
Rice Science ›› 2015, Vol. 22 ›› Issue (4): 171-179.DOI: 10.1016/S1672-6308(14)60295-X
• Orginal Article • Previous Articles Next Articles
Received:2015-03-07
Accepted:2015-04-21
Online:2015-07-28
Published:2015-05-27
About author:Corresponding author:Thomas H. TAI(Thomas.Tai@ars.usda.gov)
H. Tai Thomas. Identification and Characterization of Reduced Epicuticular Wax Mutants in Rice[J]. Rice Science, 2015, 22(4): 171-179.
Add to citation manager EndNote|Ris|BibTeX
URL: http://www.ricesci.org/EN/10.1016/S1672-6308(14)60295-X
| M1 line | M2 line | Seed source | Mutagenesis | Phenotype of M3 | Single gene recessive mutation | Mutant designation |
| 6-1 | 6-1A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Nd | wsl3 |
| 6-1 | 6-1B | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy | Nd | |
| 7-17 | 7-17A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Yes | wsl4 |
| 11-39 | 11-39A | 2009 | 1 mmol/L NaN3 / 20 h presoak | Wet leaf/glossy | Nd | wsl5 |
| 26 | 26.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl6 |
| 264 | 264.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Nd | wsl7 |
| 524 | 524.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl8 |
| 680 | 680.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl9 |
| 843 | 843.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl10 |
| 1064 | 1064.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl11 |
| 1086 | 1086.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl12 |
| 1558 | 1558.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | |
| 1558 | 1558.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | wsl13 |
| Phenotypic ratios of segregating M3 populations were evaluated using Pearson’s Chi-square test for goodness-of-fit to the 3:1 (wild-type:mutant) ratio expected for single gene recessive mutations. Nd, Not determined; wsl, Wax crystal-sparse leaf. | ||||||
Table Genetic and physical characteristics of reduced epicuticular wax mutants.
| M1 line | M2 line | Seed source | Mutagenesis | Phenotype of M3 | Single gene recessive mutation | Mutant designation |
| 6-1 | 6-1A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Nd | wsl3 |
| 6-1 | 6-1B | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy | Nd | |
| 7-17 | 7-17A | 2009 | 1 mmol/L NaN3 / 16 h presoak | Wet leaf/glossy segregating | Yes | wsl4 |
| 11-39 | 11-39A | 2009 | 1 mmol/L NaN3 / 20 h presoak | Wet leaf/glossy | Nd | wsl5 |
| 26 | 26.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl6 |
| 264 | 264.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Nd | wsl7 |
| 524 | 524.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl8 |
| 680 | 680.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl9 |
| 843 | 843.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy segregating | Yes | wsl10 |
| 1064 | 1064.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl11 |
| 1086 | 1086.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Wet leaf/glossy | Nd | wsl12 |
| 1558 | 1558.1 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | |
| 1558 | 1558.2 | 2011 | 2 mmol/L NaN3 / 16-20 h presoak | Intermediate | Nd | wsl13 |
| Phenotypic ratios of segregating M3 populations were evaluated using Pearson’s Chi-square test for goodness-of-fit to the 3:1 (wild-type:mutant) ratio expected for single gene recessive mutations. Nd, Not determined; wsl, Wax crystal-sparse leaf. | ||||||

Fig. 1. Wet leaf/glossy and intermediate-type mutant phenotypes. Leaves were subjected to a fine misting with water from a spray bottle. Water droplets adhere to the leaves of mutants and coalesce resulting in a wet, glossy appearance. A, Comparison of wild-type Sabine and the three mutants 6-1A, 7-17A, and 11-39A. Booting-stage flag leaves are shown; B, Comparison of the wild-type Sabine and the mutants 1558.2 and 6-1A. Fifth fully-expanded leaves from the base are shown. Unlike the other mutants, water droplets adhere to 1558.2 but do not coalesce and the leaves do not appear wet or glossy.
| Population | M3 seeds planted a | M3 plants phenotyped a | Segregation ratio (wild-type:mutant) | Pearson’s Chi-square test (3:1) | ||
| χ2 | P value | |||||
| 26.1 | 77 | 57 | 40:17 | 0.708 | 0.400 | |
| 680.2 | 77 | 61 | 43:18 | 0.661 | 0.416 | |
| 843.1 | 77 | 73 | 53:20 | 0.224 | 0.636 | |
| 524.2 | 95 | 68 | 53:15 | 0.314 | 0.575 | |
| a includes initial screening of M3 plants. | ||||||
Table 2 Segregation of wet leaf/glossyphenotype in four M3 populations.
| Population | M3 seeds planted a | M3 plants phenotyped a | Segregation ratio (wild-type:mutant) | Pearson’s Chi-square test (3:1) | ||
| χ2 | P value | |||||
| 26.1 | 77 | 57 | 40:17 | 0.708 | 0.400 | |
| 680.2 | 77 | 61 | 43:18 | 0.661 | 0.416 | |
| 843.1 | 77 | 73 | 53:20 | 0.224 | 0.636 | |
| 524.2 | 95 | 68 | 53:15 | 0.314 | 0.575 | |
| a includes initial screening of M3 plants. | ||||||
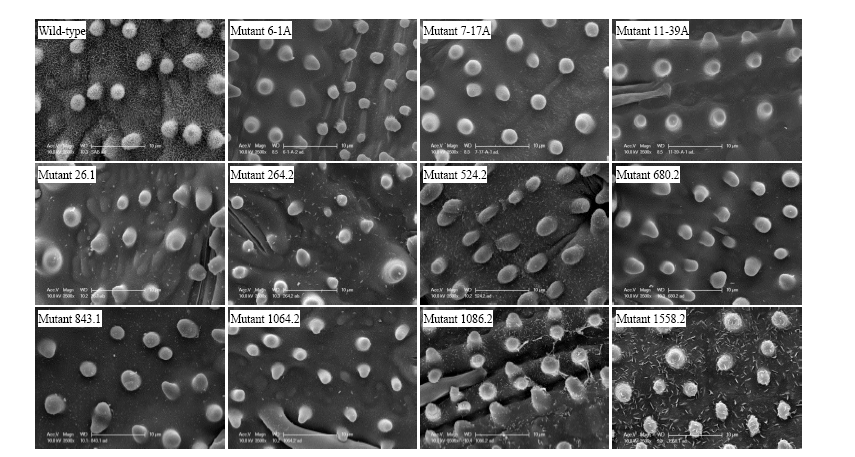
Fig. 2. Scanning electron micrographs of wild-type Sabine and 11 reduced epicuticular wax mutants.Adaxial leaf blade surface shown (knob-like structures are papillae). Scale bars, 10 μm.
| Line | Wax content (mg/g) | Reduction in wax content (%) |
| Sabine (wild-type) | 3.01 ± 0.49 | |
| 6-1A/B a | 0.91 ± 0.31 ** | 69.77 |
| 7-17A | 0.68 ± 0.42 ** | 77.41 |
| 11-39A | 1.38 ± 0.10 ** | 54.15 |
| Values are given as mean ± SD with four replicates except where noted. a, Due to insufficient tissue from 6-1A M3 mutants, additional samples were taken from 6-1B M3 mutants and content was determined using a total of five replicates (three from 6-1A plants, two from 6-1B plants included). ** means significant difference between mean values at P < 0.01 by t-test between the wild-type and each of the mutant line. | ||
Table 3 Total epicuticular wax content of wild-type and wsl mutant lines by weight method.
| Line | Wax content (mg/g) | Reduction in wax content (%) |
| Sabine (wild-type) | 3.01 ± 0.49 | |
| 6-1A/B a | 0.91 ± 0.31 ** | 69.77 |
| 7-17A | 0.68 ± 0.42 ** | 77.41 |
| 11-39A | 1.38 ± 0.10 ** | 54.15 |
| Values are given as mean ± SD with four replicates except where noted. a, Due to insufficient tissue from 6-1A M3 mutants, additional samples were taken from 6-1B M3 mutants and content was determined using a total of five replicates (three from 6-1A plants, two from 6-1B plants included). ** means significant difference between mean values at P < 0.01 by t-test between the wild-type and each of the mutant line. | ||
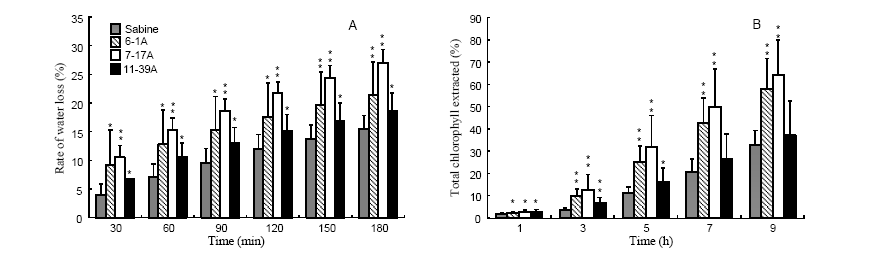
Fig. 3. Cuticle membrane permeability of three wax mutants. A, Water loss rate from detached leaves over time; B, Chlorophyll loss over time. Each bar represents the mean ± SD of two independent assays (two replicates for wild-type and four replicates for each mutant). Levels of significance between wild-type Sabine and each mutant were determined by t-test for unequal variance; * and ** mean significant difference from wild-type at P < 0.05 and P < 0.01, respectively.
| 1 | Abe A, Kosugi S, Yoshida K, Natsume S, Takagi H, Kanzaki H, Matsumura H, Yoshida K, Mitsuoka C, Tamiru M, Innan H, Cano L, Kamoun S, Terauchi R.2012. Genome sequencing reveals agronomically important loci in rice using MutMap.Nat Biotechnol, 30(2): 174-178. |
| 2 | Baker E A.1982. Chemistry and morphology of plant epicuticular waxes. In: Cutler D F, Alvin K L, Price C E. The Plant Cuticle. London: Academic Press: 139-165. |
| 3 | Clark J B, Lister G R.1975. Photosynthetic action spectra of trees: II. The relationship of cuticle structure to the visible and ultraviolet spectral properties of needles from four coniferous species.Plant Physiol, 55(2): 407-413. |
| 4 | Fiebig A, Mayfield J A, Miley N L, Chau S, Fischer R L, Preuss D.2000. Alterations in CER6, a gene identical to CUT1, differentially affect long-chain lipid content on the surface of pollen and stems.Plant Cell, 12(10): 2001-2008. |
| 5 | Islam M A, Du H, Ning J, Ye H Y, Xiong L Z.2009. Characterization of Glossy1-homologous genes in rice involved in leaf wax accumulation and drought resistance.Plant Mol Biol, 70(4): 443-456. |
| 6 | Jenks M A, Joly R J, Peters P J, Rich P J, Axtell J D, Ashworth E N.1994. Chemically induced cuticle mutation affecting epidermal conductance to water vapor and disease susceptibility in Sorghum bicolor (L.) Moench.Plant Physiol, 105(4): 1239-1245. |
| 7 | Jenks M A, Tuttle H A, Eigenbrode S D, Feldmann K A.1995. Leaf epicuticular waxes of the eceriferum mutant in Arabidopsis.Plant Physiol, 108: 369-377. |
| 8 | Jenks M A, Eigenbrode S D, Lemieux B.2002. Cuticular waxes of Arabidopsis. In: Somerville C R, Meyerowitz E M. The Arabidopsis book/American Society of Plant Biologists. |
| 9 | Jung K H, Han M J, Lee D Y, Lee Y S, Schreiber L, Franke R, Faust A, Yephremov A, Saedler H, Kim Y W, Hwang I, An G.2006. Wax-deficient anther1 is involved in cuticle and wax production in rice anther walls and is required for pollen development.Plant Cell, 18: 3015-3032. |
| 10 | Kerstiens G.1996. Cuticular water permeability and its physiological significance.J Exp Bot, 47(12): 1813-1832. |
| 11 | Lolle S J, Cheung A Y, Sussex I M.1992. Fiddlehead: An Arabidopsis mutant constitutively expressing an organ fusion program that involves interactions between epidermal cells.Dev Biol, 152: 383-392. |
| 12 | Lolle S J, Berlyn G P, Engstrom E M, Krolikowski K A, Reiter W D, Pruitt R E.1997. Developmental regulation of cell interactions in the Arabidopsis fiddlehead-1 mutant: A role for the epidermal cell wall and cuticle.Dev Biol, 189: 311-321. |
| 13 | Mao B G, Cheng Z J, Lei C L, Xu F H, Gao S W, Ren Y L, Wang J L, Zhang X, Wang J, Wu F Q, Guo X P, Liu X P, Wu C Y, Wang H Y, Wan J M.2012. Wax crystal-sparse leaf2, a rice homologue of WAX2/GL1, is involved in synthesis of leaf cuticular wax.Planta, 235: 39-52. |
| 14 | Nordström K J V, Albani M C, Velikkakam James G, Gutjahr C, Hartwig B, Turck F, Paszkowski U, Coupland G, Schneeberger K.2013. Mutation identification by direct comparison of whole-genome sequencing data from mutant and wild-type individuals using k-mers.Nat Biotechnol, 31(4): 325-330. |
| 15 | Preuss D, Lemieux B, Yen G, Davis R W.1993. A conditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signaling during fertilization.Genes Dev, 7(6): 974-985. |
| 16 | Post-Beittenmiller D.1996. Biochemistry and molecular biology of wax production in plants.Annu Rev Plant Physiol Plant Mol Biol, 47: 405-430. |
| 17 | Qin B X, Tang D, Huang J, Li M, Wu X R, Lu L L, Wang K J, Yu H X, Chen J M, Gu M H, Cheng Z K.2011. Rice OsGL1-1 is involved in leaf cuticular wax and cuticle membrane.Mol Plant, 4: 985-995. |
| 18 | Riederer M.2006. Biology of the Plant Cuticle. Oxford, Blackwell: 1-8. |
| 19 | Samuels L, Kunst L, Jetter R.2008. Sealing plant surfaces: Cutiicular wax formation by epidermal cells.Plant Biol, 59: 683-707. |
| 20 | Schneeberger K, Weigel D.2011. Fast-forward genetics enabled by new sequencing technologies.Trends Plant Sci, 16: 282-288. |
| 21 | Schneeberger K.2014. Using next-generation sequencing to isolate mutant genes from forward genetic screens.Nat Rev Genet, 15: 662-676. |
| 22 | Sieber P, Schorderet M, Ryser U, Buchala A, Kolattukudy P, Métraux J P, Nawrath C.2000. Transgenic Arabidopsis plants expressing a fungal cutinase show alterations in the structure and properties of the cuticle and postgenital organ fusions.Plant Cell, 12: 721-737. |
| 23 | Serrano M, Coluccia F, Torres M, L’Haridon F, Métraux J P.2014. The cuticle and plant defense to pathogens.Front Plant Sci, 5: 1-8. |
| 24 | Yeats T H, Rose J K C.2013. The formation and function of plant cuticles.Plant Physiol, 163: 5-20. |
| 25 | Yu D, Ranathunge K, Huang H, Pei Z, Franke R, Schreiber L, He C.2008. Wax crystal-sparse leaf1 encodes a β-ketoacyl CoA synthase involved in biosynthesis of cuticular waxes on rice leaf.Planta, 228: 675-685. |
| 26 | Zhou L Y, Ni E D, Yang J W, Zhou H, Liang H, Li J, Jiang D G, Wang Z H, Liu Z L, Zhuang C X.2013. Rice OsGL1-6 is involved in leaf cuticular wax accumulation and drought resistance.PLoS One, 8: e65139. |
| 27 | (Managing Editor: Wang Caihong) |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
