
Rice Science ›› 2019, Vol. 26 ›› Issue (5): 300-308.DOI: 10.1016/j.rsci.2019.08.004
• Research Papers • Previous Articles Next Articles
Qiong Peng1,2,3, Heping Han3, Xia Yang1,4, Lianyang Bai1,2, Qin Yu3( ), B. Powles Stephen3
), B. Powles Stephen3
Received:2018-07-27
Accepted:2018-12-09
Online:2019-09-28
Published:2019-05-24
Qiong Peng, Heping Han, Xia Yang, Lianyang Bai, Qin Yu, B. Powles Stephen. Quinclorac Resistance in Echinochloa crus-galli from China[J]. Rice Science, 2019, 26(5): 300-308.
Add to citation manager EndNote|Ris|BibTeX
| Primer | Direction | Sequence (5′→3′) |
|---|---|---|
| Tubulin | Forward | GTACCTTGAACTATTCGACTTTGC |
| Reverse | GGCGTACATCAGGTCGAACTT | |
| EcActin | Forward | CACACTGGTGTCATGGTAGG |
| Reverse | AGAAAGTGTGATGCCAGAT | |
| EcCAS | Forward | GAACAAGGTCACAGAGGGATG |
| Reverse | CCCTTGTTTTCAGCATCTTCC | |
| EcACS | Forward | AACTGTCGCTTGACCTGATC |
| Reverse | CCATCGCCTTTCTGAACTCC | |
| EcACO1 | Forward | CTGACCAAGGACCACTACAAG |
| Reverse | TCTCCCAGTCCAGGTTCTC | |
| EcACO2 | Forward | GTGATCGACTTCTCCAAGCTC |
| Reverse | AAGCTGAAAGAATCCCCACTC |
Table 1 Real-time PCR primer sequences.
| Primer | Direction | Sequence (5′→3′) |
|---|---|---|
| Tubulin | Forward | GTACCTTGAACTATTCGACTTTGC |
| Reverse | GGCGTACATCAGGTCGAACTT | |
| EcActin | Forward | CACACTGGTGTCATGGTAGG |
| Reverse | AGAAAGTGTGATGCCAGAT | |
| EcCAS | Forward | GAACAAGGTCACAGAGGGATG |
| Reverse | CCCTTGTTTTCAGCATCTTCC | |
| EcACS | Forward | AACTGTCGCTTGACCTGATC |
| Reverse | CCATCGCCTTTCTGAACTCC | |
| EcACO1 | Forward | CTGACCAAGGACCACTACAAG |
| Reverse | TCTCCCAGTCCAGGTTCTC | |
| EcACO2 | Forward | GTGATCGACTTCTCCAAGCTC |
| Reverse | AAGCTGAAAGAATCCCCACTC |
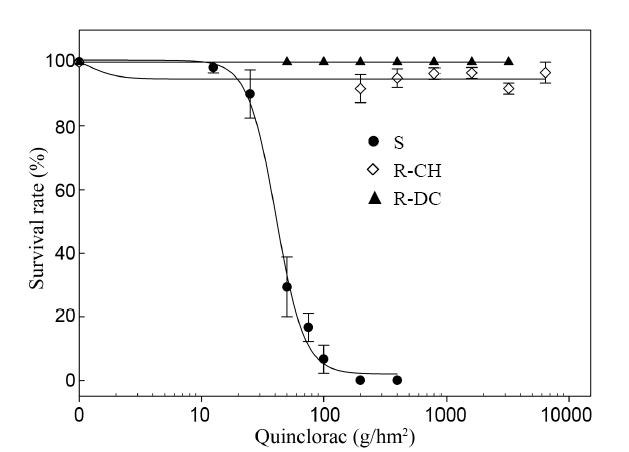
Fig. 1. Dose response to quinclorac of E. crus-galli populations at 3 weeks after treatment.‘S’ indicates the susceptible population and ‘R-DC’ and ‘R-CH’ indicate two resistant populations.
| Herbicide | Mode of action | Dose | Survival rate (%) | ||
|---|---|---|---|---|---|
| (g/hm2) | S | R-DC | R-CH | ||
| Penoxsulam | ALS inhibitor | 30 | 0 | 0 | 0 |
| Bispyribac-sodium | ALS inhibitor | 30 | 0 | 0 | 0 |
| Pyrazosulfuron-ethyl | ALS inhibitor | 120 | 5 | 52 ± 11 | 75 ± 10 |
| Imazamox | ALS inhibitor | 35 | 0 | 0 | 0 |
| Cyhalofop-butyl | ACCase inhibitor | 125 | 0 | 0 | 0 |
Table 2 Survival rate of E. crus-galli populations treated with herbicides.
| Herbicide | Mode of action | Dose | Survival rate (%) | ||
|---|---|---|---|---|---|
| (g/hm2) | S | R-DC | R-CH | ||
| Penoxsulam | ALS inhibitor | 30 | 0 | 0 | 0 |
| Bispyribac-sodium | ALS inhibitor | 30 | 0 | 0 | 0 |
| Pyrazosulfuron-ethyl | ALS inhibitor | 120 | 5 | 52 ± 11 | 75 ± 10 |
| Imazamox | ALS inhibitor | 35 | 0 | 0 | 0 |
| Cyhalofop-butyl | ACCase inhibitor | 125 | 0 | 0 | 0 |
| Time point (h) | Population | Foliar uptake | Translocation (%) | ||
|---|---|---|---|---|---|
| (%) | Root | Stem and new growth | Treated leaf | ||
| 6 | S | 96.5 ± 0.6 | 2.6 ± 0.4 b | 50.5 ± 1.3 a | 47.0 ± 1.3 b |
| R-DC | 97.6 ± 0.0 | 2.8 ± 0.8 b | 43.8 ± 1.6 b | 53.4 ± 1.6 a | |
| R-CH | 97.1 ± 0.3 | 5.8 ± 0.9 a | 51.4 ± 1.6 a | 42.8 ± 1.2 b | |
| 24 | S | 97.1 ± 0.0 | 4.2 ± 0.7 b | 54.3 ± 2.7 b | 41.5 ± 3.2 a |
| R-DC | 97.0 ± 0.2 | 4.1 ± 1.4 b | 74.1 ± 2.1 a | 21.9 ± 1.3 b | |
| R-CH | 96.9 ± 0.2 | 8.8 ± 0.9 a | 71.4 ± 0.9 a | 19.8 ± 0.7 b | |
| 48 | S | 96.2 ± 0.6 | 3.0 ± 0.8 a | 73.5 ± 2.2 b | 23.6 ± 2.7 a |
| R-DC | 96.5 ± 0.1 | 2.3 ± 0.5 a | 79.0 ± 1.3 a | 18.8 ± 0.9 ab | |
| R-CH | 96.4 ± 0.3 | 2.2 ± 0.3 a | 81.0 ± 0.7 a | 16.8 ± 0.6 b | |
Table 3 Foliar uptake and translocation of absorbed 14C in E. crus-galli populations.
| Time point (h) | Population | Foliar uptake | Translocation (%) | ||
|---|---|---|---|---|---|
| (%) | Root | Stem and new growth | Treated leaf | ||
| 6 | S | 96.5 ± 0.6 | 2.6 ± 0.4 b | 50.5 ± 1.3 a | 47.0 ± 1.3 b |
| R-DC | 97.6 ± 0.0 | 2.8 ± 0.8 b | 43.8 ± 1.6 b | 53.4 ± 1.6 a | |
| R-CH | 97.1 ± 0.3 | 5.8 ± 0.9 a | 51.4 ± 1.6 a | 42.8 ± 1.2 b | |
| 24 | S | 97.1 ± 0.0 | 4.2 ± 0.7 b | 54.3 ± 2.7 b | 41.5 ± 3.2 a |
| R-DC | 97.0 ± 0.2 | 4.1 ± 1.4 b | 74.1 ± 2.1 a | 21.9 ± 1.3 b | |
| R-CH | 96.9 ± 0.2 | 8.8 ± 0.9 a | 71.4 ± 0.9 a | 19.8 ± 0.7 b | |
| 48 | S | 96.2 ± 0.6 | 3.0 ± 0.8 a | 73.5 ± 2.2 b | 23.6 ± 2.7 a |
| R-DC | 96.5 ± 0.1 | 2.3 ± 0.5 a | 79.0 ± 1.3 a | 18.8 ± 0.9 ab | |
| R-CH | 96.4 ± 0.3 | 2.2 ± 0.3 a | 81.0 ± 0.7 a | 16.8 ± 0.6 b | |
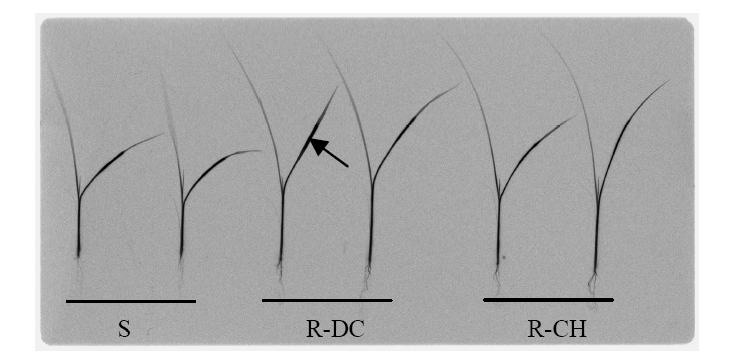
Fig. 2. Phosphor imaging comparing translocation pattern of 14C-quinclorac in E. crus-galli populations at 6 h after quinclorac treatment.14C-quinclorac was applied to the midpoint of the adaxial surface of the second fully expanded leaf (arrowed) of each plant.‘S’ indicates the susceptible population and ‘R-DC’ and ‘R-CH’ indicate two resistant populations.
| Time point (h) | Population | Radiolabel (%) | |
|---|---|---|---|
| Quinclorac | Total metabolites | ||
| 6 | S | 84.7 ± 2.3 a | 15.4 ± 2.2 a |
| R-CH | 86.9 ± 0.7 a | 13.1 ± 0.7 a | |
| 24 | S | 74.0 ± 2.6 b | 26.0 ± 2.6 a |
| R-CH | 87.2 ± 0.4 a | 12.8 ± 0.7 b | |
| Rice | 93.8 ± 0.6 | 6.2 ± 0.6 | |
| 48 | S | 77.7 ± 0.8 b | 22.3 ± 0.8 a |
| R-CH | 85.9 ± 0.9 a | 14.1 ± 0.9 b | |
Table 4 Metabolism of 14C-quinclorac in E. crus-galli populations.
| Time point (h) | Population | Radiolabel (%) | |
|---|---|---|---|
| Quinclorac | Total metabolites | ||
| 6 | S | 84.7 ± 2.3 a | 15.4 ± 2.2 a |
| R-CH | 86.9 ± 0.7 a | 13.1 ± 0.7 a | |
| 24 | S | 74.0 ± 2.6 b | 26.0 ± 2.6 a |
| R-CH | 87.2 ± 0.4 a | 12.8 ± 0.7 b | |
| Rice | 93.8 ± 0.6 | 6.2 ± 0.6 | |
| 48 | S | 77.7 ± 0.8 b | 22.3 ± 0.8 a |
| R-CH | 85.9 ± 0.9 a | 14.1 ± 0.9 b | |
| Population | β-CAS gene expression | |
|---|---|---|
| Untreated | Treated | |
| S | 0.256 ± 0.005 a | 0.156 ± 0.011 a |
| R-DC | 0.240 ± 0.022 a | 0.199 ± 0.011 a |
| R-CH | 0.192 ± 0.006 b | 0.196 ± 0.021 a |
Table 5 Relative β-CAS expression in the shoot of E. crus-galli populations at 6 h after treatment.
| Population | β-CAS gene expression | |
|---|---|---|
| Untreated | Treated | |
| S | 0.256 ± 0.005 a | 0.156 ± 0.011 a |
| R-DC | 0.240 ± 0.022 a | 0.199 ± 0.011 a |
| R-CH | 0.192 ± 0.006 b | 0.196 ± 0.021 a |
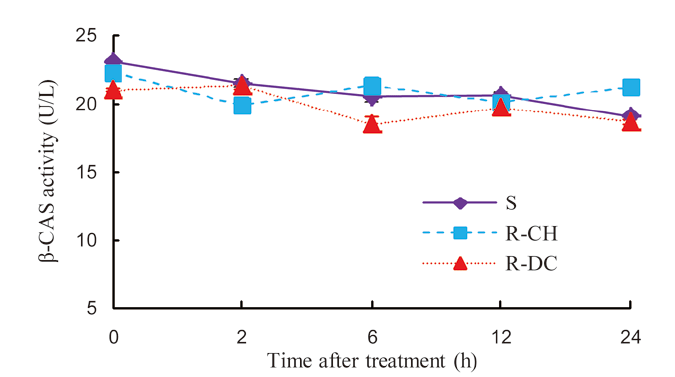
Fig. 3. Activity of β-CAS in the shoot of E. crus-galli populations treated with 50 g/hm2 quinclorac.‘S’ indicates the susceptible population and ‘R-DC’ and ‘R-CH’ indicate two resistant populations.
| Population | ACS | ACO1 | ACO2 | |||||
|---|---|---|---|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | Untreated | Treated | |||
| S | 0.019 ± 0.004 b | 0.022 ± 0.002 a | 0.034 ± 0.003 a | 0.075 ± 0.008 a | 0.218 ± 0.013 b | 0.524 ± 0.051 a | ||
| R-DC | 0.034 ± 0.004 a | 0.024 ± 0.002 a | 0.037 ± 0.001 a | 0.042 ± 0.005 b | 0.591 ± 0.065 a | 0.411 ± 0.007 ab | ||
| R-CH | 0.023 ± 0.002 b | 0.024 ± 0.002 a | 0.043 ± 0.008 a | 0.043 ± 0.008 b | 0.263 ± 0.035 b | 0.378 ± 0.059 b | ||
Table 6 Relative expression levels of ACS, ACO1 and ACO2 in the shoots of E. crus-galli populations at 6 h after quinclorac treatment.
| Population | ACS | ACO1 | ACO2 | |||||
|---|---|---|---|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | Untreated | Treated | |||
| S | 0.019 ± 0.004 b | 0.022 ± 0.002 a | 0.034 ± 0.003 a | 0.075 ± 0.008 a | 0.218 ± 0.013 b | 0.524 ± 0.051 a | ||
| R-DC | 0.034 ± 0.004 a | 0.024 ± 0.002 a | 0.037 ± 0.001 a | 0.042 ± 0.005 b | 0.591 ± 0.065 a | 0.411 ± 0.007 ab | ||
| R-CH | 0.023 ± 0.002 b | 0.024 ± 0.002 a | 0.043 ± 0.008 a | 0.043 ± 0.008 b | 0.263 ± 0.035 b | 0.378 ± 0.059 b | ||
| [1] | Bajwa A A, Jabran K, Shahid M, Ali H H, Chauhan B S, Ehsanullah.2015. Eco-biology and management ofEchinochloa crus-galli. Crop Prot, 75: 151-162. |
| [2] | Chayapakdee P, Iwakami S, Kamidate Y, Uchino A, Fan L, Sunohara S, Matsumoto H.2017. Enhanced activity of β-cyanoalanine synthase does not confer quinclorac resistance in multiple-herbicide resistant Echinochloa phyllopogon. The 26th Asian-Pacific Weed Science Society Conference, Weed Science for People, Agriculture and Nature. Kyoto, Japan. |
| [3] | Chen G Q, Wang Q, Yao Z W, Zhu L F, Dong L Y.2016. Penoxsulam-resistant barnyardgrass (Echinochloa crus-galli) in rice fields in China. Weed Biol Manag, 16(1): 16-23. |
| [4] | Chen J Y, Yu Q, Owen M, Han H P, Powles S.2018. Dinitroaniline herbicide resistance in a multiple-resistant Lolium rigidum population. Pest Manag Sci, 74: 925-932. |
| [5] | Chen T, Zhang S L, Zhao L, Zhang Y D, Zhu Z, Zhao Q Y, Zhou L H, Yao S, Zhao C F, Liang W H, Wang C L.2018. Development and verification of a functional marker associated with resistance to ALS inhibitor herbicide.Chin J Rice Sci, 32(2): 137-145. (in Chinese with English abstract) |
| [6] | Christoffoletti P J, de Figueiredo M R A, Peres L E P, Nissen S, Gaines T.2015. Auxinic herbicides, mechanisms of action, and weed resistance: A look into recent plant science advances. Sci Agric, 72(4): 356-362. |
| [7] | Fipke M V, Vidal R A.2016. Integrative theory of the mode of action of quinclorac: Literature review.Planta Dan, 34(2): 393-402. |
| [8] | Gao Y, Pan L, Sun Y, Zhang T, Dong L Y, Li J.2017. Resistance to quinclorac caused by the enhanced ability to detoxify cyanide and its molecular mechanism in Echinochloa crus-galli var. zelayensis. Pestic Biochem Physiol, 143: 231-238. |
| [9] | Gao Y, Li J, Pan X K, Liu D R, Napier R, Dong L Y.2018. Quinclorac resistance induced by the suppression of the expression of 1-aminocyclopropane-1-carboxylic acid (ACC) synthase and ACC oxidase genes in Echinochloa crus-galli var. zelayensis. Pestic Biochem Physiol, 146: 25-32. |
| [10] | Grossmann K, Kwiatkowski J.1993. Selective induction of ethylene and cyanide biosynthesis appears to be involved in the selectivity of the herbicide quinclorac between rice and barnyard grass.J Plant Physiol, 142(6): 457-466. |
| [11] | Grossmann K, Kwiatkowski J.1995. Evidence for a causative role of cyanide, derived from ethylene biosynthesis, in the herbicide mode of action of quinclorac in barnyard grass.Pestic Biochem Physiol, 51(2): 150-160. |
| [12] | Grossmann K, Kwiatkowski J.2000. The mechanism of quinclorac selectivity in grasses.Pestic Biochem Physiol, 66(2): 83-91. |
| [13] | Grossmann K.2003. Mediation of herbicide effects by hormone interactions.J Plant Growth Regul, 22(1): 109-122. |
| [14] | Grossmann K.2010. Auxin herbicides: Current status of mechanism and mode of action.Pest Manag Sci, 66: 113-120. |
| [15] | Gleason C, Foley R C, Singh K B.2011. Mutant analysis in Arabidopsis provides insight into the molecular mode of action of the auxinic herbicide dicamba. PLoS One, 6(3): e17245. |
| [16] | Guo L B, Qiu J, Ye C Y, Jin G L, Mao L F, Zhang H Q, Yang X F, Peng Q, Wang Y Y, Jia L, Lin Z X, Li G M, Fu F, Liu C, Chen L, Shen E H, Wang W D, Chu Q J, Wu S Y, Wu S L, Xia C Y, Zhang Y F, Zhou X M, Wang L F, Wu L M, Song W J, Wang Y F, Shu Q Y, Aoki D, Yumoto E, Lou Y G, Qian Q, Yamguchi H, Yamane H, Kong C H, Timko M P, Bai L Y, Fan L J.2017. Echinochloa crus-galli genome analysis provides insight into its adaptation and invasiveness as a weed. Nat Commun, 8: 1031. |
| [17] | Heap I.2018. International survey of herbicide resistant weeds. . |
| [18] | Hoyerova K, Hosek P, Quareshy M, Li J, Klima P, Kubes M, Yemm A A, Neve P, Tripathi A, Bennett M J, Napier R M.2018. Auxin molecular field maps define AUX1 selectivity: Many auxin herbicides are not substrates.New Phytol, 217(4): 1625-1639. |
| [19] | Lamoureux G L, Rusness D G.1995. Quinclorac absorption, translocation, metabolism, and toxicity in leafy spurge (Euphorbia esula). Pestic Biochem Physiol, 53(3): 210-226. |
| [20] | LeClere S, Wu C X, Westra P, Sammons R D.2018. Cross- resistance to dicamba, 2,4-D, and fluroxypyr in Kochia scoparia is endowed by a mutation in an AUX/IAA gene. Proc Natl Acad Sci USA, 115(13): 2911-2920. |
| [21] | Li G, Xu M F, Chen L P, Cai L M, Bai L Y, Wu C X.2016. A novel EcGH3 gene with a different expression pattern in quinclorac- resistant and susceptible barnyardgrass(Echinochloa crus-galli). Plant Gene, 5: 65-70. |
| [22] | Lopez-Martinez N, de Prado R.1996. Fate of quinclorac in resistant Echinochloa crus-galli. In: Second International Weed Control Congress. Copenhagen, Denmark: Eurekamag: 535-540. |
| [23] | Lopez-Martinez N, Marshall G, de Prado R.1997. Resistance of barnyardgrass (Echinochloa crus-galli) to atrazine and quinclorac. Pest Manag Sci, 51(2): 171-175. |
| [24] | Lovelace M L, Talbert R E, Hoagland R E, Scherder E F.2007. Quinclorac absorption and translocation characteristics in quinclorac- and propanil-resistant and -susceptible barnyardgrass (Echinochloa crus-galli) biotypes. Weed Technol, 21(3): 683-687. |
| [25] | Ma G L, Liu D C, Liu X Y, Tang T, Peng Y J.2013. Resistance of barnyard grass ( Echinochloa crus-galli) to quinclorac in double-harvest rice area in Hunan Province of China. Plant Dis Pests, 4: 23-27. |
| [26] | Oerke E C, Dehne H W.2004. Safeguarding production-losses in major crops and the role of crop protection.Crop Prot, 23(4): 275-285. |
| [27] | Rouse C E, Roma-Burgos N, Norsworthy J K, Tseng T M, Starkey C E, Scott R C.2018. Echinochloa resistance to herbicides continues to increase in Arkansas rice fields. Weed Technol, 32(1): 34-44. |
| [28] | Sunohara Y, Matsumoto H.2004. Oxidative injury induced by the herbicide quinclorac onEchinochloa oryzicola vasing. and the involvement of antioxidative ability in its highly selective action in grass species. Plant Sci, 167(3): 597-606. |
| [29] | Takahashi M, Umetsu K, Oono Y, Higaki T, Blancaflor E B, Rahman A.2017. Small acidic protein 1 and SCFTIR1 ubiquitin proteasome pathway act in concert to induce 2,4-dichlorophenoxyacetic acid-mediated alteration of actin in Arabidopsis roots. Plant J, 89(5): 940-956. |
| [30] | Talbert R E, Burgos N R.2007. History and management of herbicide-resistant barnyardgrass (Echinochloa crus-galli) in Arkansas rice. Weed Technol, 21(2): 324-331. |
| [31] | van Eerd L L, Stephenson G R, Kwiatkowski J, Grossmann K, Hall J C.2005. Physiological and biochemical characterization of quinclorac resistance in a false cleavers (Galium spurium L.) biotype. J Agric Food Chem, 53(4): 1144-1151. |
| [32] | Walsh T A, Neal R, Merlo A O, Honma M, Hicks G R, Wolff K, Matsumura W, Davies J P.2006. Mutations in an auxin receptor homolog AFB5 and in SGT1b confer resistance to synthetic picolinate auxins and not to 2,4-dichlorophenoxyacetic acid or indole-3-acetic acid inArabidopsis. Plant Physiol, 142(2): 542-552. |
| [33] | Wright A A, Rodriguez-Carres M, Sasidharan R, Koski L, Peterson D G, Nandula V K, Ray J D, Bond J A, Shaw D R.2018. Multiple herbicide-resistant junglerice (Echinochloa colona): Identification of genes potentially involved in resistance through differential gene expression analysis. Weed Sci, 66(3): 1-8. |
| [34] | Xu J Y, Lv B, Wang Q, Li J, Dong L Y.2013. A resistance mechanism dependent upon the inhibition of ethylene biosynthesis.Pest Manag Sci, 69(12): 1407-1414. |
| [35] | Yang X, Yu X Y, Li Y F.2013. De novo assembly and characterization of the barnyardgrass(Echinochloa crus-galli) transcriptome using next-generation pyrosequencing. PLoS One, 8(7): e69168. |
| [36] | Yang X, Zhang Z C, Gu T, Dong M C, Peng Q, Bai L Y, Li Y F.2017. Quantitative proteomics reveals ecological fitness cost of multi-herbicide resistant barnyardgrass (Echinochloa crus-galli L.). J Proteom, 150: 160-169. |
| [37] | Yasuor H, Milan M, Eckert J W, Fischer A J.2012. Quinclorac resistance: A concerted hormonal and enzymatic effort inEchinochloa phyllopogon. Pest Manag Sci, 68(1): 108-115. |
| [38] | Zhu J W, Wang J, DiTommaso A, Zhang C X, Zheng G P, Liang W, Islam F, Yang C, Chen X X, Zhou W J.2018. Weed research status, challenges, and opportunities in China. Crop Prot, doi.org/10.1016/j.cropro.2018.02.001. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [13] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||