115-136. |
| [25] | Lin W W, Guo X D, Pan X F, Li Z W.2018. Chlorophyll composition, chlorophyll fluorescence, and grain yield change inesl mutant rice. Int J Mol Sci, 19(10): 2945. |
| [26] | Lu S Y, Wang Z C, Niu Y J, Guo Z F, Huang B R.2008. Antioxidant responses of radiation-induced dwarf mutants of bermudagrass to drought stress.J Am Soc Hortic Sci, 133(3): 360-366. |
| [27] | Luo X L, Huang Q F.2011. Relationships between leaf and stem soluble sugar content and tuberous root starch accumulation in Cassava.J Agric Sci, 3(2): 64-72. |
| [28] | Ma L, Zhang D C, Miao Q S, Yang J, Xuan Y H, Hu Y B.2017. Essential role of sugar transporter OsSWEET11 during the early stage of rice grain filling.Plant Cell Physiol, 58(5): 863-873. |
| [29] | Masclaux C, Valadier M H, Brugiere N, Morot-Gaudry J F, Hirel B.2000. Characterization of the sink/source transition in tobacco (Nicotiana tabacum L.) shoots in relation to nitrogen management and leaf senescence. Planta, 211(4): 510-518. |
| [30] | Noh Y S, Amasino R M.1999. Identification of a promoter region responsible for the senescence-specific expression ofSAG12. Plant Mol Biol, 41(2): 181-194. |
| [31] | Noodén L D, Guiamét J J, John I.1997. Senescence mechanisms.Physiol Plant, 101(4): 746-753. |
| [32] | Parrott D, Yang L, Shama L, Fischer A M.2005. Senescence is accelerated, and several proteases are induced by carbon ‘feast’ conditions in barley (Hordeum vulgare L.) leaves. Planta, 222(6): 989-1000. |
| [33] | Pourtau N, Jennings R, Pelzer E, Pallas J, Wingler A.2006. Effect of sugar-induced senescence on gene expression and implications for the regulation of senescence inArabidopsis. Planta, 224(3): 556-568. |
| [34] | Quirino B F, Noh Y S, Himelblau E, Amasino R M.2000. Molecular aspects of leaf senescence.Trends Plant Sci, 5(7): 278-282. |
| [35] | Rolland F, Moore B, Sheen J.2002. Sugar sensing and signaling in plants.Plant Cell, 14: S185-S205. |
| [36] | Rose T L, Bonneau L, Der C, Marty-Mazars D, Marty F.2006. Starvation-induced expression of autophagy-related genes inArabidopsis. Biol Cell, 98(1): 53-67. |
| [37] | Saher S, Piqueras A, Hellin E, Olmos E.2004. Hyperhydricity in micropropagated carnation shoots: The role of oxidative stress.Physiol Plant, 120(1): 152-161. |
| [38] | Schmittgen T D, Livak K J.2008. Analyzing real-time PCR data by the comparative C(T) method.Nat Protoc, 3: 1101-1108. |
| [39] | Smeekens S, Ma J K, Hanson J, Rolland F.2010. Sugar signals and molecular networks controlling plant growth.Curr Opin Plant Biol, 13(3): 274-279. |
| [40] | Stein O, Granot D.2018. Plant fructokinases: Evolutionary, developmental, and metabolic aspects in sink tissues.Front Plant Sci, 9: 339. |
| [41] | Stein O, Secchi F, German M A, Damari-Weissler H, Aloni R, Holbrook N M, Zwieniecky M A, Granot D.2017. The tomato cytosolic fructokinaseFRK1 is important for phloem fiber development. Biol Plant, 62(2): 353-361. |
| [42] | Takahashi A, Kawasaki T, Wong H L, Suharsono U, Hirano H, Shimamoto K.2003. Hyperphosphorylation of a mitochondrial protein, prohibitin, is induced by calyculin A in a rice lesionmimic mutantcdr1. Plant Physiol, 132(4): 1861-1869. |
| [43] | Tang G Q, Luscher M, Sturm A.1999. Antisense repression and vacuolar and cell wall invertase in transgenic carrot alters early plant development and sucrose partitioning.Plant Cell, 11(2): 177-189. |
| [44] | van Doorn W G.2008. Is the onset of senescence in leaf cells of intact plants due to low or high sugar levels?J Exp Bot, 59(8): 1963-1972. |
| [45] | Wingler A, Purdy S, MacLean J A, Pourtau N.2006. The role of sugars in integrating environmental signals during the regulation of leaf senescence.J Exp Bot, 57(2): 391-399. |
| [46] | Wingler A, Roitsch T.2008. Metabolic regulation of leaf senescence: Interactions of sugar signaling with biotic and abiotic stress responses.Plant Biol, 10(S1): 50-62. |
| [47] | Woo H R, Kim H J, Nam H G, Lim P O.2013. Plant leaf senescence and death: Regulation by multiple layers of control and implications for aging in general.J Cell Sci, 126: 4823-4833. |
| [48] | Wu X Y, Kuai B K, Jia J Z, Jing H C.2012. Regulation of leaf senescence and crop genetic improvement.J Integr Plant Biol, 54(12): 936-952. |
| [49] | Xiao W, Sheen J, Jang J C.2000. The role of hexokinase in plant sugar signal transduction and growth and development.Plant Mol Biol, 44(4): 451-461. |
| [50] | Yang X, Gong P, Li K Y, Huang F D, Cheng F M, Pan G.2016. A single cytosine deletion in theOsPLS1 gene encoding vacuolar- type H+-ATPase subunit A1 leads to premature leaf senescence and seed dormancy in rice. J Exp Bot, 67(9): 2761-2776. |
| [51] | Yoshida S.2003. Molecular regulation of leaf senescence.Curr Opin Plant Biol, 6(1): 79-84. |
| [52] | Zhao H, Wang F B, Han Z Y, Shamsu-Ado Z, Li Z W, Pan G, Cheng F M.2018. Senescence-specific changes in endogenous IAA content and its conjugated status in rice flag leaf as affected by nitrogen level.Chin J Rice Sci, 32(6): 591-600. (in Chinese with English abstract) |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||