
Rice Science ›› 2021, Vol. 28 ›› Issue (2): 146-155.DOI: 10.1016/j.rsci.2021.01.004
• Research Paper • Previous Articles Next Articles
Qian Sun1, Shuo Yang1, Xiaofan Guo2, Siting Wang1, Xintong Jia1, Shuang Li3, Yuanhu Xuan1( )
)
Received:2020-02-07
Accepted:2020-07-06
Online:2021-03-28
Published:2021-03-28
About author:#These authors contributed equally to this work
Qian Sun, Shuo Yang, Xiaofan Guo, Siting Wang, Xintong Jia, Shuang Li, Yuanhu Xuan. RAVL1 Activates IDD3 to Negatively Regulate Rice Resistance to Sheath Blight Disease[J]. Rice Science, 2021, 28(2): 146-155.
Add to citation manager EndNote|Ris|BibTeX

Fig. 1. RAVL1 directly activated IDD3. A, Expression of IDD3 was examined and Ubiquitin was used as the reference gene to normalize gene expression level. The leaves from one-month-old seedlings of wild type (WT), ravl1 mutants (ravl1-1 and ravl1-2) and RAVL1 overexpressors (RAVL1 OX1 and RAVL1 OX2) were used for RNA extraction using the TRIzol.B, Diagrammatic representation of the 1.0 kb IDD3 promoter. P1 and P2 indicate the regions detected in the chromatin immunoprecipitation (ChIP)-PCR assay (C). C, ChIP-PCR was performed to analyze the binding affinity of RAVL1 to P1 and P2 regions (B). The anti-GFP antibody was used for immune- precipitation. D, Yeast one-hybrid assay was performed to analyze RAVL1 activation of the 1.0 kb IDD3 promoter. Yeast cells harboring either AD-RAVL1 or AD together with pIDD3-His or mpIDD3-His were grown on synthetic dropout media lacking either Leu (-L) or Leu and His (-LH). mp, Mutated at the P1 region.E, Transient expression assay was performed by co-transfection with p35S:RAVL1 and each of the vectors expressing the beta-glucuronidase gene (GUS) under the control of native (pIDD3) and E-box motif-mutated (mpIDD3) promoters in protoplast cells. The luciferase gene driven by the 35S promoter was used as an internal control to normalize the expression of GUS. Data in A, C and E represent Mean ± SE (n = 3). Different lowercase lowercase letters indicate significant differences at P < 0.05.
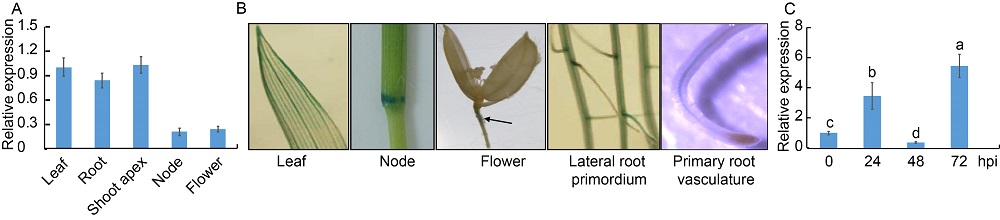
Fig. 2. IDD3 expression pattern in rice. A, Quantitative real-time PCR was performed on the mRNA extracted from the roots and leaves of one-week-old plants, shoot apecies of one- month-old plants, nodes and flowers of three-month-old plants. Ubiquitin was used as the control.B, Expression of IDD3 from one-week-old plant leaves or roots as well as nodes and flowers of three-month-old plants was analyzed using transgenic plants, in which GUS was expressed from an endogenous IDD3 promoter. Black arrowhead indicates GUS staining site in the flower photo.C, R. solani-infection mediated expression of IDD3 was analyzed at 0, 24, 48 and 72 h post-inoculation (hpi). The RNA was extracted by using the TRIzol, and Ubiquitin was used as the reference gene to normalize gene expression level.Data in A and C represent Mean ± SE (n = 3). Different lowercase letters indicate significant differences at P < 0.05.
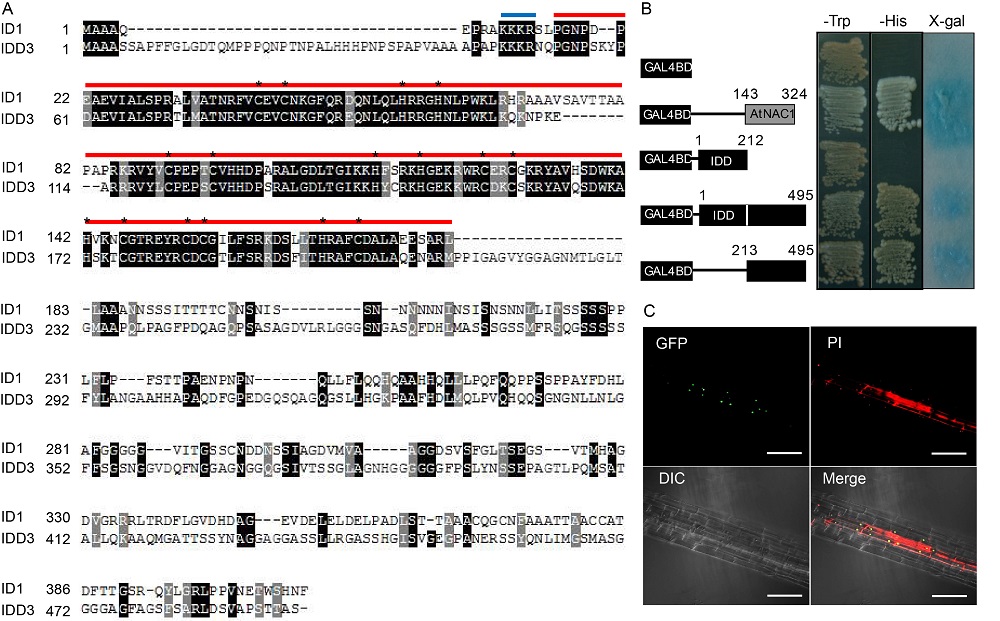
Fig. 3. Transactivation and nuclear localization of IDD3. A, Sequence alignment of rice IDD3 and ID1. Identical and similar amino acids are shown in black and gray boxes, respectively. Blue and red horizontal bars indicate the putative nuclear localization sequence and indeterminate domains, respectively. Asterisks in the red horizontal bar indicate the core amino acids of the two C2H2 and two C2HC zinc fingers. B, Transactivation activity of different regions of IDD3. DNA encoding the full-length IDD3 (495 aa), the N-terminal (212 aa), and the C-terminal (283 aa) regions of IDD3 was fused to the GAL4 DNA-binding domain and transformed into the yeast cells. AtNAC1 was used as the positive control. C, Confocal microscopic images of GFP overlapped with propidium iodide (PI) staining in a lateral root of IDD3-GFP transgenic plants (top left). The lower left panel shows light microscopy of the same tissue (DIC). Bars, 20 µm.
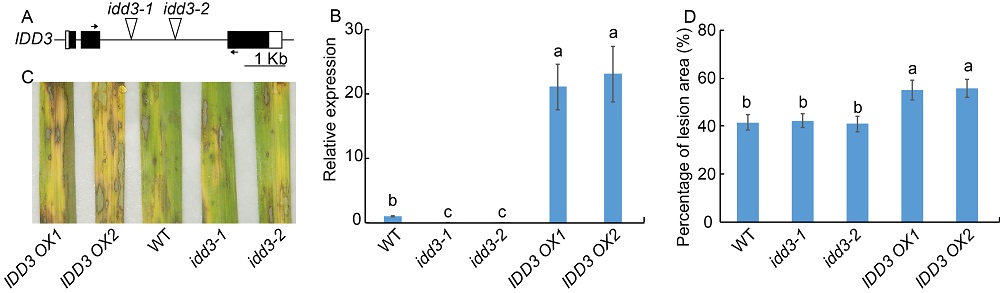
Fig. 4. Response of idd3 mutants and IDD3 overexpressors against sheath blight. A, Diagrammatic representation of the genomic structure along with the T-DNA insertion site. Black and white boxes indicate the exons and the untranslated regions, respectively. The triangles in the second intron indicate the T-DNA insertion site in the idd3 mutants (idd3-1 and idd3-2). Short horizontal arrows indicate the location of primers that were used in quantitative real-time PCR (qRT-PCR). B, qRT-PCR was performed to test the expression of IDD3 in wild type (WT), idd3 mutants and IDD3 overexpressors (IDD3 OX1 and IDD3 OX2). The leaves from one-month-old plants were used for RNA extraction using the TRIzol. Ubiquitin was used as the reference gene to normalize gene expression level.C, Phenotype response of WT, idd3 mutants and IDD3 overexpressors. D, Percentage of lesion area in WT, idd3mutants and IDD3 overexpressors.Data represent Mean ± SE (n = 3 in B, and n > 10 in D). Different lowercase letters indicate significant differences at P < 0.05.

Fig. 5. Response of IDD3 repressors to sheath blight. A, Quantitative real-time PCR was performed to examine the expression of IDD3 in wild type (WT) and IDD3 repressors (#1, #2, #3, #4 and #5). The leaves from one-month-old plants were used for RNA extraction using the TRIzol. Ubiquitin was used as the reference gene to normalize gene expression level.B, Phenotype response of IDD3 repressors (#1 and #3) to R. solani AG1-IA compared with WT. C, Percentage of lesion area in the IDD3 repressors (#1 and #3) compared with WT. Data represent Mean ± SE (n = 3 in A, and n > 10 in C). Different lowercase letters indicate significant differences at P < 0.05.
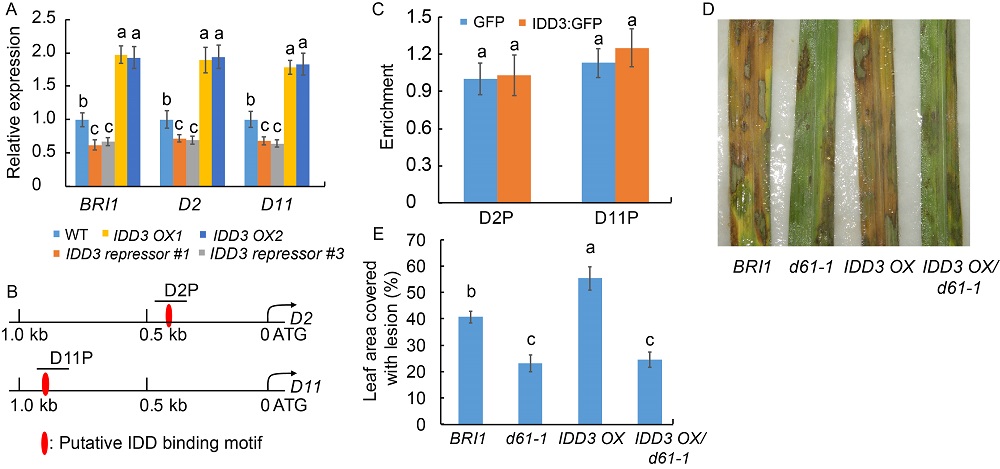
Fig. 6. Regulatory role of IDD3 and brassinosteroid (BR) signaling in rice defense to sheath blight (ShB). A, Quantitative real-time PCR was done to analyze BRI1, D2 and D11 expression in wild type (WT), idd3 mutants (idd3-1 and idd3-2) and IDD3 overexpressors (IDD3 OX1 and IDD3 OX2). The leaves from one-month-old plants were used for RNA extraction using the TRIzol. Ubiquitin was used as the reference gene to normalize gene expression level.B, Schematic diagram of 1.0 kb of D2 and D11 promoters. D2P and D11P indicate the regions detected in the chromatin immunoprecipitation (ChIP)-PCR assay (C). C, ChIP-PCR was done to analyze the binding affinity of IDD3 to D2P and D11P regions shown in (B). The anti-GFP antibody was used for immunoprecipitation. D, Phenotype response of BRI1, d61-1, IDD3 OX and IDD3 OX/d61-1 to R. solani AG1-IA compared with WT. E, Percentage of the lesion area in BRI1, d61-1, IDD3 OX and IDD3 OX/d61-1 compared with WT. Data represent Mean ± SE (n = 3 in A and C, and n > 10 in E). Different lowercase letters indicate significant differences at P < 0.05.
| [1] | Colasanti J, Yuan Z, Sundaresan V.1998. The indeterminate gene encodes a zinc finger protein and regulates a leaf-generated signal required for the transition to flowering in maize.Cell, 93(4): 593-603. |
| [2] | Cui D Y, Zhao J B, Jing Y J, Fan M Z, Liu J, Wang Z C, Xin W, Hu Y X.2013. The Arabidopsis IDD14, IDD15, and IDD16 cooperatively regulate lateral organ morphogenesis and gravitropism by promoting auxin biosynthesis and transport.PLoS Genet, 9(9): e1003759. |
| [3] | Dou M Z, Cheng S, Zhao B T, Xuan Y H, Shao M L.2016. The indeterminate domain protein ROC1 regulates chilling tolerance via activation of DREB1B/CBF1 in rice.Int J Mol Sci, 17(3): 233. |
| [4] | E Z G, Cheng B Y, Sun H W, Wang Y J, Zhu L F, Lin H, Wang L, Tong H H, Chen H Q.2019. Analysis on Chinese improved rice varieties in recent four decades.Chin J Rice Sci, 33(6): 523-531. (in Chinese with English abstract) |
| [5] | Eizenga G C, Lee F N, Rutger J N.2002. Screening Oryza species plants for rice sheath blight resistance.Plant Dis, 86(7): 808-812. |
| [6] | Feurtado J A, Huang D Q, Wicki-Stordeur L, Hemstock L E, Potentier M S, Tsang E W T, Cutler A J.2011. The Arabidopsis C2H2 zinc finger INDETERMINATE DOMAIN1/ENHYDROUS promotes the transition to germination by regulating light and hormonal signaling during seed maturation.Plant Cell, 23(5): 1772-1794. |
| [7] | Gao Y, Zhang C, Han X, Wang Z Y, Ma L, Yuan D P, Wu J N, Zhu X F, Liu J M, Li D P, Hu Y B, Xuan Y H.2018. Inhibition of OsSWEET11 function in mesophyll cells improves resistance of rice to sheath blight disease.Mol Plant Pathol, 19(9): 2149-2161. |
| [8] | Helliwell E E, Wang Q, Yang Y N.2013. Transgenic rice with inducible ethylene production exhibits broad-spectrum disease resistance to the fungal pathogens Magnaporthe oryzae and Rhizoctonia solani.Plant Biotechnol J, 11(1): 33-42. |
| [9] | Huang P, Yoshida H, Yano K, Kinoshita S, Kawai K, Koketsu E, Hattori M, Takehara S, Huang J, Hirano K, Ordonio R L, Matsuoka M, Ueguchi-Tanaka M.2018. OsIDD2, a zinc finger and INDETERMINATE DOMAIN protein, regulates secondary cell wall formation.J Integr Plant Biol, 60(2): 130-143. |
| [10] | Je B I, Piao H L, Park S J, Park S H, Kim C M, Xuan Y H, Park S H, Huang J, Do Choi Y, An G, Wong H L, Fujioka S, Kim M C, Shimamoto K, Han C D.2010. RAV-Like1 maintains brass- inosteroid homeostasis via the coordinated activation of BRI1 and biosynthetic genes in rice. Plant Cell, 22(6): 1777-1791. |
| [11] | Kim T W, Wang Z Y.2010. Brassinosteroid signal transduction from receptor kinases to transcription factors.Annu Rev Plant Biol, 61: 681-704. |
| [12] | Kouzai Y, Kimura M, Watanabe M, Kusunoki K, Osaka D, Suzuki T, Matsui H, Yamamoto M, Ichinose Y, Toyoda K, Matsuura T, Mori I C, Hirayama T, Minami E, Nishizawa Y, Inoue K, Onda Y, Mochida K, Noutoshi Y.2018. Salicylic acid-dependent immunity contributes to resistance against Rhizoctonia solani, a necrotrophic fungal agent of sheath blight, in rice and Brachypodium distachyon. New Phytol, 217(2): 771-783. |
| [13] | Kozaki A, Hake S, Colasanti J.2004. The maize ID1 flowering time regulator is a zinc finger protein with novel DNA binding properties.Nucl Acids Res, 32(5): 1710-1720. |
| [14] | Li J, Wen J Q, Lease K A, Doke J T, Tax F E, Walker J C.2002. BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling.Cell, 110(2): 213-222. |
| [15] | Li J M, Chory J.1997. A putative leucine-rich repeat receptor kinase involved in brassinosteroid signal transduction.Cell, 90(5): 929-938. |
| [16] | Liu J M, Park S J, Huang J, Lee E J, Xuan Y H, Je B I, Kumar V, Priatama R A, Raj K V, Kim S H, Min M K, Cho J H, Kim T H, Chandran A K N, Jung K H, Takatsuto S, Fujioka S, Han C D.2016. Loose Plant Architecture1 (LPA1) determines lamina joint bending by suppressing auxin signalling that interacts with C-22-hydroxylated and 6-deoxo brassinosteroids in rice.J Exp Bot, 67(6): 1883-1895. |
| [17] | Maeda S, Dubouzet J G, Kondou Y, Jikumaru Y, Seo S, Oda K, Matsui M, Hirochika H, Mori M.2019. The rice CYP78A gene BSR2 confers resistance to Rhizoctonia solani and affects seed size and growth in Arabidopsis and rice.Sci Rep, 9(1): 587. |
| [18] | Mao B Z, Liu X H, Hu D W, Li D B.2014. Co-expression of RCH10 and AGLU1 confers rice resistance to fungal sheath blight Rhizoctonia solani and blast Magnorpathe oryzae and reveals impact on seed germination.World J Microbiol Biotechnol, 30(4): 1229-1238. |
| [19] | Nam K H, Li J M.2002. BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling.Cell, 110(2): 203-212. |
| [20] | Park S J, Kim S L, Lee S, Je B I, Piao H L, Park S H, Kim C M, Ryu C H, Park S H, Xuan Y H, Colasanti J, An G, Han C D.2008. Rice Indeterminate 1 (OsId1) is necessary for the expression of Ehd1 (Early heading date 1) regardless of photoperiod. Plant J, 56(6): 1018-1029. |
| [21] | Prasad B, Eizenga G C.2008. Rice sheath blight disease resistance identified in Oryza spp. accessions.Plant Dis, 92(11): 1503-1509. |
| [22] | Rose M D, Winson F, Hieter P.1990. Methods in Yeast Genetics: A Laboratory Course Manual. New York, USA: Cold Spring Harbor Laboratory Press: 198. |
| [23] | Savary S, Castilla N P, Elazegui F A, McLaren C G, Ynalvez M A, Teng P S.1995. Direct and indirect effects of nitrogen supply and disease source structure on rice sheath blight spread.Phytopathology, 85(9): 959-965. |
| [24] | Savary S, Willocquet L, Elazegui F A, Castilla N P, Teng P S.2000. Rice pest constraints in tropical Asia: Quantification of yield losses due to rice pests in a range of production situations.Plant Dis, 84(3): 357-369. |
| [25] | Seo P J, Ryu J, Kang S K, Park C M.2011. Modulation of sugar metabolism by an INDETERMINATE DOMAIN transcription factor contributes to photoperiodic flowering in Arabidopsis.Plant J, 65(3): 418-429. |
| [26] | Shah J M, Raghupathy V, Veluthambi K.2009. Enhanced sheath blight resistance in transgenic rice expressing an endochitinase gene from Trichoderma virens.Biotechnol Lett, 31(2): 239-244. |
| [27] | Sun Q, Li T Y, Li D D, Wang Z Y, Li S, Li D P, Han X, Liu J M, Xuan Y H.2019. Overexpression of Loose Plant Architecture 1 increases planting density and resistance to sheath blight disease via activation of PIN-FORMED 1a in rice.Plant Biotechnol J, 17(5): 855-857. |
| [28] | Volz R, Kim S K, Mi J N, Mariappan K G, Siodmak A, Al-Babili S, Hirt H.2019. A chimeric IDD4 repressor constitutively induces immunity in Arabidopsis via the modulation of salicylic- and jasmonic acid homeostasis.Plant Cell Physiol, 60(7): 1536-1555. |
| [29] | Wang R, Lu L X, Pan X B, Hu Z L, Ling F, Yan Y, Liu Y M, Lin Y J.2015. Functional analysis of OsPGIP1 in rice sheath blight resistance.Plant Mol Biol, 87: 181-191. |
| [30] | Wang Z Y, Nakano T, Gendron J, He J X, Chen M, Vafeados D, Yang Y L, Fujioka S, Yoshida S, Asami T, Chory J.2002. Nuclear-localized BZR1 mediates brassinosteroid-induced growth and feedback suppression of brassinosteroid biosynthesis.Dev Cell, 2(4): 505-513. |
| [31] | Welch D, Hassan H, Blilou I, Immink R, Heidstra R, Scheres B.2007. Arabidopsis JACKDAW and MAGPIE zinc finger proteins delimit asymmetric cell division and stabilize tissue boundaries by restricting SHORT-ROOT action.Genes Dev, 21(17): 2196-2204. |
| [32] | Wu X R, Tang D, Li M, Wang K J, Cheng Z K.2013. Loose Plant Architecture1, an INDETERMINATE DOMAIN protein involved in shoot gravitropism, regulates plant architecture in rice.Plant Physiol, 161(1): 317-329. |
| [33] | Xie Q, Frugis G, Colgan D, Chua N H.2000. Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes Dev, 14(23): 3024-3036. |
| [34] | Xuan Y H, Priatama R A, Huang J, Je B I, Liu J M, Park S J, Piao H L, Son D Y, Lee J J, Park S H, Jung K H, Kim T H, Han C D.2013. Indeterminate domain 10 regulates ammonium-mediated gene expression in rice roots.New Phytol, 197(3): 791-804. |
| [35] | Yamaguchi M, Ohtani M, Mitsuda N, Kubo M, Ohme-Takagi M, Fukuda H, Demura T.2010. VND-INTERACTING2, a NAC domain transcription factor, negatively regulates xylem vessel formation in Arabidopsis.Plant Cell, 22(4): 1249-1263. |
| [36] | Yang C J, Zhang C, Lu Y N, Jin J Q, Wang X L.2011. The mechanisms of brassinosteroids’ action: From signal transduction to plant development.Mol Plant, 4(4): 588-600. |
| [37] | Yin Y H, Wang Z Y, Mora-Garcia S, Li J M, Yoshida S, Asami T, Chory J.2002. BES1 accumulates in the nucleus in response to brassinosteroids to regulate gene expression and promote stem elongation.Cell, 109(2): 181-191. |
| [38] | Yoo S D, Cho Y H, Sheen J.2007. Arabidopsis mesophyll protoplasts: A versatile cell system for transient gene expression analysis. Nat Protoc, 2(7): 1565-1572. |
| [39] | Yuan D P, Zhang C, Wang Z Y, Zhu X F, Xuan Y H.2018. RAVL1 activates brassinosteroids and ethylene signaling to modulate response to sheath blight disease in rice.Phytopathology, 108(9): 1104-1113. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||