
Rice Science ›› 2020, Vol. 27 ›› Issue (4): 329-344.DOI: 10.1016/j.rsci.2020.05.008
• Research Paper • Previous Articles Next Articles
Dey Nivedita, Bhattacharjee Soumen( )
)
Received:2019-05-19
Accepted:2019-12-26
Online:2020-07-28
Published:2020-03-31
Dey Nivedita, Bhattacharjee Soumen. Accumulation of Polyphenolic Compounds and Osmolytes under Dehydration Stress and Their Implication in Redox Regulation in Four Indigenous Aromatic Rice Cultivars[J]. Rice Science, 2020, 27(4): 329-344.
Add to citation manager EndNote|Ris|BibTeX
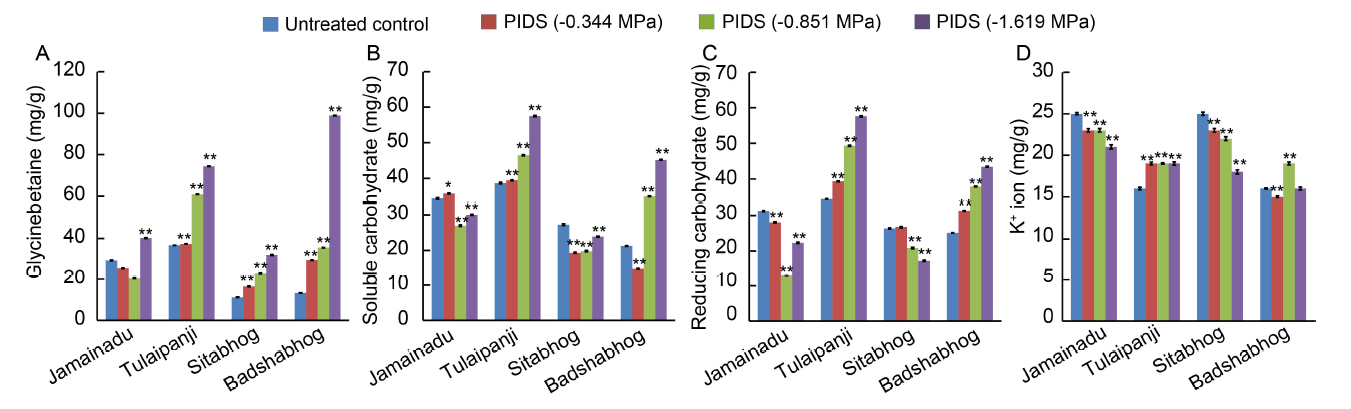
Fig. 1. Differential osmolyte accumulation (organic and inorganic).A, Accumulation of glycinebetaine. B, Soluble carbohydrate content. C, Reducing carbohydrate content. D, Accumulation of K+ ion.PIDS, Post imbibitional dehydration stress.Data are Mean ± SE (n = 3). * and **, Significant differences at the 0.05 and 0.01 levels (t-test), respectively.
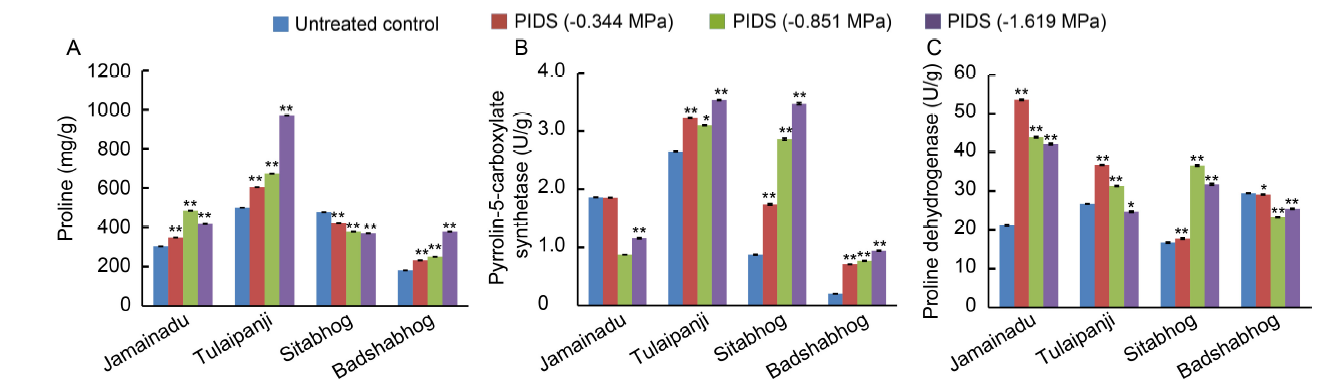
Fig. 2. Differential proline accumulation (A) and activities of pyrrolin-5-carboxylate synthetase (B) and proline dehydrogenase (C).PIDS, Post imbibitional dehydration stress.Data are Mean ± SE (n = 3). * and **, Significant differences at the 0.05 and 0.01 levels (t-test), respectively.
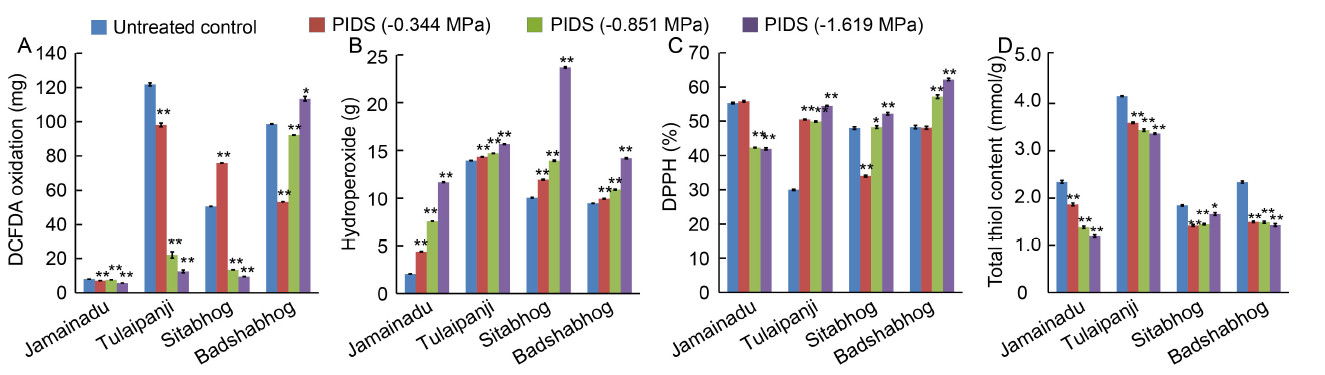
Fig. 3. Dehydration stress induced changes in internal redox cues assessed in terms of total reactive oxygen species (ROS) accumulation.A, DCFDA (2′,7′-dichlorofluorescin diacetate) oxidation. B, Hydrogen peroxide accumulation. C, DPPH assay. D, Total thiol content.PIDS, Post imbibitional dehydration stress; DCFDA, 2′,7′-dichlorofluorescin diacetate; DPPH, 2,2-diphenyl-1-pycryl hydrazyl.Data are Mean ± SE (n = 3). * and **, Significant differences at the 0.05 and 0.01 levels (t-test), respectively.
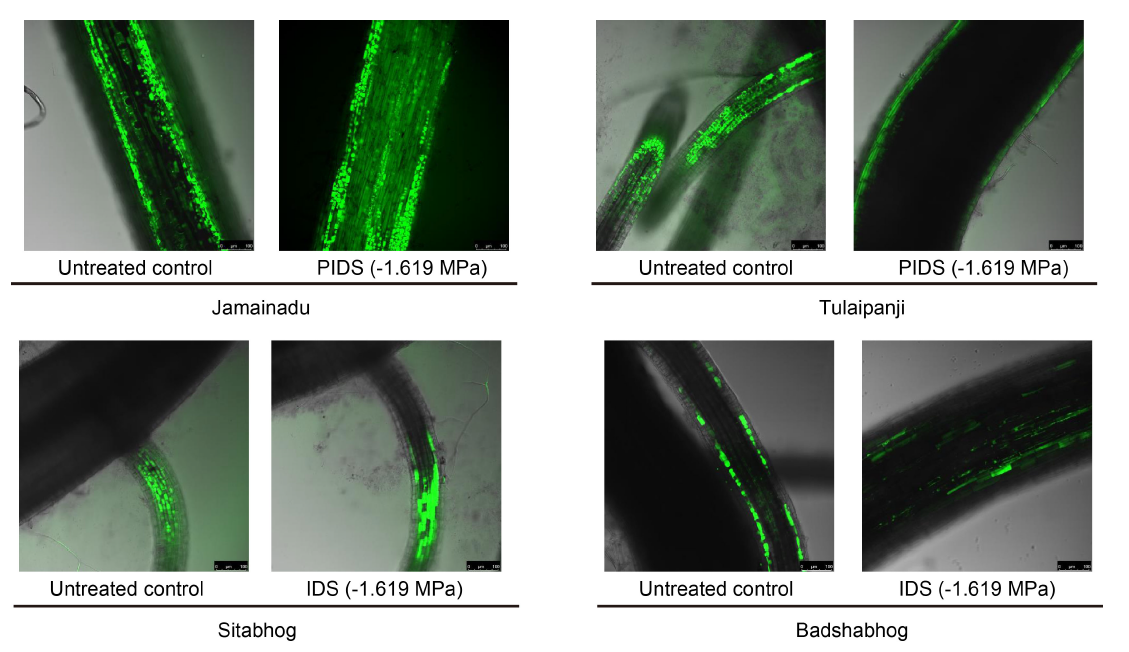
Fig. 4. Laser confocal microscopy showing accumulation of reactive oxygen species (H2O2) using Leica application suite X software.PIDS, Post imbibitional dehydration stress.
| Cultivar | Treatment | Flavonoid | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catechin | Naringin | Rutin | Quercetin | Kaempferol | Myricetin | Apigenin | ||||||||||||
| Jamainadu | Untreated control | 99.3 | 7 963.7 | 4 773.0 | 3 448.0 | 79.3 | - | - | ||||||||||
| -1.619 MPa | 163.0 | 4 732.3 a | 265.3 a | 3 026.0 a | - | - | - | |||||||||||
| Tulaipanji | Untreated control | 116.0 | 12 533.3 | 8.3 | 4 648.3 | 133.0 | 356.7 | 26.3 | ||||||||||
| -1.619 MPa | 108.0 | 5 106.3 | 401.7 b | 3 287.0 | 33.3 b | 12.7 | - | |||||||||||
| Sitabhog | Untreated control | 7 042.3 | 2 315.4. | 5 064.0 | 11 009.3 | - | 90.3 | - | ||||||||||
| -1.619 MPa | 148.7 a | 5 920.0 a | 413.3 a | 4 105.7 a | - | 44.3 | - | |||||||||||
| Badshabhog | Untreated control | 5.0 | 8 718.3 | 48.0 | 4 929.7 | 16.0 | - | - | ||||||||||
| -1.619 MPa | - | 6 957.0 b | 167.7 b | 4 369.3 | 27.0 b | 216.7 b | 77.3 b | |||||||||||
| Cultivar | Treatment | Phenolic acid | ||||||||||||||||
| Gallic acid | Protocatechuic acid | Gentistic acid | P-ohbenzoic acid | Chlorogenic acid | Caffeic acid | Syringic acid | Salicylic acid | Sinapic acid | P-coumaric acid | |||||||||
| Jamainadu | Untreated control | 1 519.7 | 647.7 | 4 042.0 | 45.0 | 25.0 | 38.7 | 62.3 | 20.0 | - | - | |||||||
| -1.619 MPa | 700.0 a | 41.7 a | 1 862.0 a | 8.7 a | - | - | 25.7 | - | - | - | ||||||||
| Tulaipanji | Untreated control | 3 027.3 | 687.7 | 3 107.3 | 76.0 | 8.7 | 23.0 | 35.3 | - | - | - | |||||||
| -1.619 MPa | 866.7 | 26.7 | 4 314.0 b | 14.0 | 104.7 b | - | 24.3 | - | - | - | ||||||||
| Sitabhog | Untreated control | 6 735.0 | 141.7 | 88 305.0 | 1 938.3 | 6 821.3 | 39.7 | 1 513.7 | - | - | - | |||||||
| -1.619 MPa | 2 345.7 a | 40.3 a | - | 94.7 a | 518.7 a | 12.3 a | 55.0 a | - | 20.3 | - | ||||||||
| Badshabhog | Untreated control | 732.0 | 1841.3 | 19 256.3 | 365.7 | 570.0 | 17.7 | 112.7 | 68.7 | - | 0.3 | |||||||
| -1.619 MPa | 863.3 b | 5.0 | - | 300.0 | 652.7 b | 18.7 b | 62.3 | - | 38.3 b | 95.7 b | ||||||||
Table 1 Changes in concentrations of flavonoids and phenolic acids associated under post imbibitional dehydration stress over their corresponding untreated control in seedlings (7-day-old) of four indigenous aromatic rice cultivars. ng/g
| Cultivar | Treatment | Flavonoid | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catechin | Naringin | Rutin | Quercetin | Kaempferol | Myricetin | Apigenin | ||||||||||||
| Jamainadu | Untreated control | 99.3 | 7 963.7 | 4 773.0 | 3 448.0 | 79.3 | - | - | ||||||||||
| -1.619 MPa | 163.0 | 4 732.3 a | 265.3 a | 3 026.0 a | - | - | - | |||||||||||
| Tulaipanji | Untreated control | 116.0 | 12 533.3 | 8.3 | 4 648.3 | 133.0 | 356.7 | 26.3 | ||||||||||
| -1.619 MPa | 108.0 | 5 106.3 | 401.7 b | 3 287.0 | 33.3 b | 12.7 | - | |||||||||||
| Sitabhog | Untreated control | 7 042.3 | 2 315.4. | 5 064.0 | 11 009.3 | - | 90.3 | - | ||||||||||
| -1.619 MPa | 148.7 a | 5 920.0 a | 413.3 a | 4 105.7 a | - | 44.3 | - | |||||||||||
| Badshabhog | Untreated control | 5.0 | 8 718.3 | 48.0 | 4 929.7 | 16.0 | - | - | ||||||||||
| -1.619 MPa | - | 6 957.0 b | 167.7 b | 4 369.3 | 27.0 b | 216.7 b | 77.3 b | |||||||||||
| Cultivar | Treatment | Phenolic acid | ||||||||||||||||
| Gallic acid | Protocatechuic acid | Gentistic acid | P-ohbenzoic acid | Chlorogenic acid | Caffeic acid | Syringic acid | Salicylic acid | Sinapic acid | P-coumaric acid | |||||||||
| Jamainadu | Untreated control | 1 519.7 | 647.7 | 4 042.0 | 45.0 | 25.0 | 38.7 | 62.3 | 20.0 | - | - | |||||||
| -1.619 MPa | 700.0 a | 41.7 a | 1 862.0 a | 8.7 a | - | - | 25.7 | - | - | - | ||||||||
| Tulaipanji | Untreated control | 3 027.3 | 687.7 | 3 107.3 | 76.0 | 8.7 | 23.0 | 35.3 | - | - | - | |||||||
| -1.619 MPa | 866.7 | 26.7 | 4 314.0 b | 14.0 | 104.7 b | - | 24.3 | - | - | - | ||||||||
| Sitabhog | Untreated control | 6 735.0 | 141.7 | 88 305.0 | 1 938.3 | 6 821.3 | 39.7 | 1 513.7 | - | - | - | |||||||
| -1.619 MPa | 2 345.7 a | 40.3 a | - | 94.7 a | 518.7 a | 12.3 a | 55.0 a | - | 20.3 | - | ||||||||
| Badshabhog | Untreated control | 732.0 | 1841.3 | 19 256.3 | 365.7 | 570.0 | 17.7 | 112.7 | 68.7 | - | 0.3 | |||||||
| -1.619 MPa | 863.3 b | 5.0 | - | 300.0 | 652.7 b | 18.7 b | 62.3 | - | 38.3 b | 95.7 b | ||||||||
| Cultivar | Treatment | Biomarker of oxidative stress | Germination performance | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MLPO (mmol/g) | Lipoxygenase (U/g) | Hydroperoxide (A560/g) | Conjugated diene (nmol/g) | CVG (%) | GRI | MGT | MDG | ||||||||||
| Jamainadu | Control | 22.48 ± 0.22 | 94.39 ± 0.27 | 2.07 ± 0.01 | 73.13 ± 0.27 | 23.35 ± 0.06 | 90.76 ± 0.08 | 4.28 ± 0.06 | 7.50 ± 0.05 | ||||||||
| -0.344 MPa | 49.87 ± 0.14** | 99.75 ± 0.41** | 4.38 ± 0.02** | 186.38 ± 0.38** | 23.63 ± 0.07* | 99.20 ± 0.09** | 4.23 ± 0.05 | 11.75 ± 0.06** | |||||||||
| -0.851 MPa | 44.28 ± 0.17** | 148.61 ± 0.52** | 7.62 ± 0.03** | 383.25 ± 0.53** | 23.36 ± 0.07* | 78.54 ± 0.10** | 4.28 ± 0.06 | 6.50 ± 0.07** | |||||||||
| -1.619 MPa | 48.61 ± 0.19** | 224.55 ± 0.57** | 11.69 ± 0.04** | 472.50 ± 0.57** | 21.96 ± 0.08** | 36.22 ± 0.10** | 4.55 ± 0.06** | 3.83 ± 0.06** | |||||||||
| Tulaipanji | Control | 24.33 ± 0.11 | 185.88 ± 0.25 | 13.94 ± 0.01 | 255.00 ± 0.30 | 59.35 ± 0.03 | 48.00 ± 0.05 | 1.68 ± 0.03 | 25.00 ± 0.03 | ||||||||
| -0.344 MPa | 18.93 ± 0.14** | 203.97 ± 0.27** | 14.33 ± 0.02** | 342.75 ± 0.41** | 61.83 ± 0.04** | 56.00 ± 0.04** | 1.62 ± 0.04 | 25.00 ± 0.04 | |||||||||
| -0.851 MPa | 26.18 ± 0.05** | 221.97 ± 0.3** | 14.71 ± 0.02** | 421.13 ± 0.40** | 60.00 ± 0.04** | 50.00 ± 0.05** | 1.67 ± 0.04 | 25.00 ± 0.05 | |||||||||
| -1.619 MPa | 28.00 ± 0.08** | 245.00 ± 0.35** | 15.67 ± 0.03** | 442.88 ± 0.51** | 45.08 ± 0.04** | 63.17 ± 0.03** | 2.22 ± 0.03** | 16.67 ± 0.03** | |||||||||
| Sitabhog | Control | 19.45 ± 0.16 | 387.89 ± 0.33 | 10.06 ± 0.04 | 198.75 ± 0.36 | 23.23 ± 0.07 | 71.01 ± 0.09 | 4.30 ± 0.07 | 9.00 ± 0.06 | ||||||||
| -0.344 MPa | 18.23 ± 0.20** | 565.58 ± 0.49** | 11.95 ± 0.05** | 199.88 ± 0.43* | 23.43 ± 0.06** | 64.97 ± 0.09** | 4.27 ± 0.08 | 8.00 ± 0.05** | |||||||||
| -0.851 MPa | 22.64 ± 0.15** | 565.66 ± 0.63** | 13.93 ± 0.06** | 229.13 ± 0.54** | 22.72 ± 0.06** | 50.68 ± 0.09** | 4.40 ± 0.06* | 5.60 ± 0.04** | |||||||||
| -1.619 MPa | 26.31 ± 0.22** | 423.47 ± 0.76** | 23.69 ± 0.08** | 303.75 ± 0.61** | 21.50 ± 0.07** | 41.19 ± 0.10** | 4.65 ± 0.08** | 4.83 ± 0.04** | |||||||||
| Badshabhog | Control | 23.33 ± 0.12 | 477.86 ± 0.15 | 9.47 ± 0.02 | 227.63 ± 0.54 | 22.39 ± 0.05 | 43.15 ± 0.07 | 4.47 ± 0.06 | 6.25 ± 0.05 | ||||||||
| -0.344 MPa | 27.15 ± 0.15** | 497.44 ± 0.33** | 9.95 ± 0.04** | 239.25 ± 0.51** | 24.45 ± 0.04** | 44.14 ± 0.07** | 4.09 ± 0.05** | 5.00 ± 0.03** | |||||||||
| -0.851 MPa | 26.04 ± 0.18** | 552.92 ± 0.38** | 10.91 ± 0.05** | 311.63 ± 0.49** | 22.26 ± 0.04 | 41.82 ± 0.03** | 4.49 ± 0.05 | 6.25 ± 0.04 | |||||||||
| -1.619 MPa | 24.62 ± 0.19* | 823.94 ± 0.46** | 14.18 ± 0.06** | 312.75 ± 0.30** | 22.10 ± 0.04* | 36.74 ± 0.05** | 4.53 ± 0.05 | 3.83 ± 0.04** | |||||||||
Table 2 Biomarkers of oxidative stress vis-à-vis their germination performances of four indigenous aromatic rice cultivars grown under post imbibitional dehydration stress.
| Cultivar | Treatment | Biomarker of oxidative stress | Germination performance | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MLPO (mmol/g) | Lipoxygenase (U/g) | Hydroperoxide (A560/g) | Conjugated diene (nmol/g) | CVG (%) | GRI | MGT | MDG | ||||||||||
| Jamainadu | Control | 22.48 ± 0.22 | 94.39 ± 0.27 | 2.07 ± 0.01 | 73.13 ± 0.27 | 23.35 ± 0.06 | 90.76 ± 0.08 | 4.28 ± 0.06 | 7.50 ± 0.05 | ||||||||
| -0.344 MPa | 49.87 ± 0.14** | 99.75 ± 0.41** | 4.38 ± 0.02** | 186.38 ± 0.38** | 23.63 ± 0.07* | 99.20 ± 0.09** | 4.23 ± 0.05 | 11.75 ± 0.06** | |||||||||
| -0.851 MPa | 44.28 ± 0.17** | 148.61 ± 0.52** | 7.62 ± 0.03** | 383.25 ± 0.53** | 23.36 ± 0.07* | 78.54 ± 0.10** | 4.28 ± 0.06 | 6.50 ± 0.07** | |||||||||
| -1.619 MPa | 48.61 ± 0.19** | 224.55 ± 0.57** | 11.69 ± 0.04** | 472.50 ± 0.57** | 21.96 ± 0.08** | 36.22 ± 0.10** | 4.55 ± 0.06** | 3.83 ± 0.06** | |||||||||
| Tulaipanji | Control | 24.33 ± 0.11 | 185.88 ± 0.25 | 13.94 ± 0.01 | 255.00 ± 0.30 | 59.35 ± 0.03 | 48.00 ± 0.05 | 1.68 ± 0.03 | 25.00 ± 0.03 | ||||||||
| -0.344 MPa | 18.93 ± 0.14** | 203.97 ± 0.27** | 14.33 ± 0.02** | 342.75 ± 0.41** | 61.83 ± 0.04** | 56.00 ± 0.04** | 1.62 ± 0.04 | 25.00 ± 0.04 | |||||||||
| -0.851 MPa | 26.18 ± 0.05** | 221.97 ± 0.3** | 14.71 ± 0.02** | 421.13 ± 0.40** | 60.00 ± 0.04** | 50.00 ± 0.05** | 1.67 ± 0.04 | 25.00 ± 0.05 | |||||||||
| -1.619 MPa | 28.00 ± 0.08** | 245.00 ± 0.35** | 15.67 ± 0.03** | 442.88 ± 0.51** | 45.08 ± 0.04** | 63.17 ± 0.03** | 2.22 ± 0.03** | 16.67 ± 0.03** | |||||||||
| Sitabhog | Control | 19.45 ± 0.16 | 387.89 ± 0.33 | 10.06 ± 0.04 | 198.75 ± 0.36 | 23.23 ± 0.07 | 71.01 ± 0.09 | 4.30 ± 0.07 | 9.00 ± 0.06 | ||||||||
| -0.344 MPa | 18.23 ± 0.20** | 565.58 ± 0.49** | 11.95 ± 0.05** | 199.88 ± 0.43* | 23.43 ± 0.06** | 64.97 ± 0.09** | 4.27 ± 0.08 | 8.00 ± 0.05** | |||||||||
| -0.851 MPa | 22.64 ± 0.15** | 565.66 ± 0.63** | 13.93 ± 0.06** | 229.13 ± 0.54** | 22.72 ± 0.06** | 50.68 ± 0.09** | 4.40 ± 0.06* | 5.60 ± 0.04** | |||||||||
| -1.619 MPa | 26.31 ± 0.22** | 423.47 ± 0.76** | 23.69 ± 0.08** | 303.75 ± 0.61** | 21.50 ± 0.07** | 41.19 ± 0.10** | 4.65 ± 0.08** | 4.83 ± 0.04** | |||||||||
| Badshabhog | Control | 23.33 ± 0.12 | 477.86 ± 0.15 | 9.47 ± 0.02 | 227.63 ± 0.54 | 22.39 ± 0.05 | 43.15 ± 0.07 | 4.47 ± 0.06 | 6.25 ± 0.05 | ||||||||
| -0.344 MPa | 27.15 ± 0.15** | 497.44 ± 0.33** | 9.95 ± 0.04** | 239.25 ± 0.51** | 24.45 ± 0.04** | 44.14 ± 0.07** | 4.09 ± 0.05** | 5.00 ± 0.03** | |||||||||
| -0.851 MPa | 26.04 ± 0.18** | 552.92 ± 0.38** | 10.91 ± 0.05** | 311.63 ± 0.49** | 22.26 ± 0.04 | 41.82 ± 0.03** | 4.49 ± 0.05 | 6.25 ± 0.04 | |||||||||
| -1.619 MPa | 24.62 ± 0.19* | 823.94 ± 0.46** | 14.18 ± 0.06** | 312.75 ± 0.30** | 22.10 ± 0.04* | 36.74 ± 0.05** | 4.53 ± 0.05 | 3.83 ± 0.04** | |||||||||
| [1] | Abbas S R, Ahmad S D, Sabir S M, Shah A H. 2014. Detection of drought tolerant sugarcane genotypes ( Saccharum officinarum) using lipid peroxidation, antioxidant activity, glycine-betaine and proline contents. J Soil Sci Plant Nutr, 14(1): 233-243. |
| [2] | Allison L E, Bernstein L, Bower C A, Brown J W, Fireman M, Hatcher J T, Hayward H E, Pearson G A, Reeve R C, Richards L A, Wilcox L V. 1954. Diagnosis and improvement of saline and alkali soils. In: Richards L A. Agriculture Handbook No. 60, Washington, United States: Department of Agriculture: 96. |
| [3] | Ashraf M, Foolad M R. 2007. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot, 59(2): 206-216. |
| [4] | Basu S, Roychoudhury A, Saha P P, Sengupta D N. 2010. Differential antioxidative responses of indica rice cultivars to drought stress. Plant Growth Regul, 60(1): 51-59. |
| [5] | Bates L S, Waldren R P, Teare I D. 1973. Rapid determination of free proline for water stress studies. Plant Soil, 39: 205-207. |
| [6] | Bhattacharjee S. 2008. Calcium-dependent signaling pathway in the heat-induced oxidative injury in Amaranthus lividus. Biol Plantarum, 52: 137. |
| [7] | Bhattacharjee S, Dey N. 2018. Redox metabolic and molecular parameters for screening drought tolerant indigenous aromatic rice cultivars. Physiol Mol Biol Plants, 24(1): 7-23. |
| [8] | Boyer J S. 1982. Plant productivity and environment. Science, 218: 443-448. |
| [9] | Brestic M, Zivcak M, Olsovska K, Kalaji H M, Shao H B, Hakeem K R. 2014. Heat signaling and stress responses in photosynthesis. In: Hakeem K R, Rehman R, Tahir I. Plant Signalling: Understanding the Molecular Cross-Talk. New Delhi: Springer: 241-256. |
| [10] | Buege J A, Aust S D. 1978. Microsomal lipid peroxidation. Method Enzymol, 52: 302-310. |
| [11] | Cao G H, Sofic E, Prior R L. 1997. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic Biol Med, 22(5): 749-760. |
| [12] | Chakraborty A, Bhattacharjee S. 2015. Differential competence of redox-regulatory mechanism under extremes of temperature determines growth performances and cross tolerance in two indica rice cultivars. J Plant Physiol, 176: 65-77. |
| [13] | Chakrabarty A, Banik N, Bhattacharjee S. 2019. Redox-regulation of germination during imbibitional oxidative and chilling stress in an indica rice cultivar(Oryza sativa L., Cultivar Ratna). Physiol Mol Biol Plants, 25(3): 649-665. |
| [14] | Chaves M M, Oliveira M M. 2004. Mechanisms underlying plant resilience to water deficits: Prospects for water-saving agriculture. J Exp Bot, 55: 2365-2384. |
| [15] | Chen T H H, Murata N. 2011. Glycinebetaine protects plants against abiotic stress: Mechanisms and biotechnological applications. Plant Cell Environ, 34(1): 1-20. |
| [16] | De Ronde J A, Cress W A, Krüger G H, Strasser R J, van Staden J. 2004. Photosynthetic response of transgenic soybean plants, containing an Arabidopsis P5CR gene, during heat and drought stress. J Plant Physiol, 161(11): 1211-1224. |
| [17] | Deb D. 2005. Seeds of Tradition, Seeds of Future: Folk Rice Varieties from East India. New Delhi: Research Foundation for Science Technology & Ecology: 12-14. |
| [18] | Devasagayam T P A, Boloor K K, Ramasarma T. 2003. Methods for estimating lipid peroxidation: An analysis of merits and demerits. Ind J Biochem Biophyiol, 40(5): 300-308. |
| [19] | Fageria N K. 2007. Yield physiology of rice. J Plant Nut, 30: 843-879. |
| [20] | Faize M, Burgos L, Faize L, Piqueras A, Nicolas E, Barba-Espin G, Clemente-Moreno M J, Alcobendas R, Artlip T, Hernandez J A. 2011. Involvement of cytosolic ascorbate peroxidase and Cu/Zn- superoxide dismutase for improved tolerance against drought. J Exp Bot, 62(8): 2599-2613. |
| [21] | Farooq M, Basra S M A, Wahid A, Cheema Z A, Cheema M A, Khaliq A. 2008. Physiological role of exogenously applied glycinebetaine to improve drought tolerance in fine grain aromatic rice ( Oryza sativa L.). J Agron Crop Sci, 194(5): 325-333. |
| [22] | Grieve C M, Grattan S R. 1983. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil, 70: 303-307. |
| [23] | Gupta K, Dey A, Gupta B. 2013. Plant polyamines in abiotic stress responses. Acta Physiol Plant, 35: 2015-2036. |
| [24] | Gusakov A V, Kondratyeva E G, Sinitsyn A P. 2011. Comparison of two methods for assaying reducing sugars in the determination of carbohydrase activities. Int J Anal Chem, 2011: 283658. |
| [25] | Hasanuzzaman M, Nahar K, Alam M M, Roychowdhury R, Fujita M. 2013. Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int J Mol Sci, 14(5): 9643-9684. |
| [26] | Hasanuzzaman M, Alam M M, Rahman A, Hasanuzzaman M, Nahar K, Fujita M. 2014. Exogenous proline and glycine betaine mediated upregulation of antioxidant defense and glyoxalase systems provides better protection against salt-induced oxidative stress in two rice ( Oryza sativa L.) varieties. Biomed Res Int, 2014: 757219. |
| [27] | Hayzer D J, Leisinger T. 1980. The gene-enzyme relationships of proline biosynthesis in Escherichia coli. J Gen Microbiol, 118(2): 287-293. |
| [28] | Heath R L, Packer L. 1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophysiol, 125(1): 189-198. |
| [29] | Hefny M, Abdel-Kader D Z. 2009. Antioxidant-enzyme system as selection criteria for salt tolerance in forage sorghum genotypes (Sorghum bicolor L. Moench). In: Ashraf M, Ozturk M, Athar H R. Salinity and Water Stress. The Netherlands: Springer: 25-36. |
| [30] | Hisyam B, Alam M A, Naimah N, Jahan M S. 2017. Roles of glycinebetaine on antioxidants and gene function in rice plants under water stress. Asian J Plant Sci, 16: 132-140. |
| [31] | Holmstrӧm K O, Somersalo S, Mandal A, Palva T E, Welin B. 2000. Improved tolerance to salinity and low temperature in transgenic tobacco producing glycinebetaine. J Exp Bot, 51: 177-185. |
| [32] | Hong Z, Lakkineni K, Zhang Z, Verma DP. 2000. Removal of feedback inhibition of delta(1)-pyrroline-5 carboxylate synthetase results in increased proline accumulation and protection of plants from osmotic stress. Plant Physiol, 122(4): 1129-1136. |
| [33] | Hossain M A, Fujita M. 2010. Evidence for a role of exogenous glycinebetaine and proline in antioxidant defense and methylglyoxal detoxification systems in mung bean seedlings under salt stress. Physiol Mol Biol Plant, 16: 19-29. |
| [34] | Hossain M A, Mostofa G M, Fujita M. 2013. Heat-shock positively modulates oxidative protection of salt and drought-stressed mustard ( Brassica campestris L.) seedlings. J Plant Sci Mol Breeding, 2: 1-13. |
| [35] | Jagadish S V K, Craufurd P Q, Wheeler T R. 2007. High temperature stress and spikelet fertility in rice ( Oryza sativa L.). J Exp Bot, 58(7): 1627-1635. |
| [36] | Janska A, Marsik P, Zelenkova S, Ovesna J. 2010. Cold stress and acclimation-what is important for metabolic adjustment? Plant Biol, 12(3): 395-405. |
| [37] | Kader M A. 2005. A comparison of seed germination calculation formulae and the association interpretation of resulting data. J P Royal Soc New South Wales, 138: 65-75. |
| [38] | Kaur N, Sharma I, Kirat K, Pati P K. 2016. Detection of reactive oxygen species in Oryza sativa L.(rice). Bio-protocol, 6(24): 1-9. |
| [39] | Kaya C, Ashraf M, Dikilitas M, Tuna A L. 2013. Alleviation of salt stress-induced adverse effects on maize plants by exogenous application of indoleacetic acid (IAA) and inorganic nutrients: A field trial. Aust J Crop Sci, 7: 249-254. |
| [40] | Khan M S, Ahmad D, Khan M A. 2015. Utilization of genes encoding osmoprotectants in transgenic plants for enhanced abiotic stress tolerance. Electronic J Biotech, 18(4): 257-266. |
| [41] | Kishor P B K, Hong Z, Miao G H, Hu C A A, Verma D P S. 1995. Overexpression of Δ1-pyrroline-5-carboxylate synthetase increases proline production and confers osmotolerance in transgenic plants. Plant Physiol, 108(4): 1387-1394. |
| [42] | Koyro H W, Ahmad P, Geissler N. 2012. Abiotic stress responses in plants: An Overview. In: Ahmad P, Prasad M N V. Environmental Adaptations and Stress Tolerance of Plants in the Era of Climate Change. New York, Springer: 1-28. |
| [43] | Kumar M, Bijo A J, Baghel R S, Reddy C R K, Jha B. 2012. Selenium and spermine alleviates cadmium induced toxicity in the red seaweed Gracilaria dura by regulating antioxidant system and DNA methylation. Plant Physiol Biochem, 51: 129-138. |
| [44] | Lang-Mladek C, Popova O, Kiok K, Berlinger M, Rakic B, Aufsatz W, Jonak C, Hauser M T, Luschnig C. 2010. Transgenerational inheritance and resetting of stress-induced loss of epigenetic gene silencing in Arabidopsis. Mol Plant, 3(3): 594-602. |
| [45] | Lesk C, Rowhani P, Ramankutty N. 2016. Influence of extreme weather disasters on global crop production. Nature, 529: 84-87. |
| [46] | Lin D R, Xiao M S, Zhao J J, Li Z H, Xing B S, Li X D, Kong M Z, Li L Y, Zhang Q, Liu Y W, Chen H, Qin W, Wu H J, Chen S Y. 2016. An overview of plant phenolic compounds and their importance in human nutrition and management of type 2 diabetes. Molecules, 21(10): 1374. |
| [47] | Liu J, Zhang F, Zhou J J, Chen F, Wang B S, Xie X Z. 2012. Phytochrome B control of total leaf area and stomatal density affects drought tolerance in rice. Plant Mol Biol, 78(3): 289-300. |
| [48] | Liu X Z, Huang B R. 2000. Carbohydrate accumulation in relation to heat stress tolerance in two creeping Bentgrass cultivars. J Amer Soc Hort Sci, 125(4): 442-447. |
| [49] | Livingston D P, Hincha D K, Heyer A G. 2009. Fructan and its relationship to abiotic stress tolerance in plants. Cell Mol Life Sci, 66(13): 2007-2023. |
| [50] | MacNevin W M, Uron P F. 1953. Spectrum of hydrogen peroxide from organic hydroperox-ides. Anal Chem, 25: 1760-1761. |
| [51] | McCready R M, Guggolz J, Silviera V, Owens H S. 1950. Determination of starch and amylose in vegetables. Anal Chem, 22(9): 1156-1158. |
| [52] | Mensor L L, Menezes F S, Leitao G G, Reis A S, dos Santos T C, Coube C S, Leitao S G. 2001. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother Res, 15(2): 127-130. |
| [53] | Mir R R, Zaman-Allah M, Sreenivasulu N, Trethowan R, Varshney R K. 2012. Integrated genomics, physiology and breeding approaches for improving drought tolerance in crops. Theor Appl Genet, 125(4): 625-645. |
| [54] | Mishra A, Kumar S, Pandey A K. 2013. Scientific validation of the medicinal efficacy of Tinospora cordifolia. Scient World J, 2013: 292934. |
| [55] | Nikolaeva M K, Maevskaya S N, Voronin P Y. 2015. Activities of antioxidant and osmoprotective systems and photosynthetic gas exchange in maize seedlings under drought conditions. Russ J Plant Physiol, 62: 314-321. |
| [56] | Nounjana N, Nghiab P T, Theerakulpisuta P. 2012. Exogenous proline and trehalose promote recovery of rice seedlings from salt-stress and differentially modulate antioxidant enzymes and expression of related genes. J Plant Physiol, 169(6): 596-604. |
| [57] | Nourooz-Zadeh J, Tajaddini-Sarmadi J, Wolff S P. 1994. Measurement of plasma hydroperoxide concentrations by the ferrous oxidation-xylenol orange assay in conjunction with triphenylphosphine. Anal Biochem, 220(2): 403-409. |
| [58] | Pandey A K, Mishra A K, Mishra A. 2012. Antifungal and antioxidative potential of oil and extracts derived from leaves of Indian spice plant Cinnamomum tamala. Cell Mol Biol, 58(1): 142-147. |
| [59] | Parida A K, Dagaonkar V S, Phalak M S, Aurangabadkar L P. 2008. Differential responses of the enzymes involved in proline biosynthesis and degradation in drought tolerant and sensitive cotton genotypes during drought stress and recovery. Acta Physiol Planta, 30: 619-627. |
| [60] | Park E J, Jeknic Z, Sakamoto A, DeNoma J, Yuwansiri R, Murata N, Chen T H. 2004. Genetic engineering of glycinebetaine synthesis in tomato protects seeds, plants, and flowers from chilling damage. Plant J, 40(4): 474-487. |
| [61] | Park E J, Jeknic´ Z, Pino M T, Murata N, Chen T H. 2007. Glycinebetaine accumulation is more effective in chloroplasts than in the cytosol for protecting transgenic tomato plants against abiotic stress. Plant Cell Environ, 30(8): 994-1005. |
| [62] | Peng Z, Lu Q, Verma D P S. 1996. Reciprocal regulation of Δ1-pyrroline-5-carboxylate synthetase and proline dehydrogenase genes controls proline levels during and after osmotic stress in plants. Mol Gen Genet, 253(3): 334-341. |
| [63] | Peterman T K, Siedow J N. 1985. Behavior of lipoxygenase during establishment, senescence, and rejuvenation of soybean cotyledons. Plant Physiol, 78(4): 690-695. |
| [64] | Petrov V, Hille J, Mueller-Roeber B, Gechev T S. 2015. ROS- mediatedabiotic stress-induced programmed cell death in plants. Front Plant Sci, 6: 69. |
| [65] | Quan N T, Xuan T D. 2018. Foliar application of vanillic and p- hydroxybenzoic acids enhanced drought tolerance and formation of phytoalexinmomilactones in rice. Arch Agron Soil Sci, 64(13): 1831-1846. |
| [66] | Ramesh R, Ramesh T, Rao P R, Shankar V G, Bhave M H V. 2017. High temperature stress effected the biochemical parameters of rice (Oryza sativa L.) varieties and hybrids. Int J Pure App Biosc, 5(4): 1478-1490. |
| [67] | Ranganayakulu G S, Veeranagamallaiah G, Sudhakar C. 2013. Effect of salt stress on osmolyte accumulation in two groundnut cultivars ( Arachis hypogaea L.) with contrasting salt tolerance. Afr J Plant Sci, 7: 586-592. |
| [68] | Rasheed R, Wahid A, Farooq M, Hussain I, Basra S M A. 2011. Role of proline and glycine betaine pretreatments in improving heat tolerance of sprouting sugarcane ( Saccharum sp.) buds. Plant Growth Regul, 65: 35-45. |
| [69] | Rena A B, Splittstoesser W E. 1975. Proline dehydrogenase and pyrroline-5-carboxylate reductase from pumpkin cotyledons. Phytochemistry, 14(3): 657-661. |
| [70] | Rhodes D, Samaras Y. 1994. Genetic control of osmoregulation in plants. In: Strange K. Cellular and Molecular Physiology of Cell Volume Regulation. Boca Raton: CRC Press: 347-367. |
| [71] | Rice-Evans C, Miller N, Paganga G. 1997. Antioxidant properties of phenolic compounds. Trends Plant Sci, 2(4): 152-159. |
| [72] | Rizhsky L, Liang H J, Shuman J, Shulaev V, Davletova S, Mittler R. 2004. When defense pathways collide: The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol, 134(4): 1683-1696. |
| [73] | Rubio-Casal A E, Castillo J M, Luque C J, Figueroa E M. 2003. Influence of salinity on germination and seeds viability of two primary colonizers of Mediterranean salt pans. J Arid Environ, 53(2): 145-154. |
| [74] | Sahebi M, Hanafi M M, Rafii M Y, Mahmud T M M, Azizi P, Osman M, Abiri R, Taheri S, Kalhori N, Shabanimofrad M, Miah G, Atabaki N. 2018. Improvement of drought tolerance in rice ( Oryza sativa L.): Genetics, genomic tools, and the WRKY gene family. Bio Med Res Int, 2018: 3158474. |
| [75] | Sakamoto A, Murata A N. 1998. Metabolic engineering of rice leading to biosynthesis of glycinebetaine and tolerance to salt and cold. Plant Mol Biol, 38: 1011-1019. |
| [76] | Sakamoto A, Murata N. 2000. Genetic engineering of glycinebetaine synthesis in plants: Current status and implications for enhancement of stress tolerance. J Exp Bot, 51(342): 81-88. |
| [77] | Samarah N H. 2016. Understanding how plants respond to drought stress at the molecular and whole plant levels. In: Hossain M A, Wani S H, Bhattacharjee S, Burritt D J, Phan Tran L S. Drought Stress Tolerance in Plants. Switzerland: Springer Nature: 1-37. |
| [78] | Silvente S, Sobolev A P, Lara M. 2012. Metabolite adjustments in drought tolerant and sensitive soybean genotypes in response to water stress. PLoS One, 7(6): e38554. |
| [79] | Swapna S, Shylaraj K S. 2017. Screening for osmotic stress responses in rice varieties under drought condition. Rice Sci, 24(5): 253-263. |
| [80] | Simontacchi M, Caro A, Fraga C G, Puntarulo S. 1993. Oxidative stress affects-tocopherol content in soyabean embryonic axes upon imbibitions. Plant Physiol, 103(3): 949-953. |
| [81] | Singh S, Srivastava P K, Kumar D, Tripathi D K, Chauhan D K, Prasad S M. 2015. Morpho-anatomical and biochemical adapting strategies of maize ( Zea mays L.) seedlings against lead and chromium stresses. Biocatal Agric Biotechnol, 4(3): 286-295. |
| [82] | Smirnoff N, Cumes Q J. 1989. Hydroxyl radicals scavenging activity of compatible isolates. Phytochemistry, 28(4): 1057-1060. |
| [83] | Steffensena S K, Rinnan A, Mortenseng A G, Laursen B, de Troiani R M, Noellemeyer E J, Janovska D, Dusek K, Delano-Frier J, Taberner A, Christophersen C, Fomsgaard I S. 2011. Variations in the polyphenol content of seeds of field grown Amaranthus genotypes. Food Chem, 129(1): 131-138. |
| [84] | Sugio A, Dreos R, Aparicio F, Maule A J. 2009. The cytosolic protein response as a sub component of the wider heat shock response in Arabidopsis. Plant Cell, 21(2): 642-654. |
| [85] | Tang R S, Zheng J C, Jin Z Q, Zhang D D, Huang Y H, Chen L G. 2008. Possible correlation between high temperature induced floret sterility and endogenous levels of IAA, GAs and ABA in rice ( Oryza sativa L.). Plant Growth Regul, 54: 37-43. |
| [86] | Tari I, Kiss G, Deér A K, Csiszár J, Erdei L, Gallé Á, Gémes K, Horváth F, Poór P, Szepesi Á, Simon L M. 2010. Salicylic acid increased aldose reductase activity and sorbitol accumulation in tomato plants under salt stress. Biol Plant, 54: 677-683. |
| [87] | Tausz M, Sircelj H, Grill D. 2004. The glutathione system as a stress marker in plant ecophysiology: Is a stress-response concept valid? J Exp Bot, 55: 1955-1962. |
| [88] | Tietze F. 1969. Enzymatic method for quantitative determination of nanogram amounts of total and oxidised glutathione: Application to mammalian blood and other tissues. Anal Biochem, 27(3): 502-522. |
| [89] | Turkan I, Demiral T. 2009. Recent developments in understanding salinity tolerance. Environ Exp Bot, 67(1): 2-9. |
| [90] | Velderrain-Rodríguez G R, Palafox-Carlos H, Wall-Medrano A, Ayala-Zavala J F, Chen C Y O, Robles-Sanchez M, Astiazaran- García H, Alvarez-Parrilla E, González-Aguilar G A. 2014. Phenolic compounds: Their journey after intake. Food Funct, 5: 189-197. |
| [91] | Verbruggen N, Hermans C. 2008. Proline accumulation in plants: A review. Amino Acids, 35(4): 753-759. |
| [92] | Waditee R, Bhuiyan M N H, Rai V, Aoki K, Tanaka Y, Hibino T, Suzuki S, Takano J, Jagendorf A T, Takabe T, Takabe T,. 2005. Genes for direct methylation of glycine provide high levels of glycinebetaine and abiotic-stress tolerance in Synechococcus and Arabidopsis. Proc Natl Acad Sci USA, 102(5): 1318-1323. |
| [93] | Wahid A, Perveen M, Gelani S, Basra S M A. 2007. Pretreatment of seed with H2O2 improves salt tolerance of wheat seedlings by alleviation of oxidative damage and expression of stress proteins. J Plant Physiol, 164(3): 283-294. |
| [94] | Wani S H, Singh N B, Haribhushan A, Mir J I. 2013. Compatible solute engineering in plants for abiotic stress tolerance-role of glycine betaine. Curr Genomics, 14(3): 157-165. |
| [95] | Yamada M, Morishita H, Urano K, Shiozaki N, Yamaguchi- Shinozaki K, Shinozaki K, Yoshiba Y. 2005. Effects of free proline accumulation in petunias under drought stress. J Exp Bot, 56: 1975-1981. |
| [96] | Yang X L, Wang B F, Chen L, Li P, Cao C G. 2019. The different influences of drought stress at the flowering stage on rice physiological traits, grain yield, andquality. Sci Rep, 9: 3742. |
| [97] | Zagorchev L, Teofanova D, Odjakova M. 2016. Ascorbate-glutathione cycle: Controlling the redox environment for drought tolerance. In: Hossain M A, Wani S H, Bhattacharjee S, Burritt D J, Phan Tran L S. Drought Stress Tolerance in Plants. Springer International Publishing: 187-226. |
| [98] | Zhang N, Liu B L, Ma C Y, Zhang G D, Chang J, Si H J, Wang D. 2014. Transcriptome characterization and sequencing-based identification of drought-responsive genes in potato. Mol Biol Rep, 41(1): 505-517. |
| [99] | Zivcak M, Brestic M, Sytar O. 2016. Osmotic adjustment and plant adaptation to drought stress. In: Hossain M A, Wani S H, Bhattacharjee S, Burritt D J, Phan Tran L S. Drought Stress Tolerance in Plants. Springer International Publishing: 105-143. |
| [100] | Zu X F, Lu Y K, Wang Q Q, Chu P F, Miao W, Wang H Q, La H G. 2017. A new method for evaluating the drought tolerance of upland rice cultivars. Crop J, 5(6): 488-498. |
| [1] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [2] | Prafulla Kumar Behera, Debabrata Panda. Germplasm Resources, Genes and Perspective for Aromatic Rice [J]. Rice Science, 2023, 30(4): 294-305. |
| [3] | Liu Yantong, Li Ting, Jiang Zhishu, Zeng Chuihai, He Rong, Qiu Jiao, Lin Xiaoli, Peng Limei, Song Yongping, Zhou Dahu, Cai Yicong, Zhu Changlan, Fu Junru, He Haohua, Xu Jie. Characterization of a Novel Weak Allele of RGA1/D1 and Its Potential Application in Rice Breeding [J]. Rice Science, 2022, 29(6): 522-534. |
| [4] | Kossi Lorimpo Adjah, Maxwell Darko Asante, Aboubacar Toure, Mawuli Aziadekey, Francis Osei Amoako-Andoh, Michael Frei, Yacouba Diallo, Komi Agboka. Improvement of Rice Production under Drought Conditions in West Africa: Application of QTLs in Breeding for Drought Resistance [J]. Rice Science, 2022, 29(6): 512-521. |
| [5] | Gao Xiuying, Zhang Ye, Zhang Hongsheng, Huang Ji. A β-ketoacyl-CoA Synthase OsCUT1 Confers Increased Drought Tolerance in Rice [J]. Rice Science, 2022, 29(4): 353-362. |
| [6] | Liu Kai, Li Minjuan, Zhang Bin, Yin Xuming, Xia Xinjie, Wang Manling, Cui Yanchun. Poaceae Orthologs of Rice OsSGL, DUF1645 Domain-Containing Genes, Positively Regulate Drought Tolerance, Grain Length and Weight in Rice [J]. Rice Science, 2022, 29(3): 257-267. |
| [7] | Chen Eryong, Shen Bo. OsABT, a Rice WD40 Domain-Containing Protein, Is Involved in Abiotic Stress Tolerance [J]. Rice Science, 2022, 29(3): 247-256. |
| [8] | Muhammad Mahran Aslam, Muhammad Abdul Rehman Rashid, Mohammad Aquil Siddiqui, Muhammad Tahir Khan, Fozia Farhat, Shafquat Yasmeen, Imtiaz Ahmad Khan, Shameem Raja, Fatima Rasool, Mahboob Ali Sial, Zhao Yan. Recent Insights into Signaling Responses to Cope Drought Stress in Rice [J]. Rice Science, 2022, 29(2): 105-117. |
| [9] | Balija Vishalakshi, Bangale Umakanth, Ponnuvel Senguttuvel, Makarand Barbadikar Kalyani, Prasad Madamshetty Srinivas, Rao Durbha Sanjeeva, Yadla Hari, Madhav Maganti Sheshu. Improvement of Upland Rice Variety by Pyramiding Drought Tolerance QTL with Two Major Blast Resistance Genes for Sustainable Rice Production [J]. Rice Science, 2021, 28(5): 493-500. |
| [10] | Vera Jesus Da Costa Maria, Ramegowda Yamunarani, Ramegowda Venkategowda, N. Karaba Nataraja, M. Sreeman Sheshshayee, Udayakumar Makarla. Combined Drought and Heat Stress in Rice: Responses, Phenotyping and Strategies to Improve Tolerance [J]. Rice Science, 2021, 28(3): 233-242. |
| [11] | Silas Akos Ibrahim, Y. Rafii Mohd, Razi Ismail Mohd, Izan Ramlee Shairul, Abd Aziz Shamsudin Noraziyah, Ramli Asfaliza, Chibuike Chukwu Samuel, Swaray Senesie, Jalloh Momodu. Evaluation of Inherited Resistance Genes of Bacterial Leaf Blight, Blast and Drought Tolerance in Improved Rice Lines [J]. Rice Science, 2021, 28(3): 279-288. |
| [12] | Baoxiang Wang, Yan Liu, Yifeng Wang, Jingfang Li, Zhiguang Sun, Ming Chi, Yungao Xing, Bo Xu, Bo Yang, Jian Li, Jinbo Liu, Tingmu Chen, Zhaowei Fang, Baiguan Lu, Dayong Xu, Kazeem Bello Babatunde. OsbZIP72 Is Involved in Transcriptional Gene-Regulation Pathway of Abscisic Acid Signal Transduction by Activating Rice High-Affinity Potassium Transporter OsHKT1;1 [J]. Rice Science, 2021, 28(3): 257-267. |
| [13] | Panda Debabrata, Sakambari Mishra Swati, Kumar Behera Prafulla. Drought Tolerance in Rice: Focus on Recent Mechanisms and Approaches [J]. Rice Science, 2021, 28(2): 119-132. |
| [14] | Pandit Elssa, Kumar Panda Rajendra, Sahoo Auromeera, Ranjan Pani Dipti, Kumar Pradhan Sharat. Genetic Relationship and Structure Analysis of Root Growth Angle for Improvement of Drought Avoidance in Early and Mid-Early Maturing Rice Genotypes [J]. Rice Science, 2020, 27(2): 124-132. |
| [15] | Hossain Prodhan Zakaria, Qingyao Shu. Rice Aroma: A Natural Gift Comes with Price and the Way Forward [J]. Rice Science, 2020, 27(2): 86-100. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||