
Rice Science ›› 2017, Vol. 24 ›› Issue (5): 253-263.DOI: 10.1016/j.rsci.2017.04.004
• Orginal Article • Previous Articles Next Articles
Swapna Simon( ), Samban Shylaraj Korukkanvilakath
), Samban Shylaraj Korukkanvilakath
Received:2016-12-01
Accepted:2017-04-10
Online:2017-09-15
Published:2017-08-31
Swapna Simon, Samban Shylaraj Korukkanvilakath. Screening for Osmotic Stress Responses in Rice Varieties under Drought Condition[J]. Rice Science, 2017, 24(5): 253-263.
Add to citation manager EndNote|Ris|BibTeX
| Variety | Origin | Variety | Origin | Variety | Origin |
|---|---|---|---|---|---|
| Sahbhagi Dhan (CK) | IRRI, the Philippines | Revathy | RARS, India | Mattathriveni | RARS, India |
| Jyothi (CK) | RARS, India | Karishma | RARS, India | Samyuktha | RARS, India |
| Bhadra | RARS, India | Krishnannjana | RARS, India | Bhagya | RARS, India |
| Asha | RARS, India | Gouri | RARS, India | Onam | RARS, India |
| Pavizham | RARS, India | Prathyasa | RARS, India | Sagara | RARS, India |
| Karthika | RARS, India | Parambuvattan | RARS, India | VTL1 | RRS, India |
| Aruna | RARS, India | Kattamodan | RARS, India | VTL2 | RRS, India |
| Makam | RARS, India | Karuthammodan | RARS, India | VTL3 | RRS, India |
| Remya | RARS, India | Chuvannamodan | RARS, India | VTL4 | RRS, India |
| Kanakam | RARS, India | Swarnaprabha | RARS, India | VTL5R | RRS, India |
| Renjini | RARS, India | Neeraja | RARS, India | VTL5W | RRS, India |
| Pavithra | RARS, India | Kanchana | RARS, India | VTL6 | RRS, India |
| Panchami | RARS, India | Aiswarya | RARS, India | VTL7 | RRS, India |
| Remanika | RARS, India | Harsha | RARS, India | VTL8 | RRS, India |
| Uma | RARS, India | Vaishakh | RARS, India | ||
| IRRI, International Rice Research Institute; RARS, Regional Agricultural Research Station, Kerala Agricultural University; RRS, Rice Research Station, Kerala Agricultural University. | |||||
Table 1 Varieties used in this study.
| Variety | Origin | Variety | Origin | Variety | Origin |
|---|---|---|---|---|---|
| Sahbhagi Dhan (CK) | IRRI, the Philippines | Revathy | RARS, India | Mattathriveni | RARS, India |
| Jyothi (CK) | RARS, India | Karishma | RARS, India | Samyuktha | RARS, India |
| Bhadra | RARS, India | Krishnannjana | RARS, India | Bhagya | RARS, India |
| Asha | RARS, India | Gouri | RARS, India | Onam | RARS, India |
| Pavizham | RARS, India | Prathyasa | RARS, India | Sagara | RARS, India |
| Karthika | RARS, India | Parambuvattan | RARS, India | VTL1 | RRS, India |
| Aruna | RARS, India | Kattamodan | RARS, India | VTL2 | RRS, India |
| Makam | RARS, India | Karuthammodan | RARS, India | VTL3 | RRS, India |
| Remya | RARS, India | Chuvannamodan | RARS, India | VTL4 | RRS, India |
| Kanakam | RARS, India | Swarnaprabha | RARS, India | VTL5R | RRS, India |
| Renjini | RARS, India | Neeraja | RARS, India | VTL5W | RRS, India |
| Pavithra | RARS, India | Kanchana | RARS, India | VTL6 | RRS, India |
| Panchami | RARS, India | Aiswarya | RARS, India | VTL7 | RRS, India |
| Remanika | RARS, India | Harsha | RARS, India | VTL8 | RRS, India |
| Uma | RARS, India | Vaishakh | RARS, India | ||
| IRRI, International Rice Research Institute; RARS, Regional Agricultural Research Station, Kerala Agricultural University; RRS, Rice Research Station, Kerala Agricultural University. | |||||
| Trait | Score | Description |
|---|---|---|
| Drought resistance | 0 | Highly resistant: No symptoms |
| 1 | Resistant: Light tip drying | |
| 3 | Moderately resistant: Tip drying to ¼ length in most leaves | |
| 5 | Moderately susceptible: ¼ to ½ of leaves fully dried | |
| 7 | Susceptible: More than 2/3 of all leaves fully dried | |
| 9 | Highly susceptible: All plants apparently dead | |
| Drought recovery score | 1 | 90%-100% of plants recovered |
| 3 | 70%-89% of plants recovered | |
| 5 | 40%-69% of plants recovered | |
| 7 | 20%-39% of plants recovered | |
| 9 | 0%-19% of plants recovered | |
| Leaf rolling score | 0 | Leaves are healthy |
| 1 | Leaves start be to folded | |
| 3 | Leaves are folded (Deep-V-shaped) | |
| 5 | Leaves are fully cupped (U-shaped) | |
| 7 | Leaves margins touching (O-shaped) | |
| 9 | Leaves are tightly rolled |
Table 2 Drought scoring system in rice (IRRI, 1976).
| Trait | Score | Description |
|---|---|---|
| Drought resistance | 0 | Highly resistant: No symptoms |
| 1 | Resistant: Light tip drying | |
| 3 | Moderately resistant: Tip drying to ¼ length in most leaves | |
| 5 | Moderately susceptible: ¼ to ½ of leaves fully dried | |
| 7 | Susceptible: More than 2/3 of all leaves fully dried | |
| 9 | Highly susceptible: All plants apparently dead | |
| Drought recovery score | 1 | 90%-100% of plants recovered |
| 3 | 70%-89% of plants recovered | |
| 5 | 40%-69% of plants recovered | |
| 7 | 20%-39% of plants recovered | |
| 9 | 0%-19% of plants recovered | |
| Leaf rolling score | 0 | Leaves are healthy |
| 1 | Leaves start be to folded | |
| 3 | Leaves are folded (Deep-V-shaped) | |
| 5 | Leaves are fully cupped (U-shaped) | |
| 7 | Leaves margins touching (O-shaped) | |
| 9 | Leaves are tightly rolled |
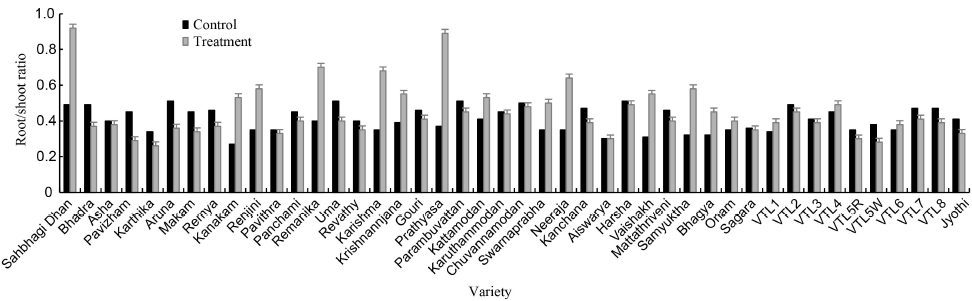
Fig. 1. Graphical representation of root/shoot ratio of rice varieties under 25% PEG6000 mediated osmotic stress (-6.75 MPa). ^Sahbhagi Dhan and Jyothi are the drought tolerant and susceptible check, respectively.^Values are expressed as Mean ± SD (n = 3).
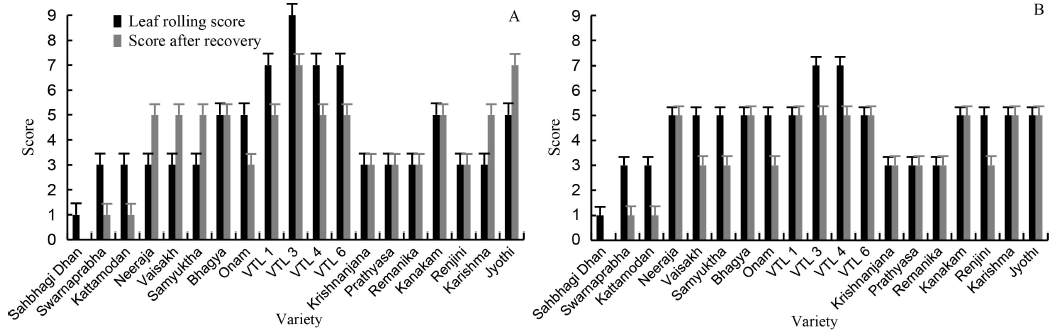
Fig. 2. Leaf rolling injury score of rice varieties under drought stress for 18 d by withdrawal of irrigation (A) and morphological drought injury score and its recovery score after rewatering (B).^Sahbhagi Dhan and Jyothi are the drought tolerant and susceptible check, respectively.^Values are expressed as Mean ± SD (n = 3).
| Variety | Cell membrane stability index | RWC (%) | Proline content (μmol/mg) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Root | Shoot | CK | DT | CK | DT | After drought recovery | ||||||
| CK | DT | CK | DT | |||||||||
| Sahbhagi Dhan | 41.86 | 36.62 | 17.70 | 20.19 | 80.08 | 77.77 a | 0.80 | 25.45 a | 2.16 g | |||
| Swarnaprabha | 37.04 | 35.16 | 17.89 | 18.40 | 78.58 | 70.22 c | 0.76 | 20.62 c | 4.38 d | |||
| Kattamodan | 14.29 | 29.29 | 17.21 | 15.67 | 80.38 | 65.45 d | 0.67 | 24.97 b | 6.85 c | |||
| Neeraja | 40.82 | 36.34 | 11.38 | 17.46 | 81.26 | 74.37 b | 0.86 | 9.20 d | 4.06 d | |||
| Vaisakh | 30.16 | 43.70 | 13.11 | 18.03 | 81.87 | 41.29 h | 0.66 | 5.85 e | 19.43 b | |||
| Samyuktha | 36.21 | 42.50 | 12.04 | 23.53 | 82.48 | 36.07 j | 1.74 | 4.82 f | 22.20 a | |||
| Bhagya | 43.33 | 52.56 | 13.39 | 17.16 | 85.29 | 57.05 f | 0.31 | 3.46 g | 6.30 c | |||
| Onam | 40.43 | 37.33 | 17.69 | 23.83 | 73.89 | 64.26 e | 0.24 | 0.80 k | 6.68 c | |||
| VTL1 | 30.77 | 36.36 | 11.11 | 11.11 | 76.78 | 32.63 j | 0.89 | 3.11 g | 4.00 d | |||
| VTL3 | 31.67 | 48.78 | 12.90 | 20.18 | 79.21 | 36.00 i | 0.99 | 0.11 l | 3.10 e | |||
| VTL4 | 31.67 | 35.00 | 12.50 | 27.39 | 79.89 | 32.69 k | 0.99 | 2.00 h | 2.85 f | |||
| VTL6 | 40.00 | 36.54 | 12.12 | 28.57 | 75.00 | 42.66 g | 0.78 | 3.09 g | 2.90 f | |||
| Krishnannjana | 35.42 | 34.78 | 19.15 | 8.11 | 68.46 | 37.99 l | 1.00 | 3.14 g | 3.00 e | |||
| Prathyasa | 30.19 | 33.11 | 22.78 | 18.57 | 82.99 | 38.30 o | 1.10 | 1.01 j | 2.00 g | |||
| Remanika | 34.00 | 38.33 | 33.33 | 13.75 | 84.26 | 34.20 m | 1.11 | 2.16 h | 2.13 g | |||
| Kanakam | 37.25 | 36.59 | 41.67 | 15.56 | 87.55 | 42.92 h | 0.89 | 2.00 h | 2.00 g | |||
| Renjini | 40.98 | 48.04 | 21.77 | 29.09 | 87.19 | 42.90 n | 1.00 | 1.01 j | 1.98 g | |||
| Karishma | 44.64 | 90.74 | 23.08 | 16.52 | 77.50 | 33.15 o | 1.00 | 1.99 h | 2.01 g | |||
| Jyothi | 32.11 | 39.94 | 26.87 | 25.69 | 86.91 | 38.78 ij | 1.00 | 1.47 i | 4.11 d | |||
| CD (0.05) | 1.569 | 1.613 | 1.591 | 1.789 | 2.283 | |||||||
| CK, Under control condition; DT, Under drought treatment. In each column, values followed by the different letters are significantly different at P < 0.05 by the Duncan’s test. | ||||||||||||
Table 3 Effect of drought stress on percent reduction in regards with electrolytic leakage (cell membrane stability index), relative water content (RWC) and proline content of rice varieties.
| Variety | Cell membrane stability index | RWC (%) | Proline content (μmol/mg) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Root | Shoot | CK | DT | CK | DT | After drought recovery | ||||||
| CK | DT | CK | DT | |||||||||
| Sahbhagi Dhan | 41.86 | 36.62 | 17.70 | 20.19 | 80.08 | 77.77 a | 0.80 | 25.45 a | 2.16 g | |||
| Swarnaprabha | 37.04 | 35.16 | 17.89 | 18.40 | 78.58 | 70.22 c | 0.76 | 20.62 c | 4.38 d | |||
| Kattamodan | 14.29 | 29.29 | 17.21 | 15.67 | 80.38 | 65.45 d | 0.67 | 24.97 b | 6.85 c | |||
| Neeraja | 40.82 | 36.34 | 11.38 | 17.46 | 81.26 | 74.37 b | 0.86 | 9.20 d | 4.06 d | |||
| Vaisakh | 30.16 | 43.70 | 13.11 | 18.03 | 81.87 | 41.29 h | 0.66 | 5.85 e | 19.43 b | |||
| Samyuktha | 36.21 | 42.50 | 12.04 | 23.53 | 82.48 | 36.07 j | 1.74 | 4.82 f | 22.20 a | |||
| Bhagya | 43.33 | 52.56 | 13.39 | 17.16 | 85.29 | 57.05 f | 0.31 | 3.46 g | 6.30 c | |||
| Onam | 40.43 | 37.33 | 17.69 | 23.83 | 73.89 | 64.26 e | 0.24 | 0.80 k | 6.68 c | |||
| VTL1 | 30.77 | 36.36 | 11.11 | 11.11 | 76.78 | 32.63 j | 0.89 | 3.11 g | 4.00 d | |||
| VTL3 | 31.67 | 48.78 | 12.90 | 20.18 | 79.21 | 36.00 i | 0.99 | 0.11 l | 3.10 e | |||
| VTL4 | 31.67 | 35.00 | 12.50 | 27.39 | 79.89 | 32.69 k | 0.99 | 2.00 h | 2.85 f | |||
| VTL6 | 40.00 | 36.54 | 12.12 | 28.57 | 75.00 | 42.66 g | 0.78 | 3.09 g | 2.90 f | |||
| Krishnannjana | 35.42 | 34.78 | 19.15 | 8.11 | 68.46 | 37.99 l | 1.00 | 3.14 g | 3.00 e | |||
| Prathyasa | 30.19 | 33.11 | 22.78 | 18.57 | 82.99 | 38.30 o | 1.10 | 1.01 j | 2.00 g | |||
| Remanika | 34.00 | 38.33 | 33.33 | 13.75 | 84.26 | 34.20 m | 1.11 | 2.16 h | 2.13 g | |||
| Kanakam | 37.25 | 36.59 | 41.67 | 15.56 | 87.55 | 42.92 h | 0.89 | 2.00 h | 2.00 g | |||
| Renjini | 40.98 | 48.04 | 21.77 | 29.09 | 87.19 | 42.90 n | 1.00 | 1.01 j | 1.98 g | |||
| Karishma | 44.64 | 90.74 | 23.08 | 16.52 | 77.50 | 33.15 o | 1.00 | 1.99 h | 2.01 g | |||
| Jyothi | 32.11 | 39.94 | 26.87 | 25.69 | 86.91 | 38.78 ij | 1.00 | 1.47 i | 4.11 d | |||
| CD (0.05) | 1.569 | 1.613 | 1.591 | 1.789 | 2.283 | |||||||
| CK, Under control condition; DT, Under drought treatment. In each column, values followed by the different letters are significantly different at P < 0.05 by the Duncan’s test. | ||||||||||||
| Variety | SPAD under stress | SPAD after recovery | Total chlorophyll | Chlorophyll a | Chlorophyll b | Carotenoid |
|---|---|---|---|---|---|---|
| Sahbhagi Dhan | 3.43 | 6.68 | -5.13 | -26.11 | -17.77 | 2.03 |
| Swarnaprabha | 15.46 | 14.65 | -12.69 | -37.21 | -15.00 | -19.72 |
| Kattamodan | 5.33 | -6.67 | 18.80 | 22.09 | 32.70 | 29.21 |
| Neeraja | 8.20 | 12.50 | 34.32 | 48.77 | 57.33 | 50.80 |
| Vaisakh | 3.28 | 21.16 | 21.25 | 19.41 | 24.29 | 30.18 |
| Samyuktha | 1.46 | -7.69 | 10.77 | 5.97 | 19.67 | 22.96 |
| Bhagya | 9.42 | 5.72 | 40.00 | 37.62 | 44.32 | 43.23 |
| Onam | -19.23 | -4.00 | 25.44 | 17.59 | 31.48 | 43.11 |
| VTL1 | 34.31 | -16.42 | 52.38 | -25.40 | 53.33 | 30.23 |
| VTL3 | 0.00 | -5.42 | 31.14 | -11.03 | 33.83 | 29.49 |
| VTL4 | 0.00 | -2.96 | 3.16 | 26.88 | 57.61 | 31.27 |
| VTL6 | 34.61 | 4.44 | 7.57 | -9.34 | 55.05 | 34.25 |
| Krishnannjana | 9.99 | 9.02 | 52.74 | 51.30 | 55.56 | 30.34 |
| Prathyasa | -20.00 | -6.41 | 57.71 | 56.56 | 58.82 | -12.13 |
| Remanika | 2.46 | -3.45 | -3.09 | -37.35 | 37.29 | 9.61 |
| Kanakam | 9.41 | -0.82 | 47.58 | 46.30 | 51.28 | 93.84 |
| Renjini | -10.74 | 9.01 | 55.36 | 44.66 | 19.67 | 42.95 |
| Karishma | 5.70 | 16.18 | 34.86 | 35.71 | 32.43 | 24.01 |
| Jyothi | 4.99 | 4.08 | 22.71 | 23.96 | 30.17 | 24.00 |
| CD (0.05) | 2.22 | 2.03 | 1.11 | 0.74 | 2.43 | 0.71 |
| SPAD, Soil and plant analyzer development. | ||||||
Table 4 Percent reductions of all parameter within each treatment were calculated as given below. %
| Variety | SPAD under stress | SPAD after recovery | Total chlorophyll | Chlorophyll a | Chlorophyll b | Carotenoid |
|---|---|---|---|---|---|---|
| Sahbhagi Dhan | 3.43 | 6.68 | -5.13 | -26.11 | -17.77 | 2.03 |
| Swarnaprabha | 15.46 | 14.65 | -12.69 | -37.21 | -15.00 | -19.72 |
| Kattamodan | 5.33 | -6.67 | 18.80 | 22.09 | 32.70 | 29.21 |
| Neeraja | 8.20 | 12.50 | 34.32 | 48.77 | 57.33 | 50.80 |
| Vaisakh | 3.28 | 21.16 | 21.25 | 19.41 | 24.29 | 30.18 |
| Samyuktha | 1.46 | -7.69 | 10.77 | 5.97 | 19.67 | 22.96 |
| Bhagya | 9.42 | 5.72 | 40.00 | 37.62 | 44.32 | 43.23 |
| Onam | -19.23 | -4.00 | 25.44 | 17.59 | 31.48 | 43.11 |
| VTL1 | 34.31 | -16.42 | 52.38 | -25.40 | 53.33 | 30.23 |
| VTL3 | 0.00 | -5.42 | 31.14 | -11.03 | 33.83 | 29.49 |
| VTL4 | 0.00 | -2.96 | 3.16 | 26.88 | 57.61 | 31.27 |
| VTL6 | 34.61 | 4.44 | 7.57 | -9.34 | 55.05 | 34.25 |
| Krishnannjana | 9.99 | 9.02 | 52.74 | 51.30 | 55.56 | 30.34 |
| Prathyasa | -20.00 | -6.41 | 57.71 | 56.56 | 58.82 | -12.13 |
| Remanika | 2.46 | -3.45 | -3.09 | -37.35 | 37.29 | 9.61 |
| Kanakam | 9.41 | -0.82 | 47.58 | 46.30 | 51.28 | 93.84 |
| Renjini | -10.74 | 9.01 | 55.36 | 44.66 | 19.67 | 42.95 |
| Karishma | 5.70 | 16.18 | 34.86 | 35.71 | 32.43 | 24.01 |
| Jyothi | 4.99 | 4.08 | 22.71 | 23.96 | 30.17 | 24.00 |
| CD (0.05) | 2.22 | 2.03 | 1.11 | 0.74 | 2.43 | 0.71 |
| SPAD, Soil and plant analyzer development. | ||||||
| Variety | Superoxide dismutase | Catalase | Peroxidase | Polyphenol oxidase | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | ||||
| Sahbhagi Dhan | 27.39 g | 24.65 f | 41.10 g | 29.97 f | 14.49 d | 10.70 d | -19.19 i | -24.04 l | |||
| Swarnaprabha | 222.34 d | 293.26 b | 153.28 c | 144.85 b | 11.81 def | 7.99 e | -22.07 j | -17.53 j | |||
| Kattamodan | 81.68 e | 24.36 f | 59.31 e | 37.04 e | 21.10 c | 11.24 d | -24.31 k | -20.27 k | |||
| Neeraja | 294.50 b | 1076.47 a | 267.18 a | 258.15 a | 23.76 c | 28.81 b | -18.54 i | -14.47 i | |||
| Vaisakh | 249.82 c | -93.42 n | 40.93 g | -54.57 k | -8.08 i | -12.20 g | -14.76 gh | -9.08 g | |||
| Samyuktha | 15.00 j | 24.65 f | 32.70 i | 85.78 c | 6.49 g | 3.12 f | -4.29 c | -0.68 c | |||
| Bhagya | 1509.65 a | -76.16 m | 203.98 b | -79.66 n | -56.66 k | -83.18 l | -16.17 h | -11.33 h | |||
| Onam | -2.08 n | -21.90 i | -6.46 j | -10.66 j | -22.27 j | -40.23 i | -0.09 b | -1.76 cd | |||
| VTL1 | 6.76 l | 12.01 h | 15.01 k | 14.95 h | 8.98 fg | 2.78 f | -9.26 ef | -15.47 i | |||
| VTL3 | 25.94 h | 27.85 e | 95.98 d | 34.51 e | 11.02 ef | 9.92 d | 1.90 a | 1.81 b | |||
| VTL4 | -16.77 q | 80.63 d | 37.70 h | 66.09 d | 32.69 b | 26.54 c | -3.02 c | 5.51 a | |||
| VTL6 | 28.45 f | 21.13 g | 24.85 j | 89.18 c | 41.33 a | 38.52 a | -6.49 d | 0.96 b | |||
| Krishnanjana | -33.62 r | -68.99 l | -36.05 o | 2.38 i | -83.06 m | -91.76 m | 2.97 a | 1.02 b | |||
| Prathyasa | 1.10 m | -60.04 k | 11.24 l | -68.70 m | -91.75 n | -56.48 j | -8.15 de | -2.64 d | |||
| Remanika | 20.88 i | 19.71 g | 49.38 f | 23.34 g | -21.28 j | -21.45 h | -13.91 g | -7.88 fg | |||
| Kanakam | -97.30 s | 24.91 f | -80.02 q | 24.82 g | -2.98 h | 1.62 f | -10.60 f | -4.35 e | |||
| Renjini | -9.23 o | -43.58 j | -17.77 n | -60.43 l | -63.02 l | -61.40 k | -11.14 f | -12.54 h | |||
| Karishma | 9.67 k | 176.76 c | 25.33 j | -119.82 o | 12.60 de | 6.91 e | -7.13 d | -7.42 f | |||
| CD (0.05) | 0.981 | 1.436 | 2.102 | 3.909 | 3.114 | 1.711 | 1.947 | 1.372 | |||
| In each column, values followed by the different letters are significantly different at P < 0.05 by the Duncan’s test. | |||||||||||
Table 5 Effect of drought stress on several enzyme activities in rice varieties. mg/(min∙protein)
| Variety | Superoxide dismutase | Catalase | Peroxidase | Polyphenol oxidase | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | ||||
| Sahbhagi Dhan | 27.39 g | 24.65 f | 41.10 g | 29.97 f | 14.49 d | 10.70 d | -19.19 i | -24.04 l | |||
| Swarnaprabha | 222.34 d | 293.26 b | 153.28 c | 144.85 b | 11.81 def | 7.99 e | -22.07 j | -17.53 j | |||
| Kattamodan | 81.68 e | 24.36 f | 59.31 e | 37.04 e | 21.10 c | 11.24 d | -24.31 k | -20.27 k | |||
| Neeraja | 294.50 b | 1076.47 a | 267.18 a | 258.15 a | 23.76 c | 28.81 b | -18.54 i | -14.47 i | |||
| Vaisakh | 249.82 c | -93.42 n | 40.93 g | -54.57 k | -8.08 i | -12.20 g | -14.76 gh | -9.08 g | |||
| Samyuktha | 15.00 j | 24.65 f | 32.70 i | 85.78 c | 6.49 g | 3.12 f | -4.29 c | -0.68 c | |||
| Bhagya | 1509.65 a | -76.16 m | 203.98 b | -79.66 n | -56.66 k | -83.18 l | -16.17 h | -11.33 h | |||
| Onam | -2.08 n | -21.90 i | -6.46 j | -10.66 j | -22.27 j | -40.23 i | -0.09 b | -1.76 cd | |||
| VTL1 | 6.76 l | 12.01 h | 15.01 k | 14.95 h | 8.98 fg | 2.78 f | -9.26 ef | -15.47 i | |||
| VTL3 | 25.94 h | 27.85 e | 95.98 d | 34.51 e | 11.02 ef | 9.92 d | 1.90 a | 1.81 b | |||
| VTL4 | -16.77 q | 80.63 d | 37.70 h | 66.09 d | 32.69 b | 26.54 c | -3.02 c | 5.51 a | |||
| VTL6 | 28.45 f | 21.13 g | 24.85 j | 89.18 c | 41.33 a | 38.52 a | -6.49 d | 0.96 b | |||
| Krishnanjana | -33.62 r | -68.99 l | -36.05 o | 2.38 i | -83.06 m | -91.76 m | 2.97 a | 1.02 b | |||
| Prathyasa | 1.10 m | -60.04 k | 11.24 l | -68.70 m | -91.75 n | -56.48 j | -8.15 de | -2.64 d | |||
| Remanika | 20.88 i | 19.71 g | 49.38 f | 23.34 g | -21.28 j | -21.45 h | -13.91 g | -7.88 fg | |||
| Kanakam | -97.30 s | 24.91 f | -80.02 q | 24.82 g | -2.98 h | 1.62 f | -10.60 f | -4.35 e | |||
| Renjini | -9.23 o | -43.58 j | -17.77 n | -60.43 l | -63.02 l | -61.40 k | -11.14 f | -12.54 h | |||
| Karishma | 9.67 k | 176.76 c | 25.33 j | -119.82 o | 12.60 de | 6.91 e | -7.13 d | -7.42 f | |||
| CD (0.05) | 0.981 | 1.436 | 2.102 | 3.909 | 3.114 | 1.711 | 1.947 | 1.372 | |||
| In each column, values followed by the different letters are significantly different at P < 0.05 by the Duncan’s test. | |||||||||||
| [1] | Agarie S, Hanaoka N, Kubota F, Agata W, Kaufman B P.1995. Measurement of cell membrane stability evaluated by electrolyte leakage as a drought and heat tolerance test in rice (Oryza sativa L.).J Fac Agric, 40(1/2): 233-240. |
| [2] | Alscher R G, Erturk N, Heath L S.2002. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants.J Exp Bot, 53: 1331-1341. |
| [3] | Anami S, de Block M, Machuka J, van Lijsebettens M.2009. Molecular improvement of tropical maize for drought stress tolerance in Sub-Saharan Africa.Crit Rev Plant Sci, 28: 16-35. |
| [4] | Arnon D I.1949. Copper enzymes in isolated chloroplasts.Polyph Bet Vulg Plant Physiol, 24(1): 1-15. |
| [5] | Ashraf M, Foolad M R.2007. Role of glycine betaine and proline in improving plant abiotic stress resistance.Environ Exp Bot, 59(2): 206-216. |
| [6] | Bates L S, Waldren R P, Teare I D.1973. Rapid determination of free proline for water stress studies.Plant Soil, 39(1): 205-207. |
| [7] | Beauchamp C, Fridovich I.1971. SOD improved assays and an assay applicable to acrylamide gel.Anal Biochem, 44(1): 276-287. |
| [8] | Blokhina O, Virolainen E, Fagerstedt K V.2003. Anti-oxidative damage and oxygen deprivation stress.Ann Bot, 91: 179-194. |
| [9] | Bray E A.2002. Abscisic acid regulation of gene expression during water-deficit stress in the era of the Arabidopsis genome.Plant Cell Environ, 25(2): 153-161. |
| [10] | Cabuslay G S, Ito O, Alejar A A.1999. Genotypic differences in physiological responses to water deficit in rice. In: Genetic Improvement of Rice for Water-Limited Environments. Los Banos, the Phillipine: International Rice Research Institute: 99-116. |
| [11] | Cabuslay G S, Ito O, Alejar A A.2002. Physiological evaluation of responses of rice (Oryza sativa L.) to water deficit.Plant Sci, 163(4): 815-827. |
| [12] | Chaturvedi G S, Singh A, Bahadur R.2012. Screening techniques for evaluating crop germplasm for drought tolerance.Plant Arch, 12(1): 11-18. |
| [13] | Chempakam B, Kastui B K V, Rajagopal V.1993. Lipid peroxidation in relation to drought tolerance in coconut (Cocos nucifera L.).Plant Physiol Biochem, 20(1): 5-10. |
| [14] | Chen D Q, Wang S W, Cao B B, Cao D, Leng G H, Li H B, Yin L N, Shan L, Deng X P.2015. Genotypic variation in growth and physiological response to drought stress and re-watering reveals the critical role of recovery in drought adaptation in maize seedlings.Front Plant Sci, 6: 1241. |
| [15] | Central Rice Research Institute (CRRI). 2009-2010. CRRI Annual Report. Indian Council of Agricultural Research, Cuttack, India. |
| [16] | de Datta S K, Chang T T, Yoshida S.1975. Drought tolerance in upland rice. In: IRRI. Major Research in Upland Rice. Los Banos, the Philippines: International Rice Research Institute: 101-116. |
| [17] | Directorate of Economics and Statistics.2009. Area, Production and Yield of Principal Crops. Department of Agriculture and Cooperation, Ministry of Agriculture, Government of India. |
| [18] | Fanizza G, Ricciardi L, Bagnulo C.1991. Leaf greenness measurements to evaluate water stressed genotypes in Vitis vinifera.Euphytica, 55(1): 27-31. |
| [19] | Fang Y J, Xiong L Z.2015. General mechanisms of drought response and their application in drought resistance improvement in plants.Cell Mol Life Sci, 72(4): 673-689. |
| [20] | Food and Agriculture Organization (FAO). 2002. Crops and Drops-Making the Best Use of Water for Agriculture. Rome: Food and Agriculture Organization of the United Nations. |
| [21] | Fen L L, Ismail M R, Zulkarami B, Rahman M S A, Robiul Ismail M.2015. Physiological and molecular characterization of drought responses and screening of drought tolerant rice varieties.Biosci J, 31(3): 709-718. |
| [22] | Fischer R A.1980. Influence of water stress on crop yield in semi arid regions. In: Turner N C, Kramer P. Adaptation of Plants to Water and High Temperature Stress. New York: Willey and Son: 323-340. |
| [23] | Floyd R A, Zs-Nagy I.1984. Formation of long lived hydroxyl free radical adducts of proline and hydroxyproline in a fenton reaction.Biochem Biophys Acta, 790(1): 94-97. |
| [24] | Foyer C H, Lelandais M, Kunert K J.1994. Photo oxidative stress in plants.Physiol Plant, 92: 696-717. |
| [25] | Foyer C H, Noctor G.2003. Redox sensing and signaling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria.Physiol Plant, 119(3): 355-364. |
| [26] | Franco J A, Bañón S, Vicente M J, Miralles J, Martínez-Sánchez J J.2011. Root development in horticultural plants grown under abiotic stress conditions: A review.J Hort Sci Biotechnol, 86: 543-556. |
| [27] | Franco J A, Martínez-Sánchez J J, Fernández J A, Bañón S.2006. Selection and nursery production of ornamental plants for landscaping and xerogardening in semi-arid environments.J Hort Sci Biotech, 81(1): 3-17. |
| [28] | Gill S S, Anjum N A, Gill R, Tuteja N.2016. Abiotic stress signaling in plants: An overview, In: Tuteja N, Gill S S. Abiotic Stress Response in Plants. Weinheim, Germany: Wiley: 3-22. |
| [29] | Gowda V R P, Henry A, Yamauchi A, Shashidhar H E, Serraj R.2011. Root biology and genetics improvement for drought avoidance in rice.Field Crops Res, 122(1): 1-13. |
| [30] | Hossain M.1995. Sustaining food security for fragile environments in Asia: Achievements, challenges, and implications for rice research. In: Hossain M. Fragile Lives in Fragile Ecosystem. Proceedings of the International Rice Research Conference. Los Banos, The Philippines: International Rice Research Institute: 3-23. |
| [31] | Ingram J, Bartels D.1996. The molecular basis of dehydration tolerance in plants.Annu Rev Plant Physiol Plant Mol Biol, 47: 377-403. |
| [32] | International Rice Research Institute (IRRI). 1996. Standard Evaluation System for Rice. Los Banos, the Philippines: International Rice Research Institute. |
| [33] | Jaleel C A, Manivannan P, Lakshmanan G M A, Gomathinayagam M, Panneerselvam R.2008. Alterations in morphological parameters and photosynthetic pigment responses of Catharanthus roseus under soil water deficits.Coll Surf B: Biointer, 61(2): 298-303. |
| [34] | Kalefetoglu Macar T, Ekmekci Y.2009. Alterations in photochemical and physiological activities of chickpea (Cicer arietinum L.) cultivars under drought stress.J Agron Crop Sci, 195(5): 335-346. |
| [35] | Kar M, Mishra D.1976. Catalase, peroxidase, and polyphenoloxidase activities during rice leaf senescence.Plant Physiol, 57(2): 315-319. |
| [36] | Kasturi Bai K V, Rajagopal V, Chempakam B, Prabha C.1996. Assay of enzymes in coconut cultivars and hybrids under non-stress and stress condition.J Plant Crops, 24: 548-554. |
| [37] | Kennedy G, Burlingame B, Nguyen V N.2002. Proceedings of the 20th Session of the International Rice Commission. Bangkok, Thailand. |
| [38] | Kerala Agricultural University (KAU). 2011. Package of Practices Recommendations: Crops. 14th Edition. Thrissur: Kerala Agricultural University: 360. |
| [39] | Kriedemann P E, Barrs H D.1981. Citrus orchards. In: Kozlowski T T. Water Deficits and Plant Growth. New York: Academic Press: 325-416. |
| [40] | Lafitte H R, Courtois B, Arraydeau M.2002. Genetic improvement of rice in aerobic systems: Progress from yield to genes.Field Crops Res, 75: 171-190. |
| [41] | Lessani H, Mojtahedi M.2002. Introduction to Plant Physiology (Translation). 6th edn. Iran: Tehran University Press: 726. |
| [42] | Lodh S B, Bhattacharjee D P, Ramakrishnaya G, Deb S P.1977. Studies on the oxidative enzymes of rice in relation to soil moisture stress.Ind Agric, 21: 181-186. |
| [43] | Maji A T.1994. Vegetative stage drought tolerance and agronomic characteristics of Oryza glaberrima accessions. Nigeria: University of Ibadan. |
| [44] | McKersie B D, Bowley S R, Jones K S.1999. Winter survival of transgenic alfalfa overexpressing superoxide dismutase.Plant Physiol, 119(3): 839-848. |
| [45] | Moaveni P.2011. Effect of water deficit stress on some physiological traits of wheat (Triticum aestivum).J Agric Sci Res, 1(1): 64-68. |
| [46] | Morgan J M.1984. Osmoregulation and water stress in higher plants.Annu Rev Plant Physiol, 35: 299-319. |
| [47] | Mullet J E, Whitsitt M S.1996. Plant cellular responses to water deficits.Plant Growth Regul, 20: 119-124. |
| [48] | O’Neill P M, Shanahan J F, Schepers J S.2006. Use of chlorophyll fluorescence assessments to differentiate corn hybrid response to variable water conditions.Crop Sci, 46(2): 681-687. |
| [49] | Pandey V, Shukla A.2015. Acclimation and tolerance strategies of rice under drought stress.Rice Sci, 22(4): 147-161. |
| [50] | Pinhero R G, Fletcher R A.1994. Paclobutrazol and ancymidol protect corn seedlings from high and low temperature stresses.Plant Growth Regul, 15(1): 47-53. |
| [51] | Tirado R, Cotter J.2010. Ecological farming: Drought-resistant agriculture. In: Erwood S, Truchi N, Stabinsky D. Greenpeace Research Laboratories Technical Note. Amsterdam, Netherland: the Netherlands Greenpeace International. |
| [52] | Selote D S, Khanna-Chopra R.2010. Antioxidant response of wheat roots to drought acclimation.Protoplasma, 245: 153-163. |
| [53] | Silva M de A, Jifon J L, Silva J A Gda, Sharma V.2007. Use of physiological parameters as fast tools to screen for drought tolerance in sugarcane.Braz J Plant Physiol, 19(3): 193-201. |
| [54] | Sikuku P A, Onyango J C, Netondo G W.2012. Physiological and biochemical responses of five nerica rice varieties (Oryza sativa L.) to water deficit at vegetative and reproductive stage.Agric Biol J N Am, 3(3): 93-104. |
| [55] | Smirnoff N.1993. The role of active oxygen in the response of plants to water deficit and desiccation.New Phytol, 125(1): 27-58. |
| [56] | Sunoj V S J, Naresh Kumar S, Muralikrishna K S.2014. Effect of elevated CO2 and temperature on oxidative stress and antioxidant enzymes activity in coconut (Cocosnucifera L.) seedlings.Ind J Plant Physiol, 19(4): 382-387. |
| [57] | Swamy B P M, Kumar A.2012. Sustainable rice yield in water-short drought-prone environments: Conventional and molecular approaches. In: Lee T S. Irrigation Systems and Practices in Challenging Environments. German: InTech. |
| [58] | Turner N C.1986. Adaptation to water deficits: A changing perspective.Aust J Plant Physiol, 13(1): 175-189. |
| [59] | Vendruscolo E C G, Schuster I, Pileggi M, Scapim C A, Molinari H B C, Marur C J, Vieira L G E.2007. Stress induced synthesis of proline confers tolerance to water deficit in transgenic wheat.J Plant Physiol, 164(10): 1367-1376. |
| [60] | Vikram P, Swamy B P M, Dixit S, Ahmed H U, Cruz M T S, Singh A K, Kumar A.2011. qDTY1.1, a major QTL for rice grain yield under reproductive-stage drought stress with a consistent effect in multiple elite genetic backgrounds.BMC Genet, 12: 89. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [13] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||