
Rice Science ›› 2023, Vol. 30 ›› Issue (2): 148-159.DOI: 10.1016/j.rsci.2023.01.007
• Research Paper • Previous Articles Next Articles
Jiratchaya Wisetkomolmat1, Chaiwat Arjin1, Surat Hongsibsong2, Warintorn Ruksiriwanich3,8, Chutamat Niwat4, Pimsiri Tiyayon5, Sansanee Jamjod6, Supapohn Yamuangmorn7, Chanakan Prom-U-Thai6,7, Korawan Sringarm1,7,8( )
)
Received:2022-05-29
Accepted:2022-11-03
Online:2023-03-28
Published:2023-01-16
Contact:
Korawan Sringarm (korawan.s@cmu.ac.th)
Jiratchaya Wisetkomolmat, Chaiwat Arjin, Surat Hongsibsong, Warintorn Ruksiriwanich, Chutamat Niwat, Pimsiri Tiyayon, Sansanee Jamjod, Supapohn Yamuangmorn, Chanakan Prom-U-Thai, Korawan Sringarm. Antioxidant Activities and Characterization of Polyphenols from Selected Northern Thai Rice Husks: Relation with Seed Attributes[J]. Rice Science, 2023, 30(2): 148-159.
Add to citation manager EndNote|Ris|BibTeX
| Variety name | Abbreviation | Pericarp color | Husk color | Ecotype | Endosperm type | Special quality | Yield (%) |
|---|---|---|---|---|---|---|---|
| Buebang 3 CMU | BB3CMU | White | Straw | Upland | Non-glutinous rice | Vitamin E | 2.09 |
| Buebang 4 CMU | BB4CMU | White | Straw | Upland | Non-glutinous rice | Zinc | 2 |
| Bien Koo 5 CMU | BKU5CMU | Purple | Straw | Upland | Non-glutinous rice | Iron | 1.71 |
| K 4 CMU | K4CMU | Purple | Purple | Wetland | Non-glutinous rice | Antioxidant | 2.1 |
| Kum Akha 1 CMU | KAK1CMU | Purple | Straw | Upland | Glutinous rice | Antioxidant | 2.5 |
| Kum Doi Saket | KDK | Purple | Purple | Wetland | Glutinous rice | Gamma oryzanol | 1.91 |
| Khao Dawk Mali 105 | KDML105 | White | Straw | Wetland | Non-glutinous rice | Aroma | 1.7 |
| Kum Chao Morchor 107 | KCCMU107 | Purple | Straw | Wetland | Non-glutinous rice | Antioxidant | 1.99 |
| Sang 5 CMU | Sang5CMU | Purple | Straw with brown line | Upland | Glutinous rice | Phenol | 1.79 |
| Pieisu 1 CMU | PES1CMU | Purple | Straw with brown line | Upland | Glutinous rice | Anthocyanin | 2.44 |
| Rice Department 6 | RD6 | White | Straw | Wetland | Glutinous rice | Aroma | 2.25 |
| Yamuechaebia 3 CMU | YM3CMU | Red | Straw | Upland | Non-glutinous rice | Gamma oryzanol | 2.14 |
Table 1. Rice varieties with special grain quality.
| Variety name | Abbreviation | Pericarp color | Husk color | Ecotype | Endosperm type | Special quality | Yield (%) |
|---|---|---|---|---|---|---|---|
| Buebang 3 CMU | BB3CMU | White | Straw | Upland | Non-glutinous rice | Vitamin E | 2.09 |
| Buebang 4 CMU | BB4CMU | White | Straw | Upland | Non-glutinous rice | Zinc | 2 |
| Bien Koo 5 CMU | BKU5CMU | Purple | Straw | Upland | Non-glutinous rice | Iron | 1.71 |
| K 4 CMU | K4CMU | Purple | Purple | Wetland | Non-glutinous rice | Antioxidant | 2.1 |
| Kum Akha 1 CMU | KAK1CMU | Purple | Straw | Upland | Glutinous rice | Antioxidant | 2.5 |
| Kum Doi Saket | KDK | Purple | Purple | Wetland | Glutinous rice | Gamma oryzanol | 1.91 |
| Khao Dawk Mali 105 | KDML105 | White | Straw | Wetland | Non-glutinous rice | Aroma | 1.7 |
| Kum Chao Morchor 107 | KCCMU107 | Purple | Straw | Wetland | Non-glutinous rice | Antioxidant | 1.99 |
| Sang 5 CMU | Sang5CMU | Purple | Straw with brown line | Upland | Glutinous rice | Phenol | 1.79 |
| Pieisu 1 CMU | PES1CMU | Purple | Straw with brown line | Upland | Glutinous rice | Anthocyanin | 2.44 |
| Rice Department 6 | RD6 | White | Straw | Wetland | Glutinous rice | Aroma | 2.25 |
| Yamuechaebia 3 CMU | YM3CMU | Red | Straw | Upland | Non-glutinous rice | Gamma oryzanol | 2.14 |
| Variety | ABTS | DPPH | FRAP | Total phenolic content | Total flavonoid content |
|---|---|---|---|---|---|
| [mmol/(L·g) TE] | [mmol/(L·g) TE] | [mmol/(L·g) Fe2+] | (mg/g GE) | (mg/g CE) | |
| BB3CMU | 252.34 ± 3.90 h | 4.39 ± 0.77 b | 0.46 ± 0.09 de | 16.53 ± 1.83 d | 4.58 ± 0.00 cd |
| BB4CMU | 385.95 ± 6.18 f | 3.24 ± 0.15 d | 0.53 ± 0.15 bcd | 15.86 ± 0.34 d | 4.01 ± 0.13 cd |
| BKU5CMU | 159.06 ± 6.17 j | 2.55 ± 0.11 fgh | 0.31 ± 0.07 f | 10.63 ± 1.00 f | 3.78 ± 0.08 cd |
| K4CMU | 515.79 ± 4.14 c | 4.81 ± 0.25 a | 0.60 ± 0.12 bc | 21.90 ± 1.18 b | 8.37 ± 0.65 b |
| KAK1CMU | 409.90 ± 21.26 e | 3.82 ± 0.25 c | 0.63 ± 0.10 b | 20.18 ± 0.54 bc | 9.04 ± 0.32 b |
| KDK | 436.37 ± 11.72 d | 2.77 ± 0.15 efg | 0.60 ± 0.11 bc | 20.79 ± 2.11 b | 5.78 ± 4.02 c |
| KDML105 | 359.48 ± 5.69 g | 2.23 ± 0.06 h | 0.47 ± 0.08 cde | 15.38 ± 0.94 de | 4.36 ± 0.09 cd |
| KCCMU107 | 191.83 ± 3.90 i | 3.01 ± 0.33 de | 0.39 ± 0.08 ef | 14.75 ± 1.16 de | 3.29 ± 0.09 d |
| Sang5CMU | 546.04 ± 10.43 b | 2.90 ± 0.17 def | 0.53 ± 0.09 bcd | 18.66 ± 0.80 c | 4.68 ± 3.27 cd |
| PES1CMU | 679.66 ± 19.96 a | 4.16 ± 0.18 b | 0.87 ± 0.16 a | 29.90 ± 3.52 a | 12.16 ± 3.51 a |
| RD6 | 164.10 ± 24.85 j | 2.64 ± 0.24 fg | 0.43 ± 0.10 def | 13.56 ± 0.98 e | 3.36 ± 0.34 cd |
| YM3CMU | 166.62 ± 12.35 j | 2.41 ± 0.12 gh | 0.37 ± 0.10 ef | 11.39 ± 0.32 f | 3.36 ± 0.54 cd |
Table 2. Antioxidant activities, total phenolic and total flavonoid contents of rice husk extracts.
| Variety | ABTS | DPPH | FRAP | Total phenolic content | Total flavonoid content |
|---|---|---|---|---|---|
| [mmol/(L·g) TE] | [mmol/(L·g) TE] | [mmol/(L·g) Fe2+] | (mg/g GE) | (mg/g CE) | |
| BB3CMU | 252.34 ± 3.90 h | 4.39 ± 0.77 b | 0.46 ± 0.09 de | 16.53 ± 1.83 d | 4.58 ± 0.00 cd |
| BB4CMU | 385.95 ± 6.18 f | 3.24 ± 0.15 d | 0.53 ± 0.15 bcd | 15.86 ± 0.34 d | 4.01 ± 0.13 cd |
| BKU5CMU | 159.06 ± 6.17 j | 2.55 ± 0.11 fgh | 0.31 ± 0.07 f | 10.63 ± 1.00 f | 3.78 ± 0.08 cd |
| K4CMU | 515.79 ± 4.14 c | 4.81 ± 0.25 a | 0.60 ± 0.12 bc | 21.90 ± 1.18 b | 8.37 ± 0.65 b |
| KAK1CMU | 409.90 ± 21.26 e | 3.82 ± 0.25 c | 0.63 ± 0.10 b | 20.18 ± 0.54 bc | 9.04 ± 0.32 b |
| KDK | 436.37 ± 11.72 d | 2.77 ± 0.15 efg | 0.60 ± 0.11 bc | 20.79 ± 2.11 b | 5.78 ± 4.02 c |
| KDML105 | 359.48 ± 5.69 g | 2.23 ± 0.06 h | 0.47 ± 0.08 cde | 15.38 ± 0.94 de | 4.36 ± 0.09 cd |
| KCCMU107 | 191.83 ± 3.90 i | 3.01 ± 0.33 de | 0.39 ± 0.08 ef | 14.75 ± 1.16 de | 3.29 ± 0.09 d |
| Sang5CMU | 546.04 ± 10.43 b | 2.90 ± 0.17 def | 0.53 ± 0.09 bcd | 18.66 ± 0.80 c | 4.68 ± 3.27 cd |
| PES1CMU | 679.66 ± 19.96 a | 4.16 ± 0.18 b | 0.87 ± 0.16 a | 29.90 ± 3.52 a | 12.16 ± 3.51 a |
| RD6 | 164.10 ± 24.85 j | 2.64 ± 0.24 fg | 0.43 ± 0.10 def | 13.56 ± 0.98 e | 3.36 ± 0.34 cd |
| YM3CMU | 166.62 ± 12.35 j | 2.41 ± 0.12 gh | 0.37 ± 0.10 ef | 11.39 ± 0.32 f | 3.36 ± 0.54 cd |
| Variable | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| ABTS | 0.441 | 0.427 | -0.645 | -0.45 |
| DPPH | 0.377 | -0.872 | -0.294 | -0.02 |
| FRAP | 0.472 | 0.213 | 0.15 | 0.554 |
| Flavonoid | 0.461 | -0.021 | 0.689 | -0.559 |
| Phenolic | 0.478 | 0.105 | 0.015 | 0.422 |
Table 3. Eigen analysis of the loadings of significant principal components (PCs) for antioxidants, total phenolic, and total flavonoid contents in rice husk samples.
| Variable | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| ABTS | 0.441 | 0.427 | -0.645 | -0.45 |
| DPPH | 0.377 | -0.872 | -0.294 | -0.02 |
| FRAP | 0.472 | 0.213 | 0.15 | 0.554 |
| Flavonoid | 0.461 | -0.021 | 0.689 | -0.559 |
| Phenolic | 0.478 | 0.105 | 0.015 | 0.422 |
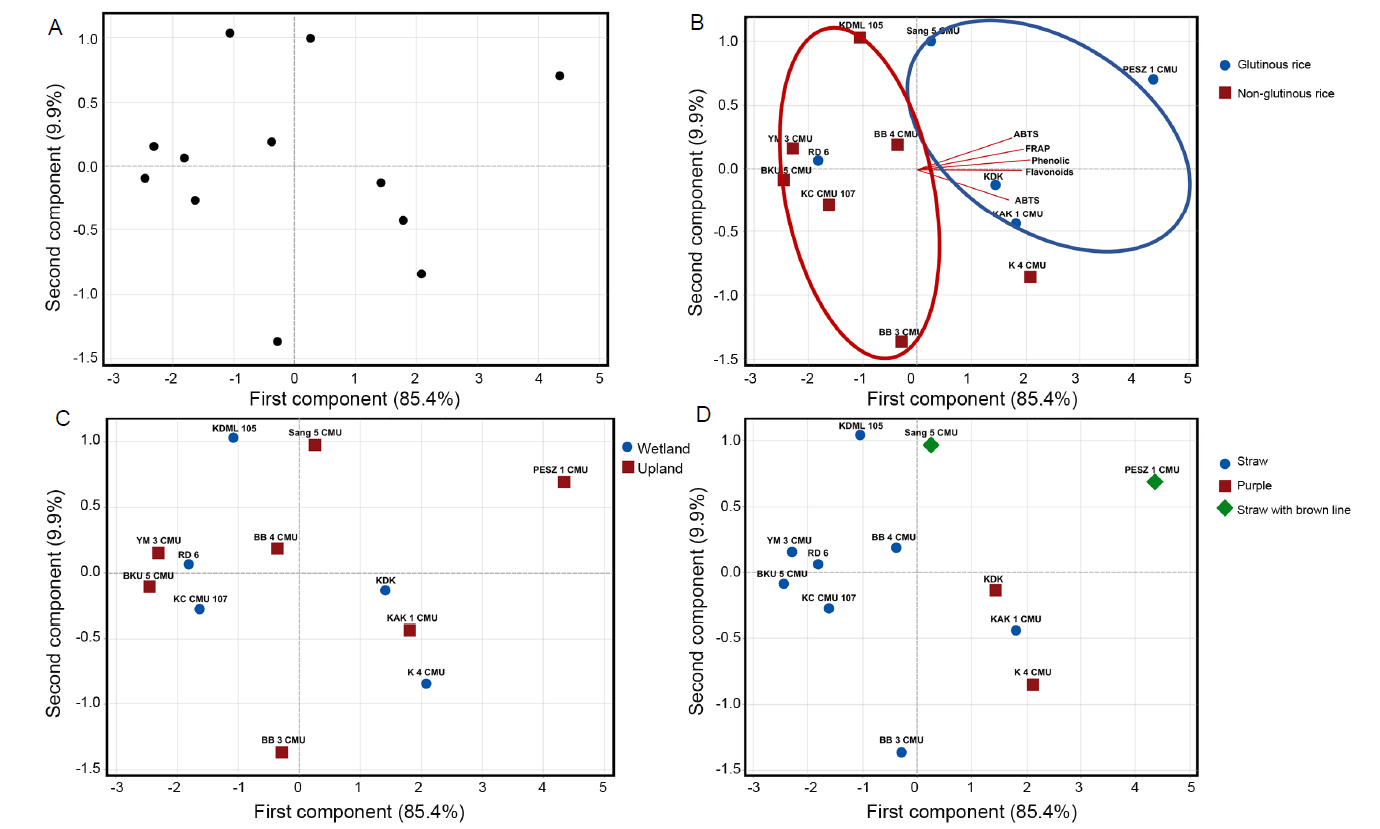
Fig. 2. Principle component analysis (PCA) of antioxidant activity, total phenolic content, and total flavonoid contents in rice husk. A, Score plot of antioxidant activity, total phenolic content and total flavonoid content. B, Biplot acclimations labeled as endosperm type. C, Score plot acclimations labeled as ecotype. D, Score plot acclimations labeled as husk color.
| Variable | DPPH | FRAP | TFC | Total phenolic content |
|---|---|---|---|---|
| ABTS | 0.497 | 0.907** | 0.795** | 0.909** |
| DPPH | 0.583* | 0.671* | 0.643* | |
| FRAP | 0.913** | 0.977** | ||
| TFC | 0.914** |
Table 4. Pearson correlation analysis of antioxidants, total phenolic, and total flavonoid contents in rice husks categorized by endosperm type.
| Variable | DPPH | FRAP | TFC | Total phenolic content |
|---|---|---|---|---|
| ABTS | 0.497 | 0.907** | 0.795** | 0.909** |
| DPPH | 0.583* | 0.671* | 0.643* | |
| FRAP | 0.913** | 0.977** | ||
| TFC | 0.914** |
| Variety | Caffeic acid | EG | o-coumaric acid | Quercetin | Phytic acid | Ferulic acid | CA | Kaempferol | HA |
|---|---|---|---|---|---|---|---|---|---|
| BB3CMU | 0.21 ± 0.02 de | 0.35 ± 0.00 h | 1.95 ± 0.08 cdef | 0.25 ± 0.00 ef | 15.45 ± 0.09 g | 0.34 ± 0.02 fg | 0.33 ± 0.14 c | 1.79 ± 0.01 e | 0.40 ± 0.02 f |
| BB4CMU | 0.25 ± 0.01 d | 0.45 ± 0.02 fg | 2.81 ± 0.01 a | 0.24 ± 0.01 f | 21.79 ± 0.07 e | 1.36 ± 0.15 a | 0.35 ± 0.12 c | 0.75 ± 0.01 g | 0.62 ± 0.04 c |
| BKU5CMU | 0.16 ± 0.02 f | 0.40 ± 0.01 gh | 2.14 ± 0.67 bcd | 0.25 ± 0.01 ef | 5.68 ± 0.24 h | 0.39 ± 0.04 fg | 0.23 ± 0.07 c | 1.25 ± 0.20 f | 0.60 ± 0.04 ed |
| K4CMU | 0.36 ± 0.01 b | 0.82 ± 0.03 e | 1.54 ± 0.08 fg | 0.61 ± 0.01 b | 31.40 ± 0.48 d | 0.32 ± 0.01 g | 2.05 ± 0.25 a | 3.63 ± 0.16 a | 1.07 ± 0.03 a |
| KAK1CMU | 0.18 ± 0.06 ef | 1.45 ± 0.00 c | 2.44 ± 0.07 abc | 0.46 ± 0.01 c | 14.75 ± 0.44 g | 0.43 ± 0.00 efg | 0.81 ± 0.03 b | 0.56 ± 0.06 h | 0.55 ± 0.02 de |
| KDK | 0.16 ± 0.01 ef | 0.15 ± 0.00 i | 0.37 ± 0.01 i | 0.27 ± 0.00 de | 6.99 ± 0.03 h | 0.18 ± 0.00 h | 0.15 ± 0.00 c | 0.32 ± 0.00 i | 0.23 ± 0.01 g |
| KDML105 | 0.40 ± 0.01 ab | 0.50 ± 0.02 f | 1.00 ± 0.03 h | 0.18 ± 0.00 g | 6.57 ± 0.13 h | 0.60 ± 0.04 d | 0.80 ± 0.07 b | 2.92 ± 0.03 c | 0.40 ± 0.01 f |
| KCCMU107 | 0.30 ± 0.00 c | 0.17 ± 0.02 i | 1.14 ± 0.01 gh | 0.24 ± 0.01 f | 67.36 ± 0.86 c | 0.52 ± 0.02 de | 0.25 ± 0.11 c | 2.47 ± 0.02 d | 0.54 ± 0.03 de |
| Sang5CMU | 0.20 ± 0.03 def | 2.80 ± 0.04 a | 2.49 ± 0.04 ab | 0.59 ± 0.03 b | 19.42 ± 0.34 f | 0.76 ± 0.04 bc | 1.90 ± 0.10 a | 0.13 ± 0.00 j | 0.53 ± 0.01 e |
| PES1CMU | 0.25 ± 0.02 d | 0.94 ± 0.00 d | 1.62 ± 0.00 ef | 0.69 ± 0.02 a | 5.44 ± 0.03 h | 0.83 ± 0.01 b | 0.30 ± 0.10 c | 0.21 ± 0.01 i | 0.45 ± 0.02 f |
| RD6 | 0.36 ± 0.00 b | 0.46 ± 0.00 fg | 1.80 ± 0.10 def | 0.23 ± 0.01 f | 99.81 ± 1.25 b | 0.45 ± 0.02 ef | 0.92 ± 0.05 b | 3.28 ± 0.04 b | 0.56 ± 0.02 de |
| YM3CMU | 0.42 ± 0.01 a | 2.60 ± 0.13 b | 2.06 ± 0.19 bcde | 0.29 ± 0.01 d | 118.05 ± 2.09 a | 0.72 ± 0.02 c | 0.94 ± 0.03 b | 3.59 ± 0.04 a | 0.73 ± 0.04 b |
| Variety | RA | Catechin | p-coumaric acid | Epicatechin | Naringin | Gallic acid | Naringenin | Rutin | |
| BB3CMU | 0.12 ± 0.00 de | 0.61 ± 0.01 d | 0.17 ± 0.00 c | ND | 0.15 ± 0.01 b | ND | ND | 0.33 ± 0.00 a | |
| BB4CMU | 0.09 ± 0.00 e | 2.24 ± 0.14 a | 0.25 ± 0.01 a | ND | 0.17 ± 0.01 b | 0.29 ± 0.02 c | ND | ND | |
| BKU5CMU | 0.12 ± 0.07 de | ND | ND | 0.11 ± 0.05 a | ND | 0.17 ± 0.00 e | 0.20 ± 0.02 g | ND | |
| K4CMU | 0.33 ± 0.01 a | 0.26 ± 0.03 f | 0.10 ± 0.00 f | 0.11 ± 0.00 a | 0.52 ± 0.01 b | ND | 1.64 ± 0.02 a | 0.26 ± 0.02 c | |
| KAK1CMU | 0.24 ± 0.00 b | ND | 0.11 ± 0.01 e | ND | 7.89 ± 3.47 a | 0.34 ± 0.03 a | ND | ND | |
| KDK | 0.12 ± 0.00 de | 0.07 ± 0.02 g | 0.10 ± 0.00 f | 0.10 ± 0.00 a | 0.04 ± 0.00 b | ND | 0.38 ± 0.02 d | 0.20 ± 0.00 d | |
| KDML105 | 0.16 ± 0.02 cd | 0.36 ± 0.00 e | 0.22 ± 0.00 b | ND | 2.78 ± 1.71 b | ND | 0.25 ± 0.00 f | ND | |
| KCCMU107 | 0.11 ± 0.00 e | 0.39 ± 0.01 e | 0.12 ± 0.00 e | ND | 0.06 ± 0.00 b | ND | 0.61 ± 0.02 c | 0.30 ± 0.00 b | |
| Sang5CMU | ND | ND | 0.16 ± 0.00 c | ND | 6.60 ± 2.15 a | ND | 0.33 ± 0.00 e | ND | |
| PES1CMU | ND | 1.73 ± 0.00 b | 0.14 ± 0.01 d | ND | ND | ND | ND | 0.34 ± 0.01 a | |
| RD6 | 0.24 ± 0.00 b | ND | ND | ND | 0.85 ± 0.03 b | 0.25 ± 0.01 d | ND | ND | |
| YM3CMU | 0.19 ± 0.00 c | 1.37 ± 0.03 c | ND | ND | 2.03 ± 0.01 b | 0.32 ± 0.01 b | 0.69 ± 0.05 b | 0.21 ± 0.00 d |
Table 5. Phenolic contents in different varieties of rice husk.
| Variety | Caffeic acid | EG | o-coumaric acid | Quercetin | Phytic acid | Ferulic acid | CA | Kaempferol | HA |
|---|---|---|---|---|---|---|---|---|---|
| BB3CMU | 0.21 ± 0.02 de | 0.35 ± 0.00 h | 1.95 ± 0.08 cdef | 0.25 ± 0.00 ef | 15.45 ± 0.09 g | 0.34 ± 0.02 fg | 0.33 ± 0.14 c | 1.79 ± 0.01 e | 0.40 ± 0.02 f |
| BB4CMU | 0.25 ± 0.01 d | 0.45 ± 0.02 fg | 2.81 ± 0.01 a | 0.24 ± 0.01 f | 21.79 ± 0.07 e | 1.36 ± 0.15 a | 0.35 ± 0.12 c | 0.75 ± 0.01 g | 0.62 ± 0.04 c |
| BKU5CMU | 0.16 ± 0.02 f | 0.40 ± 0.01 gh | 2.14 ± 0.67 bcd | 0.25 ± 0.01 ef | 5.68 ± 0.24 h | 0.39 ± 0.04 fg | 0.23 ± 0.07 c | 1.25 ± 0.20 f | 0.60 ± 0.04 ed |
| K4CMU | 0.36 ± 0.01 b | 0.82 ± 0.03 e | 1.54 ± 0.08 fg | 0.61 ± 0.01 b | 31.40 ± 0.48 d | 0.32 ± 0.01 g | 2.05 ± 0.25 a | 3.63 ± 0.16 a | 1.07 ± 0.03 a |
| KAK1CMU | 0.18 ± 0.06 ef | 1.45 ± 0.00 c | 2.44 ± 0.07 abc | 0.46 ± 0.01 c | 14.75 ± 0.44 g | 0.43 ± 0.00 efg | 0.81 ± 0.03 b | 0.56 ± 0.06 h | 0.55 ± 0.02 de |
| KDK | 0.16 ± 0.01 ef | 0.15 ± 0.00 i | 0.37 ± 0.01 i | 0.27 ± 0.00 de | 6.99 ± 0.03 h | 0.18 ± 0.00 h | 0.15 ± 0.00 c | 0.32 ± 0.00 i | 0.23 ± 0.01 g |
| KDML105 | 0.40 ± 0.01 ab | 0.50 ± 0.02 f | 1.00 ± 0.03 h | 0.18 ± 0.00 g | 6.57 ± 0.13 h | 0.60 ± 0.04 d | 0.80 ± 0.07 b | 2.92 ± 0.03 c | 0.40 ± 0.01 f |
| KCCMU107 | 0.30 ± 0.00 c | 0.17 ± 0.02 i | 1.14 ± 0.01 gh | 0.24 ± 0.01 f | 67.36 ± 0.86 c | 0.52 ± 0.02 de | 0.25 ± 0.11 c | 2.47 ± 0.02 d | 0.54 ± 0.03 de |
| Sang5CMU | 0.20 ± 0.03 def | 2.80 ± 0.04 a | 2.49 ± 0.04 ab | 0.59 ± 0.03 b | 19.42 ± 0.34 f | 0.76 ± 0.04 bc | 1.90 ± 0.10 a | 0.13 ± 0.00 j | 0.53 ± 0.01 e |
| PES1CMU | 0.25 ± 0.02 d | 0.94 ± 0.00 d | 1.62 ± 0.00 ef | 0.69 ± 0.02 a | 5.44 ± 0.03 h | 0.83 ± 0.01 b | 0.30 ± 0.10 c | 0.21 ± 0.01 i | 0.45 ± 0.02 f |
| RD6 | 0.36 ± 0.00 b | 0.46 ± 0.00 fg | 1.80 ± 0.10 def | 0.23 ± 0.01 f | 99.81 ± 1.25 b | 0.45 ± 0.02 ef | 0.92 ± 0.05 b | 3.28 ± 0.04 b | 0.56 ± 0.02 de |
| YM3CMU | 0.42 ± 0.01 a | 2.60 ± 0.13 b | 2.06 ± 0.19 bcde | 0.29 ± 0.01 d | 118.05 ± 2.09 a | 0.72 ± 0.02 c | 0.94 ± 0.03 b | 3.59 ± 0.04 a | 0.73 ± 0.04 b |
| Variety | RA | Catechin | p-coumaric acid | Epicatechin | Naringin | Gallic acid | Naringenin | Rutin | |
| BB3CMU | 0.12 ± 0.00 de | 0.61 ± 0.01 d | 0.17 ± 0.00 c | ND | 0.15 ± 0.01 b | ND | ND | 0.33 ± 0.00 a | |
| BB4CMU | 0.09 ± 0.00 e | 2.24 ± 0.14 a | 0.25 ± 0.01 a | ND | 0.17 ± 0.01 b | 0.29 ± 0.02 c | ND | ND | |
| BKU5CMU | 0.12 ± 0.07 de | ND | ND | 0.11 ± 0.05 a | ND | 0.17 ± 0.00 e | 0.20 ± 0.02 g | ND | |
| K4CMU | 0.33 ± 0.01 a | 0.26 ± 0.03 f | 0.10 ± 0.00 f | 0.11 ± 0.00 a | 0.52 ± 0.01 b | ND | 1.64 ± 0.02 a | 0.26 ± 0.02 c | |
| KAK1CMU | 0.24 ± 0.00 b | ND | 0.11 ± 0.01 e | ND | 7.89 ± 3.47 a | 0.34 ± 0.03 a | ND | ND | |
| KDK | 0.12 ± 0.00 de | 0.07 ± 0.02 g | 0.10 ± 0.00 f | 0.10 ± 0.00 a | 0.04 ± 0.00 b | ND | 0.38 ± 0.02 d | 0.20 ± 0.00 d | |
| KDML105 | 0.16 ± 0.02 cd | 0.36 ± 0.00 e | 0.22 ± 0.00 b | ND | 2.78 ± 1.71 b | ND | 0.25 ± 0.00 f | ND | |
| KCCMU107 | 0.11 ± 0.00 e | 0.39 ± 0.01 e | 0.12 ± 0.00 e | ND | 0.06 ± 0.00 b | ND | 0.61 ± 0.02 c | 0.30 ± 0.00 b | |
| Sang5CMU | ND | ND | 0.16 ± 0.00 c | ND | 6.60 ± 2.15 a | ND | 0.33 ± 0.00 e | ND | |
| PES1CMU | ND | 1.73 ± 0.00 b | 0.14 ± 0.01 d | ND | ND | ND | ND | 0.34 ± 0.01 a | |
| RD6 | 0.24 ± 0.00 b | ND | ND | ND | 0.85 ± 0.03 b | 0.25 ± 0.01 d | ND | ND | |
| YM3CMU | 0.19 ± 0.00 c | 1.37 ± 0.03 c | ND | ND | 2.03 ± 0.01 b | 0.32 ± 0.01 b | 0.69 ± 0.05 b | 0.21 ± 0.00 d |
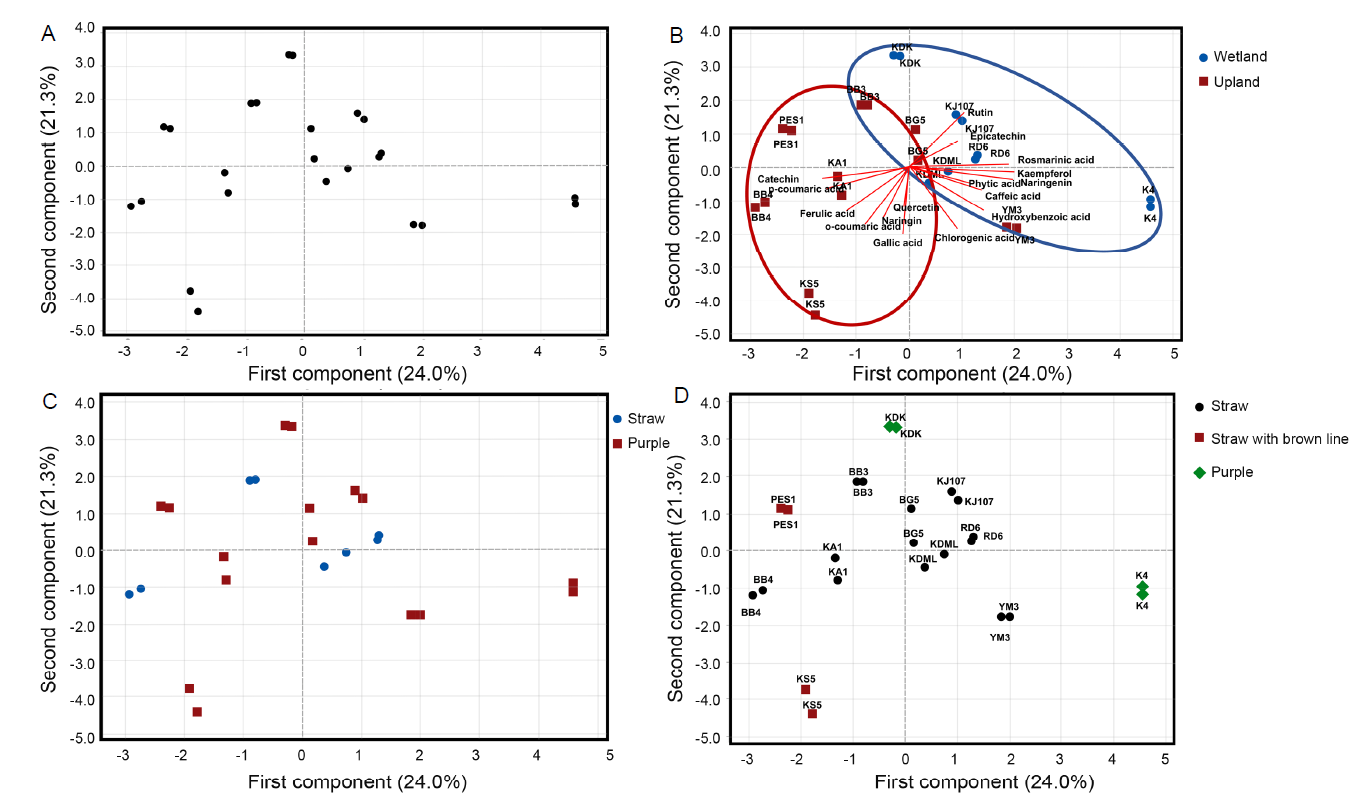
Fig. 3. Principle component analysis (PCA) of polyphenolic profile in rice husk samples. A, Score plot of polyphenolics profile. B, Biplot acclimations labeled as ecotype. C, Score plot acclimations labeled as pericarp color. D, Score plot acclimations labeled as husk color.
| [1] | Adom K K, Liu R H. 2002. Antioxidant activity of grains. J Agric Food Chem, 50(21): 6182-6187. |
| [2] | Arjin C, Pringproa K, Hongsibsong S, Ruksiriwanich W, Seel-Audom M, Mekchay S, Sringarm K. 2020. In vitro screening antiviral activity of Thai medicinal plants against porcine reproductive and respiratory syndrome virus. BMC Vet Res, 16: 102. |
| [3] |
Arranz S, Silván J M, Saura-Calixto F. 2010. Nonextractable polyphenols, usually ignored, are the major part of dietary polyphenols: A study on the Spanish diet. Mol Nutr Food Res, 54(11): 1646-1658.
PMID |
| [4] | Asadi H, Ghorbani M, Rezaei-Rashti M, Abrishamkesh S, Amirahmadi E, Chen C R, Gorji M. 2021. Application of rice husk biochar for achieving sustainable agriculture and environment. Rice Sci, 28(4): 325-343. |
| [5] | Bors W, Michel C, Stettmaier K. 2001. Structure-activity relationships governing antioxidant capacities of plant polyphenols. Methods Enzymol, 335: 166-180. |
| [6] | Bunzel M. 2010. Chemistry and occurrence of hydroxycinnamate oligomers. Phytochem Rev, 9(1): 47-64. |
| [7] |
Butsat S, Siriamornpun S. 2010. Phenolic acids and antioxidant activities in husk of different Thai rice varieties. Food Sci Technol Int, 16(4): 329-336.
PMID |
| [8] | Canan C, Cruz F T L, Delaroza F, Casagrande R, Sarmento C P M, Shimokomaki M, Ida E I. 2011. Studies on the extraction and purification of phytic acid from rice bran. J Food Compos Anal, 24(7): 1057-1063. |
| [9] | Chandrasekhar S, Satyanarayana K G, Pramada P N, Raghavan P, Gupta T N. 2003. Review processing, properties and applications of reactive silica from rice husk: An overview. J Mater Sci, 38(15): 3159-3168. |
| [10] | Chatha S S, Hussain A I, Zubair M, Khosa M K. 2011. Analytical characterization of rice (Oryza sativa) bran and bran oil from different agro-ecological regions. Pak J Agric Sci, 48: 243-249. |
| [11] | Dimitrios B. 2006. Sources of natural phenolic antioxidants. Trends Food Sci Technol, 17(9): 505-512. |
| [12] | do Nascimento L Á, Abhilasha A, Singh J, Elias M C, Colussi R. 2022. Rice germination and its impact on technological and nutritional properties: A review. Rice Sci, 29(3): 201-215. |
| [13] | Ed Nignpense B, Francis N, Blanchard C, Santhakumar A B. 2021. Bioaccessibility and bioactivity of cereal polyphenols: A review. Foods, 10(7): 1595. |
| [14] | Frank T, Meuleye B S, Miller A, Shu Q Y, Engel K H. 2007. Metabolite profiling of two low phytic acid (lpa) rice mutants. J Agric Food Chem, 55(26): 11011-11019. |
| [15] | Goffman F D, Bergman C J. 2004. Rice kernel phenolic content and its relationship with antiradical efficiency. J Sci Food Agric, 84(10): 1235-1240. |
| [16] |
Goufo P, Trindade H. 2014. Rice antioxidants: Phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, γ-oryzanol, and phytic acid. Food Sci Nutr, 2(2): 75-104.
PMID |
| [17] |
Goufo P, Trindade H. 2017. Factors influencing antioxidant compounds in rice. Crit Rev Food Sci Nutr, 57(5): 893-922.
PMID |
| [18] | Harshwardhan K, Upadhyay K. 2017. Effective utilization of agricultural waste: Review. J Fundam Renewable Energy Appl, 7(5): 237. |
| [19] | He J, Kawasaki S, Achal V. 2020. The utilization of agricultural waste as agro-cement in concrete: A review. Sustainability, 12(17): 6971. |
| [20] | Hussain A I, Chatha S A S, Noor S, Khan Z A, Arshad M U, Rathore H A, Sattar M Z A. 2012. Effect of extraction techniques and solvent systems on the extraction of antioxidant components from peanut (Arachis hypogaea L.) hulls. Food Anal Methods, 5(4): 890-896. |
| [21] | Irakli M, Kleisiaris F, Kadoglidou K, Katsantonis D. 2018. Optimizing extraction conditions of free and bound phenolic compounds from rice by-products and their antioxidant effects. Foods, 7(6): 93. |
| [22] | Jamjod S, Yimyam N, Lordkaew S, Prom-U-Thai C, Rerkasem B. 2017. Characterization of on-farm rice germplasm in an area of the crop’s center of diversity. Chiang Mai Univ J Nat Sci, 16(2): 85-98. |
| [23] | Khantham C, Yooin W, Sringarm K, Sommano S R, Jiranusornkul S, Carmona F D, Nimlamool W, Jantrawut P, Rachtanapun P, Ruksiriwanich W. 2021. Effects on steroid 5-alpha reductase gene expression of Thai rice bran extracts and molecular dynamics study on SRD5A2. Biology, 10(4): 319. |
| [24] | Khantham C, Linsaenkart P, Chaitep T, Jantrawut P, Chittasupho C, Rachtanapun P, Jantanasakulwong K, Phimolsiripol Y, Sommano S R, Prom-U-Thai C, Jamjod S, Arjin C, Sringarm K, Berrada H, Barba F J, Carmona F D, Nimlamool W, Ruksiriwanich W. 2022. Antioxidation, anti-inflammation, and regulation of SRD5A gene expression of Oryza sativa cv. bue Bang 3 CMU husk and bran extracts as androgenetic alopecia molecular treatment substances. Plants, 11(3): 330. |
| [25] | Korres N E, Norsworthy J K, Burgos N R, Oosterhuis D M. 2017. Temperature and drought impacts on rice production: An agronomic perspective regarding short- and long-term adaptation measures. Water Resour Rural Dev, 9: 12-27. |
| [26] | Kumar R, Vijayalakshmi S, Nadanasabapathi S. 2017. Health benefits of quercetin. Def Life Sci J, 2(10): 142-151. |
| [27] | Lee S C, Kim J H, Jeong S M, Kim D R, Ha J U, Nam K C, Ahn D U. 2003. Effect of far-infrared radiation on the antioxidant activity of rice hulls. J Agric Food Chem, 51(15): 4400-4403. |
| [28] | Lee S H, Park H J, Chun H K, Cho S Y, Cho S M, Lillehoj H S. 2006. Dietary phytic acid lowers the blood glucose level in diabetic KK mice. Nutr Res, 26(9): 474-479. |
| [29] | Leelayuthsoontorn P, Thipayarat A. 2006. Textural and morphological changes of Jasmine rice under various elevated cooking conditions. Food Chem, 96(4): 606-613. |
| [30] | Moure A, Cruz J M, Franco D, Domı́nguez J M, Sineiro J, Domı́nguez H, José Núñez M, Parajó J C. 2001. Natural antioxidants from residual sources. Food Chem, 72(2): 145-171. |
| [31] | Murakami M, Yamaguchi T, Takamura H, Atoba T M. 2004. Effects of thermal treatment on radical-scavenging activity of single and mixed polyphenolic compounds. J Food Sci, 69(1): FCT7-FCT10. |
| [32] | Murtey M D, Seeni A. 2020. The phytochemical analysis and pharmacological potentials of husk and straw as paddy waste products. J Sci Food Agric, 100(12): 4347-4352. |
| [33] | Nazir Y, Linsaenkart P, Khantham C, Chaitep T, Jantrawut P, Chittasupho C, Rachtanapun P, Jantanasakulwong K, Phimolsiripol Y, Sommano S R, Tocharus J, Mingmalairak S, Wongsa A, Arjin C, Sringarm K, Berrada H, Barba F J, Ruksiriwanich W. 2021. High efficiency in vitro wound healing of Dictyophora indusiata extracts via anti-inflammatory and collagen stimulating (MMP-2 inhibition) mechanisms. J Fungi, 7(12): 1100. |
| [34] | Nenadis N, Kyriakoudi A, Tsimidou M Z. 2013. Impact of alkaline or acid digestion to antioxidant activity, phenolic content and composition of rice hull extracts. LWT: Food Sci Technol, 54(1): 207-215. |
| [35] |
Park J, Gim S Y, Jeon J Y, Kim M J, Choi H K, Lee J. 2019. Chemical profiles and antioxidant properties of roasted rice hull extracts in bulk oil and oil-in-water emulsion. Food Chem, 272: 242-250.
PMID |
| [36] | Pérez-Jiménez J, Arranz S, Tabernero M, Díaz-Rubio M E, Serrano J, Goñi I, Saura-Calixto F. 2008. Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: Extraction, measurement and expression of results. Food Res Int, 41(3): 274-285. |
| [37] | Peschel W, Sánchez-Rabaneda F, Diekmann W, Plescher A, Gartzía I, Jiménez D, Lamuela-Raventós R, Buxaderas S, Codina C. 2006. An industrial approach in the search of natural antioxidants from vegetable and fruit wastes. Food Chem, 97(1): 137-150. |
| [38] | Pfaltzgraff L A, de bruyn M, Cooper E C, Budarin V, Clark J H. 2013. Food waste biomass: A resource for high-value chemicals. Green Chem, 15(2): 307-314. |
| [39] | Ramakrishna W, Kumari A, Rahman N, Mandave P. 2021. Anticancer activities of plant secondary metabolites: Rice callus suspension culture as a new paradigm. Rice Sci, 28(1): 13-30. |
| [40] | Ruksiriwanich W, Khantham C, Linsaenkart P, Chaitep T, Jantrawut P, Chittasupho C, Rachtanapun P, Jantanasakulwong K, Phimolsiripol Y, Sommano S R, Arjin C, Berrada H, Barba F J, Sringarm K. 2022. In vitro and in vivo regulation of SRD5A mRNA expression of supercritical carbon dioxide extract from Asparagus racemosus willd. root as anti-sebum and pore- minimizing active ingredients. Molecules, 27(5): 1535. |
| [41] | Salanti A, Zoia L, Orlandi M, Zanini F, Elegir G. 2010. Structural characterization and antioxidant activity evaluation of lignins from rice husk. J Agric Food Chem, 58(18): 10049-10055. |
| [42] | Sankam P, Punvittayagul C, Sringam K, Chaiyasut C, Wongpoomchai R. 2013. Antimutagenicity and anticlastogenicity of glutinous purple rice hull using in vitro and in vivo testing systems. Mol Cell Toxicol, 9(2): 169-176. |
| [43] | Sultana B, Anwar F, Rafique Asi M, Ali Shahid Chatha S. 2008. Antioxidant potential of extracts from different agro wastes: Stabilization of corn oil. Grasas Y Aceites, 59(3): 205-217. |
| [44] | Taratima W, Maneerattanarungroj P, Rattana K, Pathomsirivong W, Reanprayoon P. 2019. Nutritional composition and genetic diversity of Thai aromatic rice landraces. J Appl Bot Food Qual, 92: 281-287. |
| [45] |
Thammapat P, Meeso N, Siriamornpun S. 2016. Effects of the traditional method and an alternative parboiling process on the fatty acids, vitamin E, γ-oryzanol and phenolic acids of glutinous rice. Food Chem, 194: 230-236.
PMID |
| [46] | Verma D K, Srivastav P P. 2020. Bioactive compounds of rice (Oryza sativa L.): Review on paradigm and its potential benefit in human health. Trends Food Sci Technol, 97: 355-365. |
| [47] | Vichapong J, Sookserm M, Srijesdaruk V, Swatsitang P, Srijaranai S. 2010. High performance liquid chromatographic analysis of phenolic compounds and their antioxidant activities in rice varieties. LWT: Food Sci Technol, 43(9): 1325-1330. |
| [48] |
Wang W, Guo J, Zhang J N, Peng J, Liu T X, Xin Z H. 2015. Isolation, identification and antioxidant activity of bound phenolic compounds present in rice bran. Food Chem, 171: 40-49.
PMID |
| [49] |
Wanyo P, Meeso N, Siriamornpun S. 2014. Effects of different treatments on the antioxidant properties and phenolic compounds of rice bran and rice husk. Food Chem, 157: 457-463.
PMID |
| [50] | Wisetkomolmat J, Arjin C, Satsook A, Seel-Audom M, Ruksiriwanich W, Prom-U-Thai C, Sringarm K. 2022. Comparative analysis of nutritional components and phytochemical attributes of selected Thai rice bran. Front Nutr, 9: 833730. |
| [51] | Wu X L, Gu L W, Holden J, Haytowitz D B, Gebhardt S E, Beecher G, Prior R L. 2004. Development of a database for total antioxidant capacity in foods: A preliminary study. J Food Compos Anal, 17: 407-422. |
| [52] | Xu Q, Kanthasamy A G, Reddy M B. 2008. Neuroprotective effect of the natural iron Chelator, phytic acid in a cell culture model of Parkinson’s disease. Toxicology, 245: 101-108. |
| [53] | Yamuangmorn S, Prom-U-Thai C. 2021. The potential of high- anthocyanin purple rice as a functional ingredient in human health. Antioxidants, 10(6): 833. |
| [54] |
Young I S, Woodside J V. 2001. Antioxidants in health and disease. J Clin Pathol, 54(3): 176-186.
PMID |
| [55] | Zaky A A, Abd El-Aty A M, Ma A J, Jia Y M. 2022. An overview on antioxidant peptides from rice bran proteins: Extraction, identification, and applications. Crit Rev Food Sci Nutr, 62(5): 1350-1362. |
| [56] | Zhang M W, Zhang R F, Zhang F X, Liu R H. 2010. Phenolic profiles and antioxidant activity of black rice bran of different commercially available varieties. J Agric Food Chem, 58(13): 7580-7587. |
| [1] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [2] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [3] | Jiang Changjie, Liang Zhengwei, Xie Xianzhi. Priming for Saline-Alkaline Tolerance in Rice: Current Knowledge and Future Challenges [J]. Rice Science, 2023, 30(5): 417-425. |
| [4] | Salar Monajjem, Elias Soltani, Ebrahim Zainali, Masoud Esfahani, Farshid Ghaderi-Far, Maryam Hosseini Chaleshtori, Atefeh Rezaei. Seed Priming Improves Enzymatic and Biochemical Performances of Rice During Seed Germination under Low and High Temperatures [J]. Rice Science, 2023, 30(4): 335-347. |
| [5] | Yousef Alhaj Hamoud, Hiba Shaghaleh, Wang Ruke, Willy Franz Gouertoumbo, Amar Ali Adam hamad, Mohamed Salah Sheteiwy, Wang Zhenchang, Guo Xiangping. Wheat Straw Burial Improves Physiological Traits, Yield and Grain Quality of Rice by Regulating Antioxidant System and Nitrogen Assimilation Enzymes under Alternate Wetting and Drying Irrigation [J]. Rice Science, 2022, 29(5): 473-488. |
| [6] | Chunquan Zhu, Wenjun Hu, Xiaochuang Cao, Lianfeng Zhu, Yali Kong, Qianyu Jin, Guoxin Shen, Weipeng Wang, Hui Zhang, Junhua Zhang. Physiological and Proteomic Analyses Reveal Effects of Putrescine-Alleviated Aluminum Toxicity in Rice Roots [J]. Rice Science, 2021, 28(6): 579-593. |
| [7] | Qina Huang, Yinliang Wu, Guosheng Shao. Root Aeration Promotes Cadmium Accumulation in Rice by Regulating Iron Uptake-Associated System [J]. Rice Science, 2021, 28(5): 511-520. |
| [8] | Asadi Hossein, Ghorbani Mohammad, Rezaei-Rashti Mehran, Abrishamkesh Sepideh, Amirahmadi Elnaz, Chengrong Chen, Gorji Manouchehr. Application of Rice Husk Biochar for Achieving Sustainable Agriculture and Environment [J]. Rice Science, 2021, 28(4): 325-343. |
| [9] | Singh Priyanka, Pokharia Chitra, Shah Kavita. Exogenous Peroxidase Mitigates Cadmium Toxicity, Enhances Rhizobial Population and Lowers Root Knot Formation in Rice Seedlings [J]. Rice Science, 2021, 28(2): 166-177. |
| [10] | Dey Nivedita, Bhattacharjee Soumen. Accumulation of Polyphenolic Compounds and Osmolytes under Dehydration Stress and Their Implication in Redox Regulation in Four Indigenous Aromatic Rice Cultivars [J]. Rice Science, 2020, 27(4): 329-344. |
| [11] | B. Pedroso Giovanni, R. Philippsen Michael, F. Saldanha Loisleini, B. Araujo Raiara, F. Martins Ayrton. Strategies for Fermentable Sugar Production by Using Pressurized Acid Hydrolysis for Rice Husks [J]. Rice Science, 2019, 26(5): 319-330. |
| [12] | Yamuangmorn Supaporn, Dell Bernard, Prom-u-thai Chanakan. Effects of Cooking on Anthocyanin Concentration and Bioactive Antioxidant Capacity in Glutinous and Non-Glutinous Purple Rice [J]. Rice Science, 2018, 25(5): 270-278. |
| [13] | Kalita Jyotirmay, Kumar Pradhan Amit, Moni Shandilya Zina, Tanti Bhaben. Arsenic Stress Responses and Tolerance in Rice: Physiological, Cellular and Molecular Approaches [J]. Rice Science, 2018, 25(5): 235-249. |
| [14] | Nahar Shamsun, R. Vemireddy Lakshminarayana, Sahoo Lingaraj, Tanti Bhaben. Antioxidant Protection Mechanisms Reveal Significant Response in Drought-Induced Oxidative Stress in Some Traditional Rice of Assam, India [J]. Rice Science, 2018, 25(4): 185-196. |
| [15] | Kaur Maninder, Asthir Bavita, Mahajan Gulshan. Variation in Antioxidants, Bioactive Compounds and Antioxidant Capacity in Germinated and Ungerminated Grains of Ten Rice Cultivars [J]. Rice Science, 2017, 24(6): 349-359. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||