
Rice Science ›› 2017, Vol. 24 ›› Issue (4): 218-227.DOI: 10.1016/j.rsci.2017.04.002
• Orginal Article • Previous Articles Next Articles
Sung Jwakyung1, Lee Suyeon1, Chung Jong-Wook2, Edwards Gerald3, Ryu Hojin4, Kim Taewan5
Received:2017-01-23
Accepted:2017-04-05
Online:2017-07-10
Published:2017-04-28
Sung Jwakyung, Lee Suyeon, Chung Jong-Wook, Edwards Gerald, Ryu Hojin, Kim Taewan. Photosynthesis, Metabolite Composition and Anatomical Structure of Oryza sativa and Two Wild Relatives, O. grandiglumis and O. alta[J]. Rice Science, 2017, 24(4): 218-227.
Add to citation manager EndNote|Ris|BibTeX
| Species | Leaf length (cm) | Leaf width (cm) | SLA (mg/cm2) |
|---|---|---|---|
| O. grandiglumis | 60.2 b | 3.5 b | 8.7 b |
| O. alta | 67.8 a | 4.1 b | 9.8 ab |
| O. sativa (japonica) | 32.9 c | 0.7 c | 10.6 a |
| O. sativa | 29.8 c | 0.8 c | 10.6 a |
| (japonica × indica) |
Table 1 Length and width of upper fully expanded leaves of wild rice species measured at 80 d after transplanting.
| Species | Leaf length (cm) | Leaf width (cm) | SLA (mg/cm2) |
|---|---|---|---|
| O. grandiglumis | 60.2 b | 3.5 b | 8.7 b |
| O. alta | 67.8 a | 4.1 b | 9.8 ab |
| O. sativa (japonica) | 32.9 c | 0.7 c | 10.6 a |
| O. sativa | 29.8 c | 0.8 c | 10.6 a |
| (japonica × indica) |
| Species | Intracellular CO2 (μmol/mol) | A [μmol/(m2∙s)] | gs [μmol/(m2∙s)] | E [mmol/(m2∙s)] | A/E (μmol/mmol) |
|---|---|---|---|---|---|
| O. grandiglumis | 191 ± 14 c | 21.6 ± 1.1 ns | 0.14 ± 0.01 b | 7.7 ± 0.5 c | 2.82 ± 0.17 a |
| O. alta | 217 ± 4 b | 22.7 ± 1.2 ns | 0.20 ± 0.02 a | 10.3 ± 0.7 b | 2.20 ± 0.08 b |
| O. sativa (japonica) | 238 ± 14 a | 21.7 ± 1.4 ns | 0.22 ± 0.02 a | 11.7 ± 0.6 a | 1.85 ± 0.11 c |
| O. sativa (japonica × indica) | 233 ± 10 a | 21.3 ± 1.0 ns | 0.20 ± 0.02 a | 11.3 ± 0.9 ab | 1.89 ± 0.11 c |
Table 2 Intracellular CO2 concentration, CO2 assimilation rates (A), stomatal conductance (gs), transpiration rate (E) and instantaneous water use efficiency (A/E) of rice species.
| Species | Intracellular CO2 (μmol/mol) | A [μmol/(m2∙s)] | gs [μmol/(m2∙s)] | E [mmol/(m2∙s)] | A/E (μmol/mmol) |
|---|---|---|---|---|---|
| O. grandiglumis | 191 ± 14 c | 21.6 ± 1.1 ns | 0.14 ± 0.01 b | 7.7 ± 0.5 c | 2.82 ± 0.17 a |
| O. alta | 217 ± 4 b | 22.7 ± 1.2 ns | 0.20 ± 0.02 a | 10.3 ± 0.7 b | 2.20 ± 0.08 b |
| O. sativa (japonica) | 238 ± 14 a | 21.7 ± 1.4 ns | 0.22 ± 0.02 a | 11.7 ± 0.6 a | 1.85 ± 0.11 c |
| O. sativa (japonica × indica) | 233 ± 10 a | 21.3 ± 1.0 ns | 0.20 ± 0.02 a | 11.3 ± 0.9 ab | 1.89 ± 0.11 c |
| Species | Rubisco | Catalase | Chlorophyll content (mg/g) | Carbohydrate content | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| [μmol/(g∙min)] | [μmol/(g∙min)] | Chlorophyll a | Chlorophyll b | Total chlorophyll | Glucose | Fructose | Sucrose | Starch | ||
| (μmol/g) | (μmol/g) | (μmol/g) | (mg/g) | |||||||
| O. grandiglumis | 6.0 ± 1.1 ns | 396 ± 29 b | 3.51 ± 0.15 a | 1.09 ± 0.06 a | 4.60 ± 0.21 a | 9.13 ± 1.52 a | 5.58 ± 0.75 a | 86.1 ± 5.2 a | 56.0 ± 7.8 b | |
| O. alta | 7.2 ± 1.4 ns | 202 ± 9 c | 1.48 ± 0.34 b | 0.54 ± 0.12 c | 2.02 ± 0.46 c | 3.65 ± 0.82 b | 2.82 ± 0.76 b | 33.4 ± 4.9 c | 71.5 ± 3.5 a | |
| O. sativa (japonica) | 4.4 ± 1.0 ns | 385 ± 19 b | 2.94 ± 0.22 a | 0.86 ± 0.08 b | 3.80 ± 0.30 b | 1.21 ± 0.18 c | 0.86 ± 0.15 c | 54.3 ± 6.3 b | 27.5 ± 3.3 c | |
| O. sativa | 7.4 ± 2.0 ns | 459 ± 32 a | 3.12 ± 0.16 a | 1.02 ± 0.08 ab | 4.14 ± 0.23 ab | 1.79 ± 0.40 c | 1.20 ± 0.24 c | 39.8 ± 4.2 c | 25.0 ± 1.7 c | |
| (japonica × indica) | ||||||||||
Table 3 Total chlorophyll content, photosynthetic enzymes and soluble carbohydrates in the upper fully expanded leaves of rice species.
| Species | Rubisco | Catalase | Chlorophyll content (mg/g) | Carbohydrate content | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| [μmol/(g∙min)] | [μmol/(g∙min)] | Chlorophyll a | Chlorophyll b | Total chlorophyll | Glucose | Fructose | Sucrose | Starch | ||
| (μmol/g) | (μmol/g) | (μmol/g) | (mg/g) | |||||||
| O. grandiglumis | 6.0 ± 1.1 ns | 396 ± 29 b | 3.51 ± 0.15 a | 1.09 ± 0.06 a | 4.60 ± 0.21 a | 9.13 ± 1.52 a | 5.58 ± 0.75 a | 86.1 ± 5.2 a | 56.0 ± 7.8 b | |
| O. alta | 7.2 ± 1.4 ns | 202 ± 9 c | 1.48 ± 0.34 b | 0.54 ± 0.12 c | 2.02 ± 0.46 c | 3.65 ± 0.82 b | 2.82 ± 0.76 b | 33.4 ± 4.9 c | 71.5 ± 3.5 a | |
| O. sativa (japonica) | 4.4 ± 1.0 ns | 385 ± 19 b | 2.94 ± 0.22 a | 0.86 ± 0.08 b | 3.80 ± 0.30 b | 1.21 ± 0.18 c | 0.86 ± 0.15 c | 54.3 ± 6.3 b | 27.5 ± 3.3 c | |
| O. sativa | 7.4 ± 2.0 ns | 459 ± 32 a | 3.12 ± 0.16 a | 1.02 ± 0.08 ab | 4.14 ± 0.23 ab | 1.79 ± 0.40 c | 1.20 ± 0.24 c | 39.8 ± 4.2 c | 25.0 ± 1.7 c | |
| (japonica × indica) | ||||||||||
| Species | Total nitrogen (mg/g) | Mineral nitrogen | N assimilation enzyme [μmol/(g∙min)] | ||||
|---|---|---|---|---|---|---|---|
| Nitrate (μmol/g) | Nitrite (nmol/g) | NR | NiR | GS | |||
| O. grandiglumis | 27.9 ± 2.2 a | 20.0 ± 3.0 a | 147 ± 7 a | 2.66 ± 0.31 b | 0.67 ± 0.10 a | 0.41 ± 0.08 a | |
| O. alta | 15.3 ± 0.2 b | 22.7 ± 1.5 a | 150 ± 29 a | 4.12 ± 0.38 a | 0.79 ± 0.21 a | 0.38 ± 0.05 a | |
| O. sativa (japonica) | 28.3 ± 1.8 a | 12.0 ± 1.0 b | 141 ± 10 a | 2.82 ± 0.15 b | 0.88 ± 0.04 a | 0.21 ± 0.02 a | |
| O. sativa (japonica × indica) | 27.6 ± 1.0 a | 13.3 ± 1.2 b | 141 ± 5 a | 2.18 ± 0.55 b | 0.68 ± 0.08 a | 0.21 ± 0.02 a | |
Table 4 Total nitrogen, nitrate and nitrite contents, and nitrate assimilation enzymes in the upper fully expanded leaves of rice species.
| Species | Total nitrogen (mg/g) | Mineral nitrogen | N assimilation enzyme [μmol/(g∙min)] | ||||
|---|---|---|---|---|---|---|---|
| Nitrate (μmol/g) | Nitrite (nmol/g) | NR | NiR | GS | |||
| O. grandiglumis | 27.9 ± 2.2 a | 20.0 ± 3.0 a | 147 ± 7 a | 2.66 ± 0.31 b | 0.67 ± 0.10 a | 0.41 ± 0.08 a | |
| O. alta | 15.3 ± 0.2 b | 22.7 ± 1.5 a | 150 ± 29 a | 4.12 ± 0.38 a | 0.79 ± 0.21 a | 0.38 ± 0.05 a | |
| O. sativa (japonica) | 28.3 ± 1.8 a | 12.0 ± 1.0 b | 141 ± 10 a | 2.82 ± 0.15 b | 0.88 ± 0.04 a | 0.21 ± 0.02 a | |
| O. sativa (japonica × indica) | 27.6 ± 1.0 a | 13.3 ± 1.2 b | 141 ± 5 a | 2.18 ± 0.55 b | 0.68 ± 0.08 a | 0.21 ± 0.02 a | |
| Amino acid | O. grandiglumis | O. alta | O. sativa (japonica) | O. sativa (japonica × indica) |
|---|---|---|---|---|
| Glutamate | 1 433 ± 53 b | 1 449 ± 72 b | 1 680 ± 67 b | 2 218 ± 271 a |
| Aspartate | 729 ± 20 b | 336 ± 38 c | 332 ± 14 c | 1 316 ± 157 a |
| Alanine | 589 ± 23 a | 436 ± 25 b | 561 ± 21 a | 172 ± 21 c |
| Serine | 159 ± 15 c | 133 ± 7 c | 423 ± 14 b | 604 ± 75 a |
| Histidine | 65 ± 22 b | 51 ± 3 b | 42 ± 7 b | 106 ± 17 a |
| Glycine | 122 ± 23 b | 91 ± 4 b | 416 ± 21 a | 117 ± 19 b |
| Threonine | 215 ± 16 b | 143 ± 6 c | 254 ± 16 b | 396 ± 49 a |
| Arginine | 298 ± 37 ab | 230 ± 24 b | 235 ± 15 b | 339 ± 50 a |
| Tyrosine | 52 ± 8 bc | 39 ± 8 c | 63 ± 2 b | 124 ± 14 a |
| Valine | 199 ± 31 b | 101 ± 2 c | 165 ± 12 bc | 347 ± 42 a |
| Methionine | 24 ± 4 ab | 11 ± 1 b | 31 ± 7 a | 14 ± 3 b |
| Phenylalanine | 94 ± 12 b | 55 ± 2 c | 95 ± 4 b | 286 ± 26 a |
| Isoleucine | 92 ± 11 b | 57 ± 1 b | 97 ± 4 b | 299 ± 29 a |
| Leucine | 81 ± 14 b | 31 ± 4 c | 93 ± 2 b | 265 ± 30 a |
| Lysine | 339 ± 67 a | 223 ± 30 a | 246 ± 12 a | 333 ± 58 a |
| Proline | 186 ± 7 b | 128 ± 17 c | 275 ± 16 a | 239 ± 31 ab |
| Total | 4 675 ± 334 b | 3 513 ± 132 c | 5 007 ± 163 b | 7 176 ± 819 a |
Table 5 Amino acid composition in the upper fully expanded leaves of rice species. μmol/g
| Amino acid | O. grandiglumis | O. alta | O. sativa (japonica) | O. sativa (japonica × indica) |
|---|---|---|---|---|
| Glutamate | 1 433 ± 53 b | 1 449 ± 72 b | 1 680 ± 67 b | 2 218 ± 271 a |
| Aspartate | 729 ± 20 b | 336 ± 38 c | 332 ± 14 c | 1 316 ± 157 a |
| Alanine | 589 ± 23 a | 436 ± 25 b | 561 ± 21 a | 172 ± 21 c |
| Serine | 159 ± 15 c | 133 ± 7 c | 423 ± 14 b | 604 ± 75 a |
| Histidine | 65 ± 22 b | 51 ± 3 b | 42 ± 7 b | 106 ± 17 a |
| Glycine | 122 ± 23 b | 91 ± 4 b | 416 ± 21 a | 117 ± 19 b |
| Threonine | 215 ± 16 b | 143 ± 6 c | 254 ± 16 b | 396 ± 49 a |
| Arginine | 298 ± 37 ab | 230 ± 24 b | 235 ± 15 b | 339 ± 50 a |
| Tyrosine | 52 ± 8 bc | 39 ± 8 c | 63 ± 2 b | 124 ± 14 a |
| Valine | 199 ± 31 b | 101 ± 2 c | 165 ± 12 bc | 347 ± 42 a |
| Methionine | 24 ± 4 ab | 11 ± 1 b | 31 ± 7 a | 14 ± 3 b |
| Phenylalanine | 94 ± 12 b | 55 ± 2 c | 95 ± 4 b | 286 ± 26 a |
| Isoleucine | 92 ± 11 b | 57 ± 1 b | 97 ± 4 b | 299 ± 29 a |
| Leucine | 81 ± 14 b | 31 ± 4 c | 93 ± 2 b | 265 ± 30 a |
| Lysine | 339 ± 67 a | 223 ± 30 a | 246 ± 12 a | 333 ± 58 a |
| Proline | 186 ± 7 b | 128 ± 17 c | 275 ± 16 a | 239 ± 31 ab |
| Total | 4 675 ± 334 b | 3 513 ± 132 c | 5 007 ± 163 b | 7 176 ± 819 a |
| Species | Oxalate | Malate | Citrate | Succinate | Acetate | Total |
|---|---|---|---|---|---|---|
| O. grandiglumis | 19.9 ± 0.5 b | 3.2 ± 0.5 b | 50.0 ± 2.4 b | 2.4 ± 0.1 b | 4.8 ± 1.3 a | 80.3 ± 2.3 b |
| O. alta | 13.5 ± 0.4 c | 8.8 ± 0.2 a | 50.8 ± 0.1 b | 3.9 ± 0.3 a | 4.6 ± 0.5 a | 81.6 ± 0.3 b |
| O. sativa (japonica) | 27.3 ± 0.6 a | 1.7 ± 0.4 c | 112.4 ± 0.8 a | 1.4 ± 0.2 c | 2.8 ± 1.0 b | 145.8 ± 0.5 a |
| O. sativa (japonica × indica) | 21.5 ± 2.6 b | 1.9 ± 0.3 c | 11.8 ± 1.4 c | 0.3 ± 0.0 d | 1.9 ± 0.5 c | 37.6 ± 4.6 c |
Table 6 Organic acid composition in the upper fully expanded leaves of rice species.
| Species | Oxalate | Malate | Citrate | Succinate | Acetate | Total |
|---|---|---|---|---|---|---|
| O. grandiglumis | 19.9 ± 0.5 b | 3.2 ± 0.5 b | 50.0 ± 2.4 b | 2.4 ± 0.1 b | 4.8 ± 1.3 a | 80.3 ± 2.3 b |
| O. alta | 13.5 ± 0.4 c | 8.8 ± 0.2 a | 50.8 ± 0.1 b | 3.9 ± 0.3 a | 4.6 ± 0.5 a | 81.6 ± 0.3 b |
| O. sativa (japonica) | 27.3 ± 0.6 a | 1.7 ± 0.4 c | 112.4 ± 0.8 a | 1.4 ± 0.2 c | 2.8 ± 1.0 b | 145.8 ± 0.5 a |
| O. sativa (japonica × indica) | 21.5 ± 2.6 b | 1.9 ± 0.3 c | 11.8 ± 1.4 c | 0.3 ± 0.0 d | 1.9 ± 0.5 c | 37.6 ± 4.6 c |
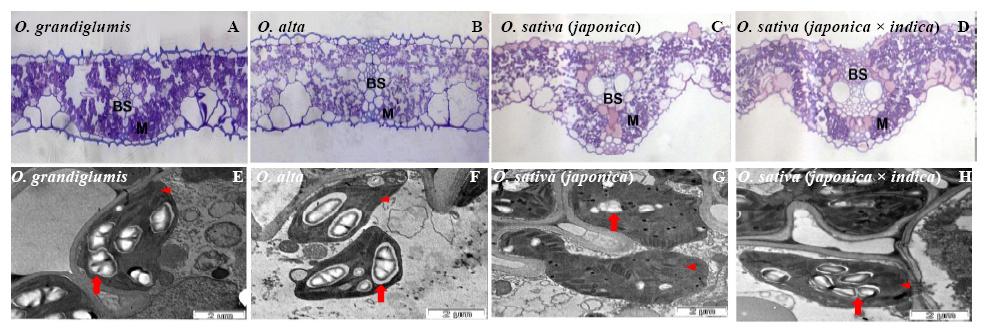
Fig. 2. Structural features of fully expanded leaves of wild and cultivated rice species observed by a photo-microscope (A-D) and transmission electron microscopy (E-H). M, Mesophyll; BS, Bundle sheath. Arrowheads highlight chloroplasts and arrows mark starch granules in chloroplasts.
| 1 | Aebi H.1974. Catalases. In: Bergmeyer H U. Methods of Enzymatic Analysis. New York: Academic Press: 673-684. |
| 2 | Azevedo R A, Arruda P, Turner W L, Lea P J.1997. The biosynthesis and metabolism of the aspartate derived amino acids in higher plants. Phytochemistry, 46(3): 395-419. |
| 3 | Bertheloot J, Martr P, Andrieu B.2008. Dynamics of light and nitrogen distribution during grain filling within wheat canopy.Plant Physiol, 148(3): 1707-1720. |
| 4 | Bradford M M.1976. A rapid and sensitive method for quantitation of microgram quantities of proteins using the principle of protein-dye binding.Anal Biochem, 72: 248-254. |
| 5 | Brar D S, Khush G S.2002. Transferring genes from wild species into rice. In: Quantitative Genetics Genomics and Plant Breeding. New York, USA: CAB International: 197-217. |
| 6 | Bykova N V, Keerberg O, Pärnik T, Bauwe H, Gardeström P.2005. Interaction between photorespiration and respiration in transgenic potato plants with antisense reduction in glycine decarboxylase.Planta, 222(1): 130-140. |
| 7 | Cataldo D A, Maroon M, Schrader L E, Youngs V L.1975. Rapid colorimetric determination of nitrate in plant tissue by nitration of salycilic acid.Comm Soil Sci Plant Anal, 6: 71-80. |
| 8 | Dawe D.2007. Agricultural research, poverty alleviation and key trends in Asia’s rice economy. In: Sheehy J E, Mitchell P L, Hardy B. Charting New Pathways to C4 Rice. Manila, the Philippines: International Rice Research Institute: 37-53. |
| 9 | Foyer C H, Valadier M H, Migge A, Becker T W.1998. Drought-induced effects on nitrate reductase activity and mRNA and on the coordination of nitrogen and carbon metabolism in maize leaves.Plant Physiol, 117(1): 283-292. |
| 10 | Gibson K, Park J S, Nagai Y, Hwang S K, Cho Y C, Roh K H, Lee S M, Kim D H, Choi S B, Ito H, Edwards G E, Okita T W.2011. Exploiting leaf starch synthesis as a transient sink to elevate photosynthesis, plant productivity and yields.Plant Sci, 181(3): 275-281. |
| 11 | Guicherd P, Peltier J P, Gout E, Bligny R, Marigo G.1997. Osmotic adjustment in Fraxinus excelsior L.: Malate and mannitol accumulation in leaves under drought conditions.Trees, 11(3): 155-161. |
| 12 | Guo L Q, Shi D C, Wang D L.2010. The key physiological response to alkali stress by the alkali-resistant halophyte Puccinellia tenuiflora is the accumulation of large quantities of organic acids and into the rhizosphere.J Agron Crop Sci, 196(2): 123-135. |
| 13 | Heinrichs E A, Medrano F G, Rapusas H R.1985. Genetic Evaluation for Insect Resistance in Rice. Manila, the Philippines: International Rice Research Institute. |
| 14 | Keys A J.2006. The re-assimilation of ammonia produced by photorespiration and the nitrogen economy of C3 higher plants.Photosynth Res, 87: 165-175. |
| 15 | Lichtenthaler H K, Buschmann C.2001. Chlorophylls and carotenoids: Measurement and characterization by UV-VIS. In: Lichtenthaler H K. Current Protocols in Food Analytical Chemistry. New York: Wiley. |
| 16 | Long S P.1999. Environmental Responses in C4 Plant Biology. San Diego, USA: Academic Press: 215-249. |
| 17 | Meister A.1985. Glutamine synthetase from mammalian tissues.Methods Enzymol, 113: 185-199. |
| 18 | Melzer E, O’Leary M H.1987. Anapleurotic CO2 fixation by phosphoenolpyruvate carboxylase in C3 plants.Plant Physiol, 84(1): 58-60. |
| 19 | Merlo L, Ferretti M, Passera C, Ghisi R.1995. Light-modulation of nitrate reductase activity in leaves and roots of maize.Physiol Plant, 2: 305-311. |
| 20 | Morcuende R, Kostadinova S, Perez P, Martin del Molino I M, Martinez-Carrasco R.2004. Nitrate is a negative signal for fructan synthesis, and the fructosyltransferase-inducing trehalose inhibits nitrogen and carbon assimilation in excised barley leaves.New Phytol, 161(3): 749-759. |
| 21 | Rachmilevitch S, Cousins A B, Bloom A J.2004. Nitrate assimilation in plant shoots depends on photorespiration.Proc Natl Acad Sci USA, 101: 11506-11510. |
| 22 | Roe J H.1955. The determination of sugar in blood and spinal fluid with anthrone reagent.J Biol Chem, 212: 335-343. |
| 23 | Rufty Jr T W, Huber S C, Volk R J.1988. Alterations in leaf carbohydrate metabolism in response to nitrogen stress.Plant Physiol, 88(3): 725-730. |
| 24 | Ruuska S A, Lewis D C, Kennedy G, Furbank R T, Jenkins C L D, Tabe L M.2008. Large scale transcriptome analysis of the effects of nitrogen nutrition on accumulation of stem carbohydrate reserves in reproductive stage wheat.Plant Mol Biol, 66(1): 15-32. |
| 25 | Sage R F.2001. Environmental and evolutionary preconditions for the origin and diversification of the C4 photosynthetic syndrome.Plant Biol, 3: 202-213. |
| 26 | Saglam A, Terzi R, Nar H, Saruhan N, Ayaz F A, Kadioglu A.2010. Inorganic and organic solutes in apoplastic and symplastic spaces contribute to osmotic adjustment during leaf rolling.Acta Biol Cracov Ser Bot, 52: 37-44. |
| 27 | Sharkey T D, Savitch L V, Butz N D.1991. Photometric method for routine determination of Kcat and carbamylation of Rubisco.Photosynth Res, 28(1): 41-48. |
| 28 | Stitt M, Muller C, Matt P, Gibon Y, Carillo P, Morcuende R, Scheible W R, Krapp A.2002. Steps towards an integrated view of nitrogen metabolism.J Exp Bot, 53: 959-970. |
| 29 | Vaughan D A.1989. The Genus Oryza L. Current Status of Taxonomy. Manila, the Philippines: International Rice Research Institute. |
| 30 | Vaughan D A.1994. The Wild Relatives of Rice: A Genetic Resources Handbook. Manila, the Philippines: International Rice Research Institute. |
| 31 | Vaughan D A, Morishima H, Kadowaki K.2003. Diversity in the Oryza genus.Curr Opin Plant Biol, 6(2): 139-146. |
| 32 | Walinga I, van Vank W, Houba V J G, van der Lee J J.1989. Soil and Plant Analysis: Part 7. Plant Analysis Procedures. Wageningen, the Netherlands: Wageningen Agriucltural University: 264. |
| 33 | Wang C, van den Ende W, Tillberg J E.2000. Fructan accumulation induced by nitrogen deficiency in barley leaves correlates with the level of sucrose:fructan 6-fructosyltransferase mRNA.Planta, 211(5): 701-707. |
| 34 | Weber A, Flügge U F.2002. Interaction of cytosolic and plastidic nitrogen metabolism in plants.J Exp Bot, 53: 865-874. |
| 35 | Woo K C, Osmond C B.1982. Stimulation of ammonia and 2-oxoglutarate-dependent oxygen evolution in isolated chloroplasts by dicarboxylates and the role of the chloroplast in photorespiratory nitrogen cycling.Plant Physiol, 69(3): 591-596. |
| 36 | Yeo M E, Yeo A R, Flowers T J.1994. Photosynthesis and photorespiration in the genus Oryza. J Exp Bot, 45(5): 553-560. |
| 37 | Yu G J, Kwak T S, Kang K H, Moon H P.1997. Efficiency of backcrossing and ovule culture in an interspecific cross between O. sativa and O. grandiglumis.Prod Kor J Breeding, 29: 448-452. |
| 38 | Zhao M, Lafitte H R, Sacks E, Dimayuga G, Acuna T L B.2008. Perennial O. sativa × O. rufipogon interspecific hybrids: I. Photosynthethetic characteristics and their inheritance.Field Crops Res, 106(3): 203-213. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||