
Rice Science ›› 2021, Vol. 28 ›› Issue (1): 89-98.DOI: 10.1016/j.rsci.2020.11.010
• Research Papers • Previous Articles Next Articles
Yanchang Luo1,2, Tingchen Ma2, Teo Joanne1, Zhixiang Luo2, Zefu Li2, Jianbo Yang2, Zhongchao Yin1,3( )
)
Received:2020-01-29
Accepted:2020-05-09
Online:2021-01-28
Published:2021-01-28
Yanchang Luo, Tingchen Ma, Teo Joanne, Zhixiang Luo, Zefu Li, Jianbo Yang, Zhongchao Yin. Marker-Assisted Breeding of Thermo-Sensitive Genic Male Sterile Line 1892S for Disease Resistance and Submergence Tolerance[J]. Rice Science, 2021, 28(1): 89-98.
Add to citation manager EndNote|Ris|BibTeX

Fig. 1. Marker-assisted breeding of 31892S.The donor line T4105 is a BC4F2 plant containing homozygous Pi9, Sub1A and Xa21 genes in KDML105 genetic background. The number of total plants screened and the number of positive plants identified are indicated in each generation. Only one plant, BC6F2-945, was identified from 1 550 BC6F2 individuals to contain homozygous alleles at the Pi9, Sub1A, tms5 and Xa21 loci. BC6F2-945 was designated as 31892S.
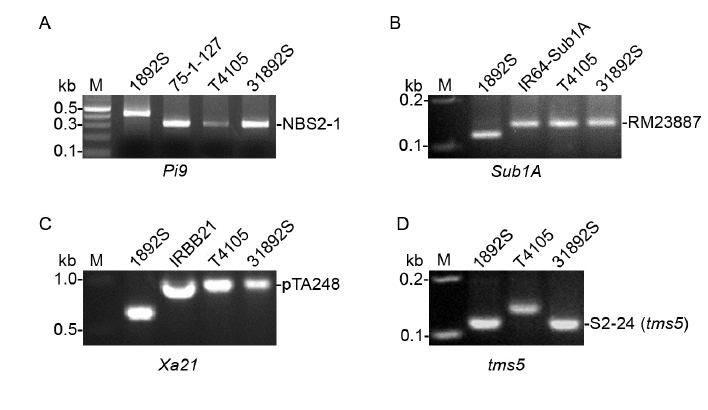
Fig. 2. Genotyping of molecular markers at the selected loci in 31892S. A, Genotyping of sequence-tagged site (STS) marker NBS2-1 at the Pi9 gene. 75-1-127 is the original donor line of the Pi9 gene.B, Genotyping of simple sequence repeat (SSR) marker RM23887 closely linked to the Sub1A gene. IR64-Sub1A is the original donor line of the Sub1A gene.C, Genotyping of STS marker pTA248 co-segregated with the Xa21 gene. IRBB21 is the original donor line of the Sub1A gene.D, Genotyping of SSR marker S2-24 closely linked to the tms5 gene in 1892S.M, Marker.
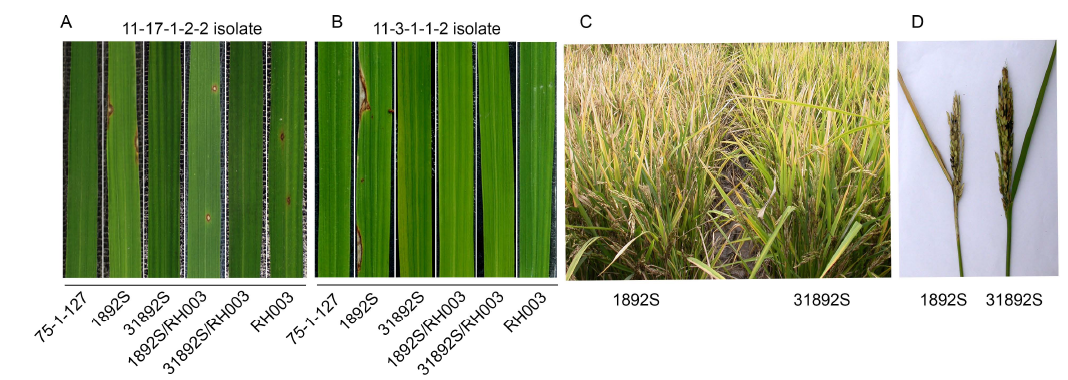
Fig. 3. Disease evaluation of rice lines for resistance to rice blast.A and B, Disease evaluation of rice lines to rice blast. Two-week-old rice seedlings were inoculated with M. oryzae isolates 11-17-1-2-2 (A) and 11-3-1-1-2 (B). The images were taken at 7 d after blast inoculation.C, Phenotypes of 1892S and 31892S in field. Image was taken at 30 d after heading in the field trial in Hefei, Anhui Province, China, in the summer season of 2017.D, A typical flag leaf and panicle of 1892S with neck blast infection and a healthy flag leaf and panicle of 31892S with no blast infection.75-1-127, Pi9 introgressive line; 1892S, Thermo-sensitive genic male sterile (TGMS) line; 31892S, Improved TGMS line in 1892S genetic background; RH003, Paternal line used for hybrid F1 production; 1892SS/RH003, F1 hybrid derived from the cross between 1892SS and RH003; 31892S/RH003, F1 hybrid derived from the cross between 31892S and RH003.
| Strain | Origin | IRBB21 | 1892S | 31892S | RH003 | 1892S/RH003 | 31892S/RH003 |
|---|---|---|---|---|---|---|---|
| Aust-2031 | Australia | 2.4 ± 0.6 (R) | 6.0 ± 1.3 (MR) | 5.1 ± 1.4 (MR) | 3.8 ± 1.5 (MR) | 5.0 ± 2.3 (MR) | 4.3 ± 1.0 (MR) |
| Aust-R3 | Australia | 2.7 ± 0.4 (R) | 1.0 ± 0.3 (R) | 0.8 ± 0.3 (R) | 0.7 ± 0.3 (R) | 0.8 ± 0.2 (R) | 1.4 ± 0.3 (R) |
| GD1358 | China | 2.6 ± 0.7 (R) | 11.2 ± 2.5 (S) | 4.6 ± 0.7 (MR) | 7.7 ± 2.8 (MS) | 9.4 ± 2.9 (S) | 5.2 ± 1.4 (MR) |
| HB17 | China | 2.3 ± 0.5 (R) | 17.8 ± 3.7 (S) | 2.5 ± 0.9 (R) | 23.1 ± 4.0 (S) | 24.0 ± 4.4 (S) | 3.1 ± 2.1 (MR) |
| HB21 | China | 2.2 ± 0.3 (R) | 20.9 ± 4.5 (S) | 4.4 ± 1.0 (MR) | 20.4 ± 4.1 (S) | 24.7 ± 1.9 (S) | 3.8 ± 1.0 (MR) |
| HLJ72 | China | 2.0 ± 0.5 (R) | 3.2 ± 0.7 (MR) | 2.6 ± 0.4 (R) | 9.1 ± 2.5 (S) | 11.5 ± 3.2 (S) | 3.4 ± 0.8 (MR) |
| JS49-6 | China | 1.7 ± 0.4 (R) | 16.6 ± 2.7 (S) | 1.5 ± 0.6 (R) | 14.3 ± 3.0 (S) | 19.2 ± 3.3 (S) | 3.5 ± 1.6 (MR) |
| LN57 | China | 3.3 ± 0.6 (MR) | 13.1 ± 2.7 (S) | 1.4 ± 0.5 (R) | 15.4 ± 3.6 (S) | 18.4 ± 2.1 (S) | 3.1 ± 1.1 (MR) |
| NX42 | China | 2.0 ± 0.7 (R) | 17.5 ± 2.9 (S) | 1.6 ± 0.8 (R) | 19.2 ± 3.7 (S) | 21.6 ± 4.9 (S) | 3.8 ± 0.9 (MR) |
| ZHE173 | China | 1.6 ± 0.6 (R) | 14.2 ± 2.7 (S) | 1.1 ± 0.4 (R) | 14.9 ± 2.3 (S) | 13.4 ± 2.8 (S) | 2.7 ± 0.6 (MR) |
| CIAT1185 | Columbia | 2.1 ± 1.0 (R) | 16.5 ± 2.0 (S) | 5.6 ± 1.3 (MR) | 17.3 ± 6.1 (S) | 15.6 ± 3.2 (S) | 5.9 ± 1.5 (MR) |
| IXO56 | Indonesia | 4.2 ± 0.9 (MR) | 15.1 ± 1.6 (S) | 4.8 ± 2.3 (MR) | 17.6 ± 3.7 (S) | 16.3 ± 2.0 (S) | 5.6 ± 1.3 (MR) |
| H75373 | Japan | 2.0 ± 0.4 (R) | 10.6 ± 1.8 (S) | 1.1 ± 0.2 (R) | 20.9 ± 3.7 (S) | 18.9 ± 3.8 (S) | 1.5 ± 0.5 (R) |
| T7174 | Japan | 1.9 ± 0.7 (R) | 0.7 ± 0.4 (R) | 1.2 ± 0.4 (R) | 18.8 ± 4.3 (S) | 17.9 ± 1.9 (S) | 2.2 ± 0.4 (R) |
| JW89011 | Korea | 21.9 ± 1.9 (S) | 0.8 ± 0.4 (R) | 1.3 ± 0.4 (R) | 1.1 ± 0.4 (R) | 1.5 ± 0.8 (R) | 1.1 ± 0.3 (R) |
| K202 | Korea | 3.0 ± 1.1 (R) | 12.2 ± 2.4 (S) | 0.6 ± 0.2 (R) | 16.4 ± 5.1 (S) | 18.8 ± 2.7 (S) | 1.8 ± 0.9 (R) |
| PXO86 (R2) | the Philippines | 1.2 ± 0.4 (R) | 16.8 ± 1.9 (S) | 4.6 ± 1.7 (MR) | 17.1 ± 2.3 (S) | 21.9 ± 0.8 (S) | 5.5 ± 1.9 (MR) |
| PXO79 (R3) | the Philippines | 1.7 ± 0.4 (R) | 16.9 ± 2.1 (S) | 4.8 ± 0.8 (MR) | 11.9 ± 2.5 (S) | 14.0 ± 3.2 (S) | 4.2 ± 0.9 (MR) |
| PXO71 (R4) | the Philippines | 2.5 ± 0.6 (R) | 15.7 ± 2.9 (S) | 1.4 ± 0.6 (R) | 21.1 ± 3.0 (S) | 21.7 ± 2.4 (S) | 2.4 ± 0.7 (R) |
| PXO113 (R4) | the Philippines | 2.5 ± 0.7 (R) | 17.6 ± 2.9 (S) | 2.0 ± 0.6 (R) | 18.8 ± 1.6 (S) | 21.1 ± 1.7 (S) | 2.2 ± 1.0 (R) |
| PXO112 (R5) | the Philippines | 3.3 ± 0.6 (MR) | 4.0 ± 0.7 (MR) | 0.7 ± 0.4 (R) | 5.2 ± 1.4 (MR) | 4.3 ± 2.1 (MR) | 1.2 ± 0.3 (R) |
| 2 | Thailand | 12.6 ± 2.6 (S) | 12.1 ± 2.4 (S) | 4.4 ± 1.0 (MR) | 16.1 ± 2.6 (S) | 8.9 ± 2.6 (MS) | 6.0 ± 1.2 (MR) |
| R-7 | Thailand | 2.4 ± 0.5 (R) | 20.4 ± 4.1 (S) | 4.7 ± 1.0 (MR) | 30.1 ± 5.1 (S) | 25.4 ± 4.2 (S) | 4.0 ± 1.8 (MR) |
| 1947 | Cameroon | 18.2 ± 3.7 (S) | 6.8 ± 2.1 (MS) | 8.6 ± 2.5 (MS) | 9.0 ± 3.2 (MS) | 11.3 ± 4.1 (S) | 19.8 ± 5.2 (S) |
Table 1 Disease evaluation of IRBB21, 1892S, 31892S and F1 hybrids to rice bacterial blight.
| Strain | Origin | IRBB21 | 1892S | 31892S | RH003 | 1892S/RH003 | 31892S/RH003 |
|---|---|---|---|---|---|---|---|
| Aust-2031 | Australia | 2.4 ± 0.6 (R) | 6.0 ± 1.3 (MR) | 5.1 ± 1.4 (MR) | 3.8 ± 1.5 (MR) | 5.0 ± 2.3 (MR) | 4.3 ± 1.0 (MR) |
| Aust-R3 | Australia | 2.7 ± 0.4 (R) | 1.0 ± 0.3 (R) | 0.8 ± 0.3 (R) | 0.7 ± 0.3 (R) | 0.8 ± 0.2 (R) | 1.4 ± 0.3 (R) |
| GD1358 | China | 2.6 ± 0.7 (R) | 11.2 ± 2.5 (S) | 4.6 ± 0.7 (MR) | 7.7 ± 2.8 (MS) | 9.4 ± 2.9 (S) | 5.2 ± 1.4 (MR) |
| HB17 | China | 2.3 ± 0.5 (R) | 17.8 ± 3.7 (S) | 2.5 ± 0.9 (R) | 23.1 ± 4.0 (S) | 24.0 ± 4.4 (S) | 3.1 ± 2.1 (MR) |
| HB21 | China | 2.2 ± 0.3 (R) | 20.9 ± 4.5 (S) | 4.4 ± 1.0 (MR) | 20.4 ± 4.1 (S) | 24.7 ± 1.9 (S) | 3.8 ± 1.0 (MR) |
| HLJ72 | China | 2.0 ± 0.5 (R) | 3.2 ± 0.7 (MR) | 2.6 ± 0.4 (R) | 9.1 ± 2.5 (S) | 11.5 ± 3.2 (S) | 3.4 ± 0.8 (MR) |
| JS49-6 | China | 1.7 ± 0.4 (R) | 16.6 ± 2.7 (S) | 1.5 ± 0.6 (R) | 14.3 ± 3.0 (S) | 19.2 ± 3.3 (S) | 3.5 ± 1.6 (MR) |
| LN57 | China | 3.3 ± 0.6 (MR) | 13.1 ± 2.7 (S) | 1.4 ± 0.5 (R) | 15.4 ± 3.6 (S) | 18.4 ± 2.1 (S) | 3.1 ± 1.1 (MR) |
| NX42 | China | 2.0 ± 0.7 (R) | 17.5 ± 2.9 (S) | 1.6 ± 0.8 (R) | 19.2 ± 3.7 (S) | 21.6 ± 4.9 (S) | 3.8 ± 0.9 (MR) |
| ZHE173 | China | 1.6 ± 0.6 (R) | 14.2 ± 2.7 (S) | 1.1 ± 0.4 (R) | 14.9 ± 2.3 (S) | 13.4 ± 2.8 (S) | 2.7 ± 0.6 (MR) |
| CIAT1185 | Columbia | 2.1 ± 1.0 (R) | 16.5 ± 2.0 (S) | 5.6 ± 1.3 (MR) | 17.3 ± 6.1 (S) | 15.6 ± 3.2 (S) | 5.9 ± 1.5 (MR) |
| IXO56 | Indonesia | 4.2 ± 0.9 (MR) | 15.1 ± 1.6 (S) | 4.8 ± 2.3 (MR) | 17.6 ± 3.7 (S) | 16.3 ± 2.0 (S) | 5.6 ± 1.3 (MR) |
| H75373 | Japan | 2.0 ± 0.4 (R) | 10.6 ± 1.8 (S) | 1.1 ± 0.2 (R) | 20.9 ± 3.7 (S) | 18.9 ± 3.8 (S) | 1.5 ± 0.5 (R) |
| T7174 | Japan | 1.9 ± 0.7 (R) | 0.7 ± 0.4 (R) | 1.2 ± 0.4 (R) | 18.8 ± 4.3 (S) | 17.9 ± 1.9 (S) | 2.2 ± 0.4 (R) |
| JW89011 | Korea | 21.9 ± 1.9 (S) | 0.8 ± 0.4 (R) | 1.3 ± 0.4 (R) | 1.1 ± 0.4 (R) | 1.5 ± 0.8 (R) | 1.1 ± 0.3 (R) |
| K202 | Korea | 3.0 ± 1.1 (R) | 12.2 ± 2.4 (S) | 0.6 ± 0.2 (R) | 16.4 ± 5.1 (S) | 18.8 ± 2.7 (S) | 1.8 ± 0.9 (R) |
| PXO86 (R2) | the Philippines | 1.2 ± 0.4 (R) | 16.8 ± 1.9 (S) | 4.6 ± 1.7 (MR) | 17.1 ± 2.3 (S) | 21.9 ± 0.8 (S) | 5.5 ± 1.9 (MR) |
| PXO79 (R3) | the Philippines | 1.7 ± 0.4 (R) | 16.9 ± 2.1 (S) | 4.8 ± 0.8 (MR) | 11.9 ± 2.5 (S) | 14.0 ± 3.2 (S) | 4.2 ± 0.9 (MR) |
| PXO71 (R4) | the Philippines | 2.5 ± 0.6 (R) | 15.7 ± 2.9 (S) | 1.4 ± 0.6 (R) | 21.1 ± 3.0 (S) | 21.7 ± 2.4 (S) | 2.4 ± 0.7 (R) |
| PXO113 (R4) | the Philippines | 2.5 ± 0.7 (R) | 17.6 ± 2.9 (S) | 2.0 ± 0.6 (R) | 18.8 ± 1.6 (S) | 21.1 ± 1.7 (S) | 2.2 ± 1.0 (R) |
| PXO112 (R5) | the Philippines | 3.3 ± 0.6 (MR) | 4.0 ± 0.7 (MR) | 0.7 ± 0.4 (R) | 5.2 ± 1.4 (MR) | 4.3 ± 2.1 (MR) | 1.2 ± 0.3 (R) |
| 2 | Thailand | 12.6 ± 2.6 (S) | 12.1 ± 2.4 (S) | 4.4 ± 1.0 (MR) | 16.1 ± 2.6 (S) | 8.9 ± 2.6 (MS) | 6.0 ± 1.2 (MR) |
| R-7 | Thailand | 2.4 ± 0.5 (R) | 20.4 ± 4.1 (S) | 4.7 ± 1.0 (MR) | 30.1 ± 5.1 (S) | 25.4 ± 4.2 (S) | 4.0 ± 1.8 (MR) |
| 1947 | Cameroon | 18.2 ± 3.7 (S) | 6.8 ± 2.1 (MS) | 8.6 ± 2.5 (MS) | 9.0 ± 3.2 (MS) | 11.3 ± 4.1 (S) | 19.8 ± 5.2 (S) |

Fig. 4. Evaluation of rice lines for submergence tolerance.A, Phenotypes of plants at 14 d after germination and before submergence treatment.B, Phenotypes of plants after 14 d of submergence treatment. C, Phenotypes of plants after 7 d of recovery from submergence treatment. D, Viability of plants after 7 d of recovery from submergence treatment. The data represent Mean ± SD (n = 3, about 30 plants for each line in each treatment). The different capital letters indicate significant difference among rice lines at the 0.01 level according to the Duncan’s multiple range tests. 1, Sub1A donor line IR64-Sub1A (genotype: Sub1ASub1A); 2, 1892S; 3, 31892S; 4, RH003; 5, 1892S/RH003; 6, 31892S/RH003.
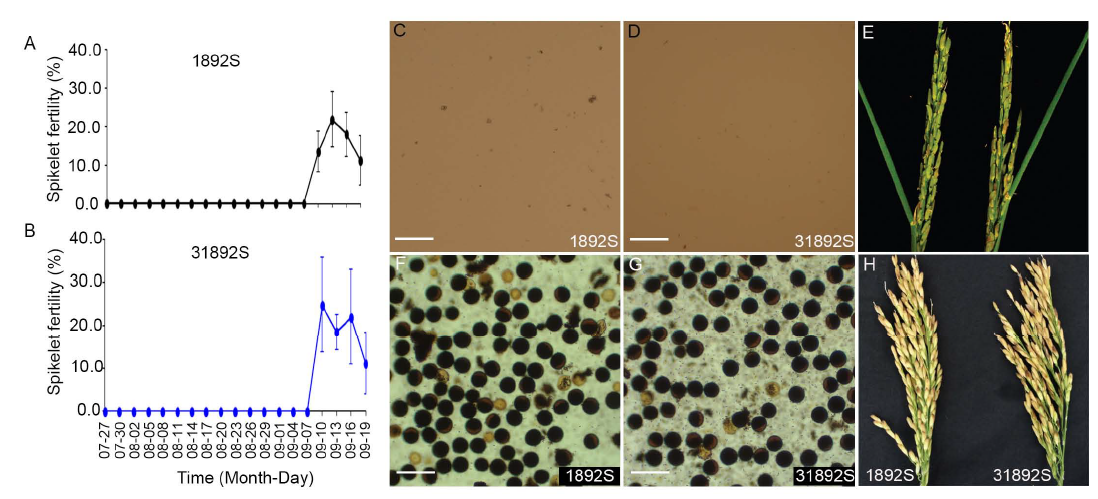
Fig. 5. Sterility-fertility conversion of 1892S and 31892S.A and B, Spikelet fertility of bagged panicles on 1892S (A) and 31892S (B) plants at different bagged time in 2017.C and D, Pollen grains of 1892S (C) and 31892S (D) plants grown under restrictive conditions with panicles flowering on 14 August, 2017. E, Panicles of 1892S and 31892S plants grown under restrictive conditions in Hefei, China with panicles flowering on 14 August 2017. F and G, Pollen grains of 1892S (C) and 31892S (D) after sterility-fertility alteration with panicles flowering on 18 September 2017 in Hefei, China. H, Panicles of 1892S and 31892S plants grown under permissive conditions in the winter season of 2016/2017 in Lingshui, China. In C, D, F and G, Pollen grains were stained with 1% I2-KI solution. Scale Bars, 100 µm.
| Trait | TGMS line under restrictive condition a | TGMS line under permissive condition b | F1 hybrid c | |||||
|---|---|---|---|---|---|---|---|---|
| 1892S | 31892S | 1892S | 31892S | 1892S/RH003 | 31892S/RH003 | |||
| Growth duration (d) | 123 | 123 | 136.3 ± 4.0 | 136.0 ± 4.6 | 138.0 ± 1.1 | 138.0 ± 1.1 | ||
| Plant height (cm) | 76.3 ± 0.5 | 80.2 ± 4.8 | 67.5 ± 7.1 | 67.9 ± 9.0 | 121.0 ± 5.9 | 126.7 ± 1.4 | ||
| Productive panicles per plant | NA | NA | 6.4 ± 1.1 | 6.6 ± 1.0 | 8.7 ± 1.1 | 8.7 ± 1.5 | ||
| Panicle length (cm) | 19.3 ± 0.1 | 19.7 ± 0.3 | 15.7 ± 1.3 | 16.7 ± 1.4 | 23.5 ± 0.6 | 23.7 ± 0.6 | ||
| Total spikelet number per panicle | 181.8 ± 7.5 | 175.6 ± 14.5 | 146.5 ± 5.3 | 138.1 ± 26.4 | 251.1 ± 34.6 | 232.4 ± 32.3 | ||
| Spikelet fertility (%) | NA | NA | 82.2 ± 8.2 | 72.3 ± 12.0 | 87.2 ± 4.6 | 82.8 ± 5.8 | ||
| 1000-grain weight (g) | NA | NA | 23.3 ± 0.5 | 23.7 ± 0.5 | 22.4 ± 0.2 | 23.4 ± 1.1 | ||
| Yield (t/hm2) | NA | NA | 5.8 ± 0.9 | 5.1 ± 0.9 | 10.5 ± 1.6 | 9.6 ± 1.8 | ||
Table 2 Agronomic traits of 1892S, 31892S and their F1 hybrids.
| Trait | TGMS line under restrictive condition a | TGMS line under permissive condition b | F1 hybrid c | |||||
|---|---|---|---|---|---|---|---|---|
| 1892S | 31892S | 1892S | 31892S | 1892S/RH003 | 31892S/RH003 | |||
| Growth duration (d) | 123 | 123 | 136.3 ± 4.0 | 136.0 ± 4.6 | 138.0 ± 1.1 | 138.0 ± 1.1 | ||
| Plant height (cm) | 76.3 ± 0.5 | 80.2 ± 4.8 | 67.5 ± 7.1 | 67.9 ± 9.0 | 121.0 ± 5.9 | 126.7 ± 1.4 | ||
| Productive panicles per plant | NA | NA | 6.4 ± 1.1 | 6.6 ± 1.0 | 8.7 ± 1.1 | 8.7 ± 1.5 | ||
| Panicle length (cm) | 19.3 ± 0.1 | 19.7 ± 0.3 | 15.7 ± 1.3 | 16.7 ± 1.4 | 23.5 ± 0.6 | 23.7 ± 0.6 | ||
| Total spikelet number per panicle | 181.8 ± 7.5 | 175.6 ± 14.5 | 146.5 ± 5.3 | 138.1 ± 26.4 | 251.1 ± 34.6 | 232.4 ± 32.3 | ||
| Spikelet fertility (%) | NA | NA | 82.2 ± 8.2 | 72.3 ± 12.0 | 87.2 ± 4.6 | 82.8 ± 5.8 | ||
| 1000-grain weight (g) | NA | NA | 23.3 ± 0.5 | 23.7 ± 0.5 | 22.4 ± 0.2 | 23.4 ± 1.1 | ||
| Yield (t/hm2) | NA | NA | 5.8 ± 0.9 | 5.1 ± 0.9 | 10.5 ± 1.6 | 9.6 ± 1.8 | ||
| Characteristic | TGMS line | F2 grains of hybrid rice | |||
|---|---|---|---|---|---|
| 1892S | 31892S | 1892S/RH003 | 31892S/RH003 | ||
| Grain length (mm) | 6.3 ± 0.1 | 6.4 ± 0.1 | 6.2 ± 0.3 | 6.3 ± 0.4 | |
| Ratio of length to width | 2.9 ± 0.1 | 2.9 ± 0.1 | 2.9 ± 0.0 | 3.0 ± 0.1 | |
| Degree of chalkiness (%) | 8.3 ± 0.6 | 9.0 ± 0.9 | 16.8 ± 3.4 | 17.7 ± 4.0 | |
| Amylose content (%) | 14.8 ± 0.2 | 15.4 ± 0.8 | 22.7 ± 0.7 | 22.9 ± 0.4 | |
| Gel consistency (mm) | 87.0 ± 8.2 | 91.0 ± 7.2 | 68.5 ± 37.5 | 74.0 ± 35.4 | |
| Alkali spreading value | 4.2 ± 0.3 | 4.7 ± 0.6 | 5.3 ± 0.7 | 6.1 ± 0.1 | |
Table 3 Grain quality of 1892S, 31892S and F2 grains of hybrid rice.
| Characteristic | TGMS line | F2 grains of hybrid rice | |||
|---|---|---|---|---|---|
| 1892S | 31892S | 1892S/RH003 | 31892S/RH003 | ||
| Grain length (mm) | 6.3 ± 0.1 | 6.4 ± 0.1 | 6.2 ± 0.3 | 6.3 ± 0.4 | |
| Ratio of length to width | 2.9 ± 0.1 | 2.9 ± 0.1 | 2.9 ± 0.0 | 3.0 ± 0.1 | |
| Degree of chalkiness (%) | 8.3 ± 0.6 | 9.0 ± 0.9 | 16.8 ± 3.4 | 17.7 ± 4.0 | |
| Amylose content (%) | 14.8 ± 0.2 | 15.4 ± 0.8 | 22.7 ± 0.7 | 22.9 ± 0.4 | |
| Gel consistency (mm) | 87.0 ± 8.2 | 91.0 ± 7.2 | 68.5 ± 37.5 | 74.0 ± 35.4 | |
| Alkali spreading value | 4.2 ± 0.3 | 4.7 ± 0.6 | 5.3 ± 0.7 | 6.1 ± 0.1 | |
| [1] | Amante-Bordeos A, Sitch L A, Nelson R, Dalmacio R D, Oliva N P, Aswidinnoor H, Leung H. 1992. Transfer of bacterial blight and blast resistance from the tetraploid wild rice Oryza minuta to cultivated rice, Oryza sativa. Theor Appl Genet, 84: 345-354. |
| [2] | Brinkman M A, Frey K J. 1977. Yield-component analysis of oat isolines that produce different grain yields. Crop Sci, 17(1): 165-168. |
| [3] | Chunwongse J, Martin G B, Tanksley S D. 1993. Pre-germination genotypic screening using PCR amplification of half-seeds. Theor Appl Genet, 86(6): 694-698. |
| [4] | Cruz N D, Khush G S. 2000. Rice grain quality evaluation procedures. In: Singh R K, Singh U S, Khush G S. Aromatic Rices. New Delhi, India: Oxford and IBH Publishing Co. Pvt. Ltd: 24-36. |
| [5] | Deng H F, Shu F B, Yuan D Y. 1999. An overview of research and utilization of Annong S-1. Hybrid Rice, 14(3): 1-3. (in Chinese) |
| [6] | Duncan D B. 1955. Multiple range and multiple F tests. Biometrics, 11: 1-41. |
| [7] | Fukao T, Yeung E, Bailey-Serres J. 2011. The submergence tolerance regulator SUB1A mediates crosstalk between submergence and drought tolerance in rice. Plant Cell, 23(1): 412-427. |
| [8] | Gnanamanickam S S, Priyadarisini V B, Narayanan N N, Vasudevan P, Kavitha S. 1999. An overview of bacterial blight disease of rice and strategies for its management. Curr Sci, 77(11): 1435-1444. |
| [9] | Huang N, Angeles E R, Domingo J, Magpantay G, Singh S, Zhang G, Kumaravadivel N, Bennett J, Khush G S. 1997. Pyramiding of bacterial blight resistance genes in rice: Marker-assisted selection using RFLP and PCR. Theor Appl Genet, 95(3): 313-320. |
| [10] | Ikeda R, Khush G S, Tabien R E. 1990. A new resistance gene to bacterial blight derived from O. longistaminata. Jpn J Breeding, 40(S1): 280-281. |
| [11] | Kauffman H E, Reddy A P K, Hsieh S P Y, Merca S D. 1973. An improved technique for evaluating resistance of rice varieties to Xanthomonas oryzae. Plant Dis Rep, 57(6): 537-541. |
| [12] | Khanna A, Sharma V, Ellur R K, Shikari A B, Gopala Krishnan S, Singh U D, Prakash G, Sharma T R, Rathour R, Variar M, Prashanthi S K, Nagarajan M, Vinod K K, Bhowmick P K, Singh N K, Prabhu K V, Singh B D, Singh A K. 2015. Development and evaluation of near-isogenic lines for major blast resistance gene(s) in Basmati rice. Theor Appl Genet, 128(7): 1243-1259. |
| [13] | Khush K S, Esperanza B, Ogawa T. 1990. A new gene for resistance to bacterial blight from O. longistaminata. Rice Genet News Lett, 7: 121-122. |
| [14] | Koide Y, Ebron L A, Kato H, Tsunematsu H, Telebanco-Yanoria M J, Kobayashi N, Yokoo M, Maruyama S, Imbe T, Fukuta Y. 2011. A set of near-isogenic lines for blast resistance genes with an indica-type rainfed lowland elite rice (Oryza sativa L.) genetic background. Field Crops Res, 123(1): 19-27. |
| [15] | Liu G, Lu G, Zeng L, Wang G L. 2002. Two broad-spectrum blast resistance genes, Pi9(t) and Pi2(t), are physically linked on rice chromosome 6. Mol Genet Genom, 267(4): 472-480. |
| [16] | Luo Y C, Ma T C, Zhang A F, Ong K H, Li Z F, Yang J B, Yin Z C. 2016. Marker-assisted breeding of the rice restorer line Wanhui 6725 for disease resistance, submergence tolerance and aromatic fragrance. Rice, 9(1): 66. |
| [17] | Luo Y C, Ma T C, Zhang A F, Ong K H, Luo Z X, Li Z F, Yang J B, Yin Z C. 2017. Marker-assisted breeding of Chinese elite rice cultivar 9311 for disease resistance to rice blast and bacterial blight and tolerance to submergence. Mol Breeding, 37(8): 106. |
| [18] | Luo Y C, Sangha J S, Wang S H, Li Z F, Yang J B, Yin Z C. 2012. Marker-assisted breeding of Xa4, Xa21 and Xa27 in the restorer lines of hybrid rice for broad-spectrum and enhanced disease resistance to bacterial blight. Mol Breeding, 30: 1601-1610. |
| [19] | Luo Y C, Yin Z C. 2013. Marker-assisted breeding of Thai fragrance rice for semi-dwarf phenotype, submergence tolerance and disease resistance to rice blast and bacterial blight. Mol Breeding, 32: 709-721. |
| [20] | Luo Y C, Zakaria S, Basyah B, Ma T C, Li Z F, Yang J B, Yin Z C. 2014. Marker-assisted breeding of Indonesia local rice variety Siputeh for semi-dwarf phonetype, good grain quality and disease resistance to bacterial blight. Rice, 7(1): 33. |
| [21] | Mackill D J, Amante M M, Vergara B S, Sarkarung S. 1993. Improved semidwarf rice lines with tolerance to submergence of seedlings. Crop Sci, 33(4): 749-753. |
| [22] | Mew T W, Alvarez A M, Leach J E, Swings J. 1993. Focus on bacterial blight of rice. Plant Dis, 77(1): 5-12. |
| [23] | Neeraja C N, Maghirang-Rodriguez R, Pamplona A, Heuer S, Collard B C Y, Septiningsih E M, Vergara G, Sanchez D, Xu K, Ismail A M, Mackill D J. 2007. A marker-assisted backcross approach for developing submergence-tolerant rice cultivars. Theor Appl Genet, 115(6): 767-776. |
| [24] | Ni D H, Song F S, Ni J L, Zhang A F, Wang C L, Zhao K J, Yang Y C, Wei P C, Yang J B, Li L. 2015. Marker-assisted selection of two-line hybrid rice for disease resistance to rice blast and bacterial blight. Field Crops Res, 184: 1-8. |
| [25] | Perez L M, Redona E D, Mendioro M S, Cruz C M V, Leung H. 2008. Introgression of Xa4, Xa7 and Xa21 for resistance to bacterial blight in thermosensitive genetic male sterile rice (Oryza sativa L.) for the development of two-line hybrids. Euphytica, 164(3): 627-636. |
| [26] | Sakthivel K, Sundaram R M, Rani N S, Balachandran S M, Neeraja C N. 2009. Genetic and molecular basis of fragrance in rice. Biotechnol Adv, 27(4): 468-473. |
| [27] | Septiningsih E M, Pamplona A M, Sanchez D L, Neeraja C N, Vergara G V, Heuer S, Ismail A M, Mackill D J. 2009. Development of submergence-tolerant rice cultivars: the Sub1 locus and beyond. Ann Bot, 103: 151-160. |
| [28] | Song F S, Ni J L, Zhang A F, Wang C L, Zhao K J, Li L, Ni D H, Yang J B. 2016. Molecular improvement of male sterile line 1892S for resistance to rice blast and bacterial blight. Mol Plant Breeding, 14(6): 1507-1515. (in Chinese with English abstract) |
| [29] | Xu J J, Wang B H, Wu Y H, Du P N, Wang J, Wang M, Yi C D, Gu M H, Liang G H. 2011. Fine mapping and candidate gene analysis of ptgms2-1, the photoperiod-thermo-sensitive genic male sterile gene in rice (Oryza sativa L.). Theor Appl Genet, 122(2): 365-372. |
| [30] | Xu K N, Mackill D J. 1996. A major locus for submergence tolerance mapped on rice chromosome 9. Mol Breeding, 2(3): 219-224. |
| [31] | Xu K N, Xu X, Fukao T, Canlas P, Maghirang-Rodriguez R, Heuer S, Ismail A M, Bailey-Serres J, Ronald P C, Mackill D J. 2006. Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature, 442: 705-708. |
| [32] | Yang L S, Bai Y S, Qian Z. 2016. Promotion and application of male sterile line 1892S breeding series resistant combination. J Anhui Agric Sci, 44(8): 63-66. (in Chinese with English abstract) |
| [33] | Yuan L P. 2014. Development of hybrid rice to ensure food security. Rice Sci, 21(1): 1-2. |
| [34] | Zhang J, Li X, Jiang G, Xu Y, He Y. 2006. Pyramiding of Xa7 and Xa21 for the improvement of disease resistance to bacterial blight in hybrid rice. Plant Breeding, 125(6): 600-605. |
| [35] | Zhou H, Zhou M, Yang Y Z, Li J, Zhu L Y, Jiang D G, Dong J F, Liu Q J, Gu L F, Zhou L Y, Feng M J, Qin P, Hu X C, Song C L, Shi J F, Song X W, Ni E D, Wu X J, Deng Q Y, Liu Z L, Chen M S, Liu Y G, Cao X F, Zhuang C X. 2014. RNase ZS1 processes UbL40 mRNAs and controls thermosensitive genic male sterility in rice. Nat Commun, 5: 4884. |
| [1] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [2] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [3] | Suhas Gorakh Karkute, Vishesh Kumar, Mohd Tasleem, Dwijesh Chandra Mishra, Krishna Kumar Chaturvedi, Anil Rai, Amitha Mithra Sevanthi, Kishor Gaikwad, Tilak Raj Sharma, Amolkumar U. Solanke. Genome-Wide Analysis of von Willebrand Factor A Gene Family in Rice for Its Role in Imparting Biotic Stress Resistance with Emphasis on Rice Blast Disease [J]. Rice Science, 2022, 29(4): 375-384. |
| [4] | Wu Zhongling, Qiu Jiehua, Shi Huanbin, Lin Chuyu, Yue Jiangnan, Liu Zhiquan, Xie Wei, Naweed I. Naqvi, Kou Yanjun, Tao Zeng. Polycomb Repressive Complex 2-Mediated H3K27 Trimethylation Is Required for Pathogenicity in Magnaporthe oryzae [J]. Rice Science, 2022, 29(4): 363-374. |
| [5] | R. Abdul Fiyaz, D. Shivani, K. Chaithanya, K. Mounika, M. Chiranjeevi, G. S. Laha, B. C. Viraktamath, L. V. Subba Rao, R. M. Sundaram. Genetic Improvement of Rice for Bacterial Blight Resistance: Present Status and Future Prospects [J]. Rice Science, 2022, 29(2): 118-132. |
| [6] | Zhou Ying, Wan Tao, Yuan Bin, Lei Fang, Chen Meijuan, Wang Qiong, Huang Ping, Kou Shuyan, Qiu Wenxiu, Liu Li. Improving Rice Blast Resistance by Mining Broad-Spectrum Resistance Genes at Pik Locus [J]. Rice Science, 2022, 29(2): 133-142. |
| [7] | Yong Yang, Qiujun Lin, Xinyu Chen, Weifang Liang, Yuwen Fu, Zhengjin Xu, Yuanhua Wu, Xuming Wang, Jie Zhou, Chulang Yu, Chengqi Yan, Qiong Mei, Jianping Chen. Characterization and Proteomic Analysis of Novel Rice Lesion Mimic Mutant with Enhanced Disease Resistance [J]. Rice Science, 2021, 28(5): 466-478. |
| [8] | Junhua Lu, Xuemei Yang, Jinfeng Chen, Tingting Li, Zijin Hu, Ying Xie, Jinlu Li, Jiqun Zhao, Mei Pu, Hui Feng, Jing Fan, Yanyan Huang, Jiwei Zhang, Wenming Wang, Yan Li. Osa-miR439 Negatively Regulates Rice Immunity Against Magnaporthe oryzae [J]. Rice Science, 2021, 28(2): 156-165. |
| [9] | Mishra Rukmini, Zheng Wei, Kumar Joshi Raj, Kaijun Zhao. Genome Editing Strategies Towards Enhancement of Rice Disease Resistance [J]. Rice Science, 2021, 28(2): 133-145. |
| [10] | Jan Mehmood, Shah Gulmeena, Yuqing Huang, Xuejiao Liu, Peng Zheng, Hao Du, Hao Chen, Jumin Tu. Development of Heat Tolerant Two-Line Hybrid Rice Restorer Line Carrying Dominant Locus of OsHTAS [J]. Rice Science, 2021, 28(1): 99-108. |
| [11] | Panda Debabrata, Barik Jijnasa. Flooding Tolerance in Rice: Focus on Mechanisms and Approaches [J]. Rice Science, 2021, 28(1): 43-57. |
| [12] | Ning Xiao, Yunyu Wu, Aihong Li. Strategy for Use of Rice Blast Resistance Genes in Rice Molecular Breeding [J]. Rice Science, 2020, 27(4): 263-277. |
| [13] | B. ANGELES-SHIM Rosalyn, P. REYES Vincent, M. del VALLE Marilyn, S. LAPIS Ruby, SHIM Junghyun, SUNOHARA Hidehiko, K. JENA Kshirod, ASHIKARI Motoyuki, DOI Kazuyuki. Marker-Assisted Introgression of Quantitative Resistance Gene pi21 Confers Broad Spectrum Resistance to Rice Blast [J]. Rice Science, 2020, 27(2): 113-123. |
| [14] | Barik Jijnasa, Kumar Vajinder, K. Lenka Sangram, Panda Debabrata. Assessment of Variation in Morpho-Physiological Traits and Genetic Diversity in Relation to Submergence Tolerance of Five Indigenous Lowland Rice Landraces [J]. Rice Science, 2020, 27(1): 32-43. |
| [15] | Ting Chen, Zheng Chen, Prakash Sathe Atul, Zhihong Zhang, Liangjian Li, Huihui Shang, Shaoqing Tang, Xiaobo Zhang, Jianli Wu. Characterization of a Novel Gain-of-Function Spotted-Leaf Mutant with Enhanced Disease Resistance in Rice [J]. Rice Science, 2019, 26(6): 372-383. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||