
Rice Science ›› 2024, Vol. 31 ›› Issue (2): 179-189.DOI: 10.1016/j.rsci.2023.11.005
• Reviews • Previous Articles Next Articles
Zhu Chengqi1, Ye Yuxuan1, Qiu Tian1, Huang Yafan2, Ying Jifeng3, Shen Zhicheng1( )
)
Received:2023-06-27
Accepted:2023-11-06
Online:2024-03-28
Published:2024-04-11
Contact:
SHEN Zhicheng (zcshen@zju.edu.cn)
Zhu Chengqi, Ye Yuxuan, Qiu Tian, Huang Yafan, Ying Jifeng, Shen Zhicheng. Drought-Tolerant Rice at Molecular Breeding Eras: An Emerging Reality[J]. Rice Science, 2024, 31(2): 179-189.
Add to citation manager EndNote|Ris|BibTeX
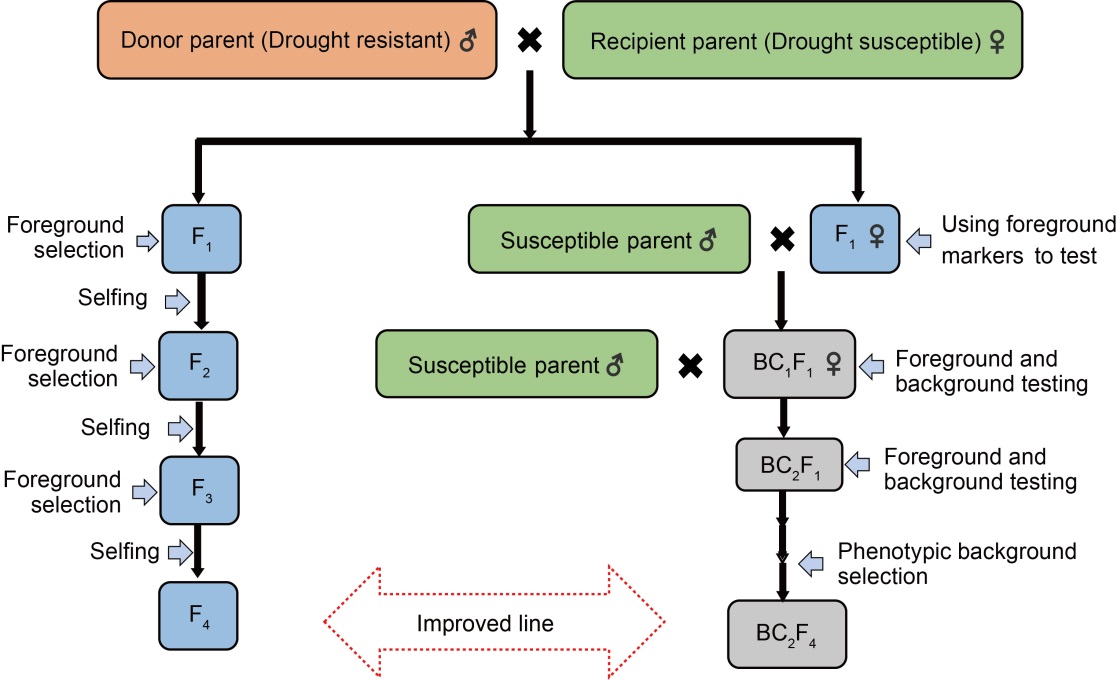
Fig. 3. General procedures for development of drought-tolerant rice varieties through marker-assisted backcrossing and pedigree selection. Fn, nth generation hybrid; BCnFn, nth generation hybrid with backcrossing × nth generation hybrids.
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| OsDREB1A | AP2/ERF | Greenhouse | Fast stomatal closure | Hsieh et al, |
| OsDREB1B, OsDREB1F | AP2/ERF | Field | Fast stomatal closure | Pellegrineschi et al, |
| AtHARDY | AP2/ERF | Greenhouse | High WUE, low transpiration, and high photosynthesis | Karaba et al, |
| AtABF3 | bZIP | Greenhouse | Less leaf rolling, wilting, and higher Fv/Fm | Oh et al, |
| AtZat10 | EAR (Zinc finger) | Greenhouse, field | High spikelet fertility | Xiao et al, |
| AtSNAC1 | NAC | Greenhouse, field | ABA-hypersensitive | Rabbani et al, |
| OsZFP252 | TFIIIA-type (Zinc finger) | Greenhouse | Proline and sugar accumulation, and better H2O2 homeostasis | Xu et al, |
| OsWRKY11 | WRKY (Zinc finger) | Greenhouse | Less leaf wilting and slow water loss | Wu et al, |
Table 1. Genes studied in transcriptional regulation of drought response and tolerance in rice.
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| OsDREB1A | AP2/ERF | Greenhouse | Fast stomatal closure | Hsieh et al, |
| OsDREB1B, OsDREB1F | AP2/ERF | Field | Fast stomatal closure | Pellegrineschi et al, |
| AtHARDY | AP2/ERF | Greenhouse | High WUE, low transpiration, and high photosynthesis | Karaba et al, |
| AtABF3 | bZIP | Greenhouse | Less leaf rolling, wilting, and higher Fv/Fm | Oh et al, |
| AtZat10 | EAR (Zinc finger) | Greenhouse, field | High spikelet fertility | Xiao et al, |
| AtSNAC1 | NAC | Greenhouse, field | ABA-hypersensitive | Rabbani et al, |
| OsZFP252 | TFIIIA-type (Zinc finger) | Greenhouse | Proline and sugar accumulation, and better H2O2 homeostasis | Xu et al, |
| OsWRKY11 | WRKY (Zinc finger) | Greenhouse | Less leaf wilting and slow water loss | Wu et al, |
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| AtLOS5 | Abscisic acid biosynthesis | Field | High spikelet fertility, and high yield | Xiao et al, |
| OsCDPK7 | Farnesylation | Greenhouse | Late embryogenesis abundant gene expression | Saijo et al, |
| OsHSP17.7 | Heat/cold-shock protein | Greenhouse | Protein protection | Sato and Yokoya, |
| CspA | Heat/cold-shock protein | Greenhouse, field | High photosynthesis, and increased yield | Castiglioni et al, |
| HVA1 | Late embryogenesis abundant protein | Greenhouse, field | High water use efficiency, high relative water content, and membrane protection | Xu et al, Sivamani et al, |
| OsLEA-3 | Late embryogenesis abundant protein | Greenhouse, field | High spikelet fertility, and high grain yield | Xiao et al, |
| Oatadc | Polyamine | Greenhouse | Spermine/spermidine accumulation, more chlorophyll, and less leaf rolling and wilting | Capell et al, |
| VaP5CS | Proline | Greenhouse | Proline accumulation, and less wilting | Zhu et al, De Ronde et al, |
| OsCIPK03, OsCIPK12, and OsCIPK15 | Protein phosphorylation | Greenhouse | Higher proline and sugar accumulation, and less leaf rolling | Xiang et al, |
| NPK1 | Protein phosphorylation | Greenhouse, field | High photosynthesis, high spikelet fertility, and high yield | Shou et al, |
| TPS | Sugar | Greenhouse | More trehalose, less leaf rolling | Garg et al, |
Table 2. Genes studied of drought tolerance in rice.
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| AtLOS5 | Abscisic acid biosynthesis | Field | High spikelet fertility, and high yield | Xiao et al, |
| OsCDPK7 | Farnesylation | Greenhouse | Late embryogenesis abundant gene expression | Saijo et al, |
| OsHSP17.7 | Heat/cold-shock protein | Greenhouse | Protein protection | Sato and Yokoya, |
| CspA | Heat/cold-shock protein | Greenhouse, field | High photosynthesis, and increased yield | Castiglioni et al, |
| HVA1 | Late embryogenesis abundant protein | Greenhouse, field | High water use efficiency, high relative water content, and membrane protection | Xu et al, Sivamani et al, |
| OsLEA-3 | Late embryogenesis abundant protein | Greenhouse, field | High spikelet fertility, and high grain yield | Xiao et al, |
| Oatadc | Polyamine | Greenhouse | Spermine/spermidine accumulation, more chlorophyll, and less leaf rolling and wilting | Capell et al, |
| VaP5CS | Proline | Greenhouse | Proline accumulation, and less wilting | Zhu et al, De Ronde et al, |
| OsCIPK03, OsCIPK12, and OsCIPK15 | Protein phosphorylation | Greenhouse | Higher proline and sugar accumulation, and less leaf rolling | Xiang et al, |
| NPK1 | Protein phosphorylation | Greenhouse, field | High photosynthesis, high spikelet fertility, and high yield | Shou et al, |
| TPS | Sugar | Greenhouse | More trehalose, less leaf rolling | Garg et al, |
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| OsCDPK7 | Farnesylation | Greenhouse | Late embryogenesis abundant gene expression | Saijo et al, |
| OsCIPK03, OsCIPK12, and OsCIPK15 | Protein phosphorylation | Greenhouse | Higher proline and sugar accumulation, and less leaf rolling | Xiang et al, |
| NPK1 | Protein phosphorylation | Greenhouse, field | High photosynthesis, high spikelet fertility, and high yield | Shou et al, |
Table 3. Genes studied of drought tolerance in rice: Post-translational regulation.
| Gene | Function | Evaluation | Physiological change | Reference |
|---|---|---|---|---|
| OsCDPK7 | Farnesylation | Greenhouse | Late embryogenesis abundant gene expression | Saijo et al, |
| OsCIPK03, OsCIPK12, and OsCIPK15 | Protein phosphorylation | Greenhouse | Higher proline and sugar accumulation, and less leaf rolling | Xiang et al, |
| NPK1 | Protein phosphorylation | Greenhouse, field | High photosynthesis, high spikelet fertility, and high yield | Shou et al, |
| Developer | Mechanism | Implementation location and status | Field trial result |
|---|---|---|---|
| Arcadia biosciences (America) | Expresses isopentenyltransferase from Agrobacterium, which catalyzes the rate-limiting step in cytokinin synthesis, and accompanies by SARK promoter from bean | Two years of field trials in rice with combined water use efficiency, nitrogen use efficiency, and salt tolerance; technology licensed to developers who have put the gene into their own varieties of soybean, wheat, rice, cotton, sugar beets, sugarcane, and tree crops | Inhibit or delay ethylene-induced senescence, while allowing ethylene to properly regulate leaf stomatal opening. The delay in senescence may enable active photosynthesis for longer periods during drought, allowing plants to synthesize osmo-protectants and other metabolites that improve drought tolerance |
| Chinese Academy of Sciences (China) | OsSPL14 regulated by OsmiR156 | Toward rice plant to ideal plant architecture, change plant tillering and leaf distribution | A point mutation in OsSPL14 perturbs OsmiR156-directed regulation of OsSPL14, generating an ‘ideal’ rice plant with a reduced tiller number, increased lodging resistance, and enhanced grain yield |
| DuPont Pioneer (America) | Expresses an ACS6 RNA construct to downregulate ACC synthase and decrease ethylene biosynthesis | Field trials in the USA and Chile | Through the expression of an RNAi construct that targets ACC synthase (ACS), which catalyzes the rate-limiting step in ethylene biosynthesis. Habben’s group downregulates ACS, which decreases the biosynthesis of ethylene, leading to increased grain yield. |
| Monsanto (America) | Expresses a cold-shock protein B from Bacillus subtilis, which stabilizes RNA | Deregulate in USA in December 2011, stewarded commercialization in US western Great Plains and Midwest | Average increase of five bushels of corn per acre during drought |
| Performance Plants (Canada) | Uses RNAi driven by conditional promoters to suppress farnesyltransferase, shuts down stomata | Licensed to Scotts (Marysville, Ohio, USA), Syngenta (Basel, Swiss), Bayer Crop Science (Monheim, Germany), DuPont Pioneer (USA), Mahyco (Jalna, India), Ricetec (Houston, USA), and DBN (Beijing, China) | Canola, 26% higher yield, petunia, double the number of flowers |
| University of Tokyo (Japan) | Expresses DrEB1a transcription factor | Field trials via collaborations with University of Calcutta (India) | Sugar production during drought in the transgenic varieties is 20%-30% higher than in conventional parental lines |
Table 4. Transgenic drought tolerant rice in commercial development and on the market.
| Developer | Mechanism | Implementation location and status | Field trial result |
|---|---|---|---|
| Arcadia biosciences (America) | Expresses isopentenyltransferase from Agrobacterium, which catalyzes the rate-limiting step in cytokinin synthesis, and accompanies by SARK promoter from bean | Two years of field trials in rice with combined water use efficiency, nitrogen use efficiency, and salt tolerance; technology licensed to developers who have put the gene into their own varieties of soybean, wheat, rice, cotton, sugar beets, sugarcane, and tree crops | Inhibit or delay ethylene-induced senescence, while allowing ethylene to properly regulate leaf stomatal opening. The delay in senescence may enable active photosynthesis for longer periods during drought, allowing plants to synthesize osmo-protectants and other metabolites that improve drought tolerance |
| Chinese Academy of Sciences (China) | OsSPL14 regulated by OsmiR156 | Toward rice plant to ideal plant architecture, change plant tillering and leaf distribution | A point mutation in OsSPL14 perturbs OsmiR156-directed regulation of OsSPL14, generating an ‘ideal’ rice plant with a reduced tiller number, increased lodging resistance, and enhanced grain yield |
| DuPont Pioneer (America) | Expresses an ACS6 RNA construct to downregulate ACC synthase and decrease ethylene biosynthesis | Field trials in the USA and Chile | Through the expression of an RNAi construct that targets ACC synthase (ACS), which catalyzes the rate-limiting step in ethylene biosynthesis. Habben’s group downregulates ACS, which decreases the biosynthesis of ethylene, leading to increased grain yield. |
| Monsanto (America) | Expresses a cold-shock protein B from Bacillus subtilis, which stabilizes RNA | Deregulate in USA in December 2011, stewarded commercialization in US western Great Plains and Midwest | Average increase of five bushels of corn per acre during drought |
| Performance Plants (Canada) | Uses RNAi driven by conditional promoters to suppress farnesyltransferase, shuts down stomata | Licensed to Scotts (Marysville, Ohio, USA), Syngenta (Basel, Swiss), Bayer Crop Science (Monheim, Germany), DuPont Pioneer (USA), Mahyco (Jalna, India), Ricetec (Houston, USA), and DBN (Beijing, China) | Canola, 26% higher yield, petunia, double the number of flowers |
| University of Tokyo (Japan) | Expresses DrEB1a transcription factor | Field trials via collaborations with University of Calcutta (India) | Sugar production during drought in the transgenic varieties is 20%-30% higher than in conventional parental lines |
| [1] | Abebe T, Guenzi A C, Martin B, Cushman J C. 2003. Tolerance of mannitol-accumulating transgenic wheat to water stress and salinity. Plant Physiol, 131(4): 1748-1755. |
| [2] | Amor Y, Babiychuk E, Inzé D, Levine A. 1998. The involvement of poly(ADP-ribose) polymerase in the oxidative stress responses in plants. FEBS Lett, 440(1/2): 1-7. |
| [3] | Bailey S J, Parker J E, Ainsworth E A, Oldroyd G E D, Schroeder J I. 2019. Genetic strategies for improving crop yields. Nature, 575: 109-118. |
| [4] | Baker S S, Wilhelm K S, Thomashow M F. 1994. The 5′-region of Arabidopsis thaliana cor15a has cis-acting elements that confer cold-, drought- and ABA-regulated gene expression. Plant Mol Biol, 24(5): 701-713. |
| [5] | Bolaños J, Edmeades G O. 1993. Eight cycles of selection for drought tolerance in lowland tropical maize. II: Responses in reproductive behavior. Field Crops Res, 31(3/4): 253-268. |
| [6] | Boudsocq M, Laurière C. 2005. Osmotic signaling in plants: Multiple pathways mediated by emerging kinase families. Plant Physiol, 138(3): 1185-1194. |
| [7] | Capell T, Escobar C, Liu H, Burtin D, Lepri O, Christou P. 1998. Over-expression of the oat arginine decarboxylase cDNA in transgenic rice (Oryza sativa L.)affects normal development patterns in vitro and results in putrescine accumulation in transgenic plants. Theor Appl Genet, 97(1): 246-254. |
| [8] | Capell T, Bassie L, Christou P. 2004. Modulation of the polyamine biosynthetic pathway in transgenic rice confers tolerance to drought stress. Proc Natl Acad Sci USA, 101(26): 9909-9914. |
| [9] | Castiglioni P, Warner D, Bensen R J, Anstrom D C, Harrison J, Stoecker M, Abad M, Kumar G, Salvador S, D’Ordine R, Navarro S, Back S, Fernandes M, Targolli J, Dasgupta S, Bonin C, Luethy M H, Heard J E. 2008. Bacterial RNA chaperones confer abiotic stress tolerance in plants and improved grain yield in maize under water-limited conditions. Plant Physiol, 147(2): 446-455. |
| [10] | Chae L, Sudat S, Dudoit S, Zhu T, Luan S. 2009. Diverse transcriptional programs associated with environmental: Stress and hormones in the Arabidopsis receptor-like kinase gene family. Mol Plant, 2(1): 84-107. |
| [11] | Chukwu S C, Rafii M Y, Ramlee S I, Ismail S I, Oladosu Y, Okporie E, Onyishi G, Utobo E, Ekwu L, Swaray S, Jalloh M. 2019. Marker- assisted selection and gene pyramiding for resistance to bacterial leaf blight disease of rice (Oryza sativa L.). Biotechnol Biotechnol Equip, 33(1): 440-455. |
| [12] | De Ronde J A, Cress W A, Krüger G H J, Strasser R J, 2004. Photosynthetic response of transgenic soybean plants, containing an Arabidopsis P5CR gene, during heat and drought stress. J Plant Physiol, 161(11): 1211-1224. |
| [13] | Dixit S, Singh A, Kumar A. 2014. Rice breeding for high grain yield under drought: A strategic solution to a complex problem. Int J Agron, 2014: 863683. |
| [14] | Doucet-Chabeaud G, Godon C, Brutesco C, de Murcia G, Kazmaier M. 2001. Ionising radiation induces the expression of PARP-1 and PARP-2 genes in Arabidopsis. Mol Gen Genomics, 265(6): 954-963. |
| [15] | Efendi B, Sabaruddin Z, Lukman H. 2017. Mutation with gamma raysirradiation to assemble green super rice tolerant to drought stress and high yield rice (Oryza sativa L.). Int J Adv Sci Eng Technol, 5: 1-5. |
| [16] | Fahad S, Bajwa A A, Nazir U, Anjum S A, Farooq A, Zohaib A, Sadia S, Nasim W, Adkins S, Saud S, Ihsan M Z, Alharby H, Wu C, Wang D P, Huang J L. 2017. Crop production under drought and heat stress: Plant responses and management options. Front Plant Sci, 8: 1147. |
| [17] | Furihata T, Maruyama K, Fujita Y, Umezawa T, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K. 2006. Abscisic acid-dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proc Natl Acad Sci USA, 103(6): 1988-1993. |
| [18] | Galichet A, Gruissem W. 2003. Protein farnesylation in plants: Conserved mechanisms but different targets. Curr Opin Plant Biol, 6(6): 530-535. |
| [19] | Garg A K, Kim J K, Owens T G, Ranwala A P, Do Choi Y, Kochian L V, Wu R J. 2002. Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA, 99(25): 15898-15903. |
| [20] | Gong D M, Zhang C Q, Chen X Y, Gong Z Z, Zhu J K. 2002. Constitutive activation and transgenic evaluation of the function of an Arabidopsis PKS protein kinase. J Biol Chem, 277(44): 42088-42096. |
| [21] | Holmström K O, Mäntylä E, Welin B, Mandal A, Palva E T, Tunnela O E, Londesborough J. 1996. Drought tolerance in tobacco. Nature, 379: 683-684. |
| [22] | Hsiao T C. 1973. Plant responses to water stress. Annu Rev Plant Physiol, 24: 519-570. |
| [23] | Hsieh T H, Lee J T, Yang P T, Chiu L H, Charng Y Y, Wang Y C, Chan M T. 2002. Heterology expression of the Arabidopsis C-repeat/ dehydration response element binding factor 1 gene confers elevated tolerance to chilling and oxidative stresses in transgenic tomato. Plant Physiol, 129(3): 1086-1094. |
| [24] | Hu H H, Xiong L Z. 2014. Genetic engineering and breeding of drought-resistant crops. Annu Rev Plant Biol, 65: 715-741. |
| [25] | Hu H H, Dai M Q, Yao J L, Xiao B Z, Li X H, Zhang Q F, Xiong L Z. 2006. Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proc Natl Acad Sci USA, 103(35): 12987-12992. |
| [26] | Huang X Y, Chao D Y, Gao J P, Zhu M Z, Shi M, Lin H X. 2009. A previously unknown zinc finger protein, DST, regulates drought and salt tolerance in rice via stomatal aperture control. Genes Dev, 23(15): 1805-1817. |
| [27] | Jang I C, Oh S J, Seo J S, Choi W B, Song S I, Kim C H, Kim Y S, Seo H S, Do Choi Y, Nahm B H, Kim J K. 2003. Expression of a bifunctional fusion of the Escherichia coli genes for trehalose-6- phosphate synthase and trehalose-6-phosphate phosphatase in transgenic rice plants increases trehalose accumulation and abiotic stress tolerance without stunting growth. Plant Physiol, 131(2): 516-524. |
| [28] | Karaba A, Dixit S, Greco R, Aharoni A, Trijatmiko K R, Marsch- Martinez N, Krishnan A, Nataraja K N, Udayakumar M, Pereira A. 2007. Improvement of water use efficiency in rice by expression of HARDY, an Arabidopsis drought and salt tolerance gene. Proc Natl Acad Sci USA, 104(39): 15270-15275. |
| [29] | Karim S, Aronsson H, Ericson H, Pirhonen M, Leyman B, Welin B, Mäntylä E, Palva E T, Van Dijck P, Holmström K O. 2007. Improved drought tolerance without undesired side effects in transgenic plants producing trehalose. Plant Mol Biol, 64(4): 371-386. |
| [30] | Khan M I R, Palakolanu S R, Chopra P, Rajurkar A B, Gupta R, Iqbal N, Maheshwari C. 2020. Improving drought tolerance in rice: Ensuring food security through multi-dimensional approaches. Physiol Plant, 172(2): 645-668. |
| [31] | Kumar A, Basu S, Ramegowda V, Pereira A. 2017. Mechanisms of drought tolerance in rice. In: Sasaki T. Achieving Sustainable Cultivation of Rice. Cambridge, the UK: Burleigh Dodds Science Publishing: 131-164. |
| [32] | Lafitte H R, Li Z K, Vijayakumar C H M, Gao Y M, Shi Y, Xu J L, Fu B Y, Yu S B, Ali A J, Domingo J, Maghirang R, Torres R, MacKill D. 2006. Improvement of rice drought tolerance through backcross breeding: Evaluation of donors and selection in drought nurseries. Field Crops Res, 97(1): 77-86. |
| [33] | Miah G, Rafii M Y, Ismail M R, Puteh A B, Rahim H A, Asfaliza R, Latif M A. 2013. Blast resistance in rice: A review of conventional breeding to molecular approaches. Mol Biol Rep, 40(3): 2369-2388. |
| [34] | Miah G, Rafii M Y, Ismail M R, Sahebi M, Hashemi F S G, Yusu O, Usman M G. 2017. Blast disease intimidation towards rice cultivation: A review of pathogen and strategies to control. J Anim Plant Sci, 27(4): 1058-1066. |
| [35] | Mundy J, Yamaguchi-Shinozaki K, Chua N H. 1990. Nuclear proteins bind conserved elements in the abscisic acid-responsive promoter of a rice rab gene. Proc Natl Acad Sci USA, 87(4): 1406-1410. |
| [36] | Nambara E, McCourt P. 1999. Protein farnesylation in plants: A greasy tale. Curr Opin Plant Biol, 2(5): 388-392. |
| [37] | Oh S J, Song S I, Kim Y S, Jang H J, Kim S Y, Kim M, Kim Y K, Nahm B H, Kim J K. 2005. Arabidopsis CBF3/DREB1A and ABF3 in transgenic rice increased tolerance to abiotic stress without stunting growth. Plant Physiol, 138(1): 341-351. |
| [38] | Oladosu Y, Rafii M Y, Abdullah N, Abdul Malek M, Rahim H A, Hussin G, Abdul Latif M, Kareem I. 2014. Genetic variability and selection criteria in rice mutant lines as revealed by quantitative traits. Sci World J, 2014: 190531. |
| [39] | Oladosu Y, Rafii M Y, Abdullah N, Malek M A, Rahim H A, Hussin G, Ismail M R, Latif M A, Kareem I. 2015. Genetic variability and diversity of mutant rice revealed by quantitative traits and molecular markers. Agrociencia, 49: 249-266. |
| [40] | Oladosu Y, Rafii M Y, Abdullah N, Hussin G, Ramli A, Rahim H A, Miah G, Usman M. 2016. Principle and application of plant mutagenesis in crop improvement: A review. Biotechnol Biotechnol Equip, 30(1): 1-16. |
| [41] | Oladosu Y, Rafii M Y, Samuel C, Fatai A, Magaji U, Kareem I, Kamarudin Z S, Muhammad I, Kolapo K. 2019. Drought resistance in rice from conventional to molecular breeding: A review. Int J Mol Sci, 20(14): 3519. |
| [42] | Oladosu Y, Rafii M Y, Arolu F, Chukwu S C, Salisu M A, Olaniyan B A, Fagbohun I K, Muftaudeen T K. 2021. Genetic diversity and utilization of cultivated eggplant germplasm in varietal improvement. Plants, 10(8): 1714. |
| [43] | Ozga J A, Kaur H, Savada R P, Reinecke D M. 2017. Hormonal regulation of reproductive growth under normal and heat-stress conditions in legume and other model crop species. J Exp Bot, 68(8): 1885-1894. |
| [44] | Panda D, Mishra S S, Behera P K. 2021. Drought tolerance in rice: Focus on recent mechanisms and approaches. Rice Sci, 28(2): 119-132. |
| [45] | Pandey V, Shukla A. 2015. Acclimation and tolerance strategies of rice under drought stress. Rice Sci, 22(4): 147-161. |
| [46] | Pang Y L, Chen K, Wang X Q, Xu J L, Ali J, Li Z K. 2017. Recurrent selection breeding by dominant male sterility for multiple abiotic stresses tolerant rice cultivars. Euphytica, 213(12): 268. |
| [47] | Pellegrineschi A, Reynolds M, Pacheco M, Brito R M, Almeraya R, Yamaguchi-Shinozaki K, Hoisington D. 2004. Stress-induced expression in wheat of the Arabidopsis thaliana DREB1A gene delays water stress symptoms under greenhouse conditions. Genome, 47(3): 493-500. |
| [48] | Polania J, Rao I M, Cajiao C, Grajales M, Rivera M, Velasquez F, Raatz B, Beebe S E. 2017. Shoot and root traits contribute to drought resistance in recombinant inbred lines of MD 23-24 × SEA 5 of common bean. Front Plant Sci, 8: 296. |
| [49] | Rabbani M A, Maruyama K, Abe H, Khan M A, Katsura K, Ito Y, Yoshiwara K, Seki M, Shinozaki K, Yamaguchi-Shinozaki K. 2003. Monitoring expression profiles of rice genes under cold, drought, and high-salinity stresses and abscisic acid application using cDNA microarray and RNA gel-blot analyses. Plant Physiol, 133(4): 1755-1767. |
| [50] | Rollins L A, Moles A T, Lam S, Buitenwerf R, Buswell J M, Brandenburger C R, Flores-Moreno H, Nielsen K B, Couchman E, Brown G S, Thomson F J, Hemmings F, Frankham R, Sherwin W B. 2013. High genetic diversity is not essential for successful introduction. Ecol Evol, 3(13): 4501-4517. |
| [51] | Sahebi M, Hanafi M M, Abdullah S N A, Rafii M Y, Azizi P, Nejat N, Idris A S. 2014. Isolation and expression analysis of novel silicon absorption gene from roots of mangrove (Rhizophora apiculata) via suppression subtractive hybridization. Biomed Res Int, 2014: 971985. |
| [52] | Sahebi M, Hanafi M M, Rafii M Y, Mahmud T M M, Azizi P, Osman M, Abiri R, Taheri S, Kalhori N, Shabanimofrad M, Miah G, Atabaki N. 2018. Improvement of drought tolerance in rice (Oryza sativa L.): Genetics, genomic tools, and the WRKY gene family. Biomed Res Int, 2018: 3158474. |
| [53] | Saijo Y, Hata S, Kyozuka J, Shimamoto K, Izui K. 2000. Over- expression of a single Ca2+-dependent protein kinase confers both cold and salt/drought tolerance on rice plants. Plant J, 23(3): 319-327. |
| [54] | Sakuma Y, Maruyama K, Osakabe Y, Qin F, Seki M, Shinozaki K, Yamaguchi-Shinozaki K. 2006. Functional analysis of an Arabidopsis transcription factor, DREB2A, involved in drought- responsive gene expression. Plant Cell, 18(5): 1292-1309. |
| [55] | Sato Y, Yokoya S. 2008. Enhanced tolerance to drought stress in transgenic rice plants overexpressing a small heat-shock protein, sHSP17.7. Plant Cell Rep, 27(2): 329-334. |
| [56] | Schroeder J I, Kwak J M, Allen G J. 2001. Guard cell abscisic acid signalling and engineering drought hardiness in plants. Nature, 410: 327-330. |
| [57] | Shou H X, Bordallo P, Wang K. 2004. Expression of the Nicotiana protein kinase (NPK1) enhanced drought tolerance in transgenic maize. J Exp Bot, 55: 1013-1019. |
| [58] | Singhal P, Jan A T, Azam M, Haq Q M R. 2016. Plant abiotic stress: A prospective strategy of exploiting promoters as alternative to overcome the escalating burden. Front Life Sci, 9(1): 52-63. |
| [59] | Sivamani E, Bahieldin A, Wraith J M, Al-Niemi T, Dyer W E, Ho T H D, Qu R D. 2000. Improved biomass productivity and water use efficiency under water deficit conditions in transgenic wheat constitutively expressing the barley HVA1 gene. Plant Sci, 155(1): 1-9. |
| [60] | Testerink C, Munnik T. 2005. Phosphatidic acid: A multifunctional stress signaling lipid in plants. Trends Plant Sci, 10(8): 368-375. |
| [61] | Usman M G, Rafii M Y, Martini M Y, Yusuff O A, Ismail M R, Miah G. 2018. Introgression of heat shock protein (Hsp70 and sHsp) genes into the Malaysian elite chilli variety Kulai (Capsicum annuum L.) through the application of marker-assisted backcrossing (MAB). Cell Stress Chaperon, 23(2): 223-234. |
| [62] | Wang Y, Ying J F, Kuzma M, Chalifoux M, Sample A, McArthur C, Uchacz T, Sarvas C, Wan J X, Dennis D T, McCourt P, Huang Y F. 2005. Molecular tailoring of farnesylation for plant drought tolerance and yield protection. Plant J, 43(3): 413-424. |
| [63] | Wang Y, Beaith M, Chalifoux M, Ying J F, Uchacz T, Sarvas C, Griffiths R, Kuzma M, Wan J X, Huang Y F. 2009. Shoot-specific down-regulation of protein farnesyltransferase (α-subunit) for yield protection against drought in canola. Mol Plant, 2(1): 191-200. |
| [64] | Wu X L, Shiroto Y, Kishitani S, Ito Y, Toriyama K. 2009. Enhanced heat and drought tolerance in transgenic rice seedlings overexpressing OsWRKY11 under the control of HSP101 promoter. Plant Cell Rep, 28(1): 21-30. |
| [65] | Xia H, Zhang X X, Liu Y, Bi J G, Ma X S, Zhang A N, Liu H Y, Chen L, Zhou S, Gao H, Xu K, Wei H B, Liu G L, Wang F M, Zhao H Y, Luo X X, Hou D P, Lou Q J, Feng F J, Zhou L G, Chen S J, Yan M, Li T F, Li M S, Wang L, Liu Z C, Yu X Q, Mei H W, Luo L J. 2022. Blue revolution for food security under carbon neutrality: A case from the water-saving and drought- resistance rice. Mol Plant, 15: 1401-1404. |
| [66] | Xiang Y, Huang Y M, Xiong L Z. 2007. Characterization of stress- responsive CIPK genes in rice for stress tolerance improvement. Plant Physiol, 144(3): 1416-1428. |
| [67] | Xiao B Z, Huang Y M, Tang N, Xiong L Z. 2007. Over-expression of a LEA gene in rice improves drought resistance under the field conditions. Theor Appl Genet, 115(1): 35-46. |
| [68] | Xiao B Z, Chen X, Xiang C B, Tang N, Zhang Q F, Xiong L Z. 2009. Evaluation of seven function-known candidate genes for their effects on improving drought resistance of transgenic rice under field conditions. Mol Plant, 2(1): 73-83. |
| [69] | Xu D, Duan X, Wang B, Hong B, Ho T H D, Wu R. 1996. Expression of a late embryogenesis abundant protein gene, HVA1, from barley confers tolerance to water deficit and salt stress in transgenic rice. Plant Physiol, 110: 249-257. |
| [70] | Xu D Q, Huang J, Guo S Q, Yang X, Bao Y M, Tang H J, Zhang H S. 2008. Overexpression of a TFIIIA-type zinc finger protein gene ZFP252 enhances drought and salt tolerance in rice (Oryza sativa L.). FEBS Lett, 582(7): 1037-1043. |
| [71] | Xue H W, Chen X, Mei Y. 2009. Function and regulation of phospholipid signalling in plants. Biochem J, 421(2): 145-156. |
| [72] | Yamaguchi-Shinozaki K, Shinozaki K. 2005. Organization of cis-acting regulatory elements in osmotic- and cold-stress- responsive promoters. Trends Plant Sci, 10(2): 88-94. |
| [73] | Yamaguchi-Shinozaki K, Shinozaki K. 2006. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol, 57: 781-803. |
| [74] | Yang S J, Vanderbeld B, Wan J X, Huang Y F. 2010. Narrowing down the targets: Towards successful genetic engineering of drought-tolerant crops. Mol Plant, 3(3): 469-490. |
| [75] | Zhu B C, Su J, Chang M C, Verma D P S, Fan Y L, Wu R. 1998. Overexpression of a Δ1-pyrroline-5-carboxylate synthetase gene and analysis of tolerance to water- and salt-stress in transgenic rice. Plant Sci, 139(1): 41-48. |
| [1] | Wei Huanhe, Geng Xiaoyu, Zhang Xiang, Zhu Wang, Zhang Xubin, Chen Yinglong, Huo Zhongyang, Zhou Guisheng, Meng Tianyao, Dai Qigen. Grain Yield, Biomass Accumulation, and Leaf Photosynthetic Characteristics of Rice under Combined Salinity-Drought Stress [J]. Rice Science, 2024, 31(1): 118-128. |
| [2] | Sundus Zafar, Xu Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 523-536. |
| [3] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [4] | M. Iqbal R. Khan, Sarika Kumari, Faroza Nazir, Risheek Rahul Khanna, Ravi Gupta, Himanshu Chhillar. Defensive Role of Plant Hormones in Advancing Abiotic Stress-Resistant Rice Plants [J]. Rice Science, 2023, 30(1): 15-35. |
| [5] | Chen Eryong, Shen Bo. OsABT, a Rice WD40 Domain-Containing Protein, Is Involved in Abiotic Stress Tolerance [J]. Rice Science, 2022, 29(3): 247-256. |
| [6] | Muhammad Mahran Aslam, Muhammad Abdul Rehman Rashid, Mohammad Aquil Siddiqui, Muhammad Tahir Khan, Fozia Farhat, Shafquat Yasmeen, Imtiaz Ahmad Khan, Shameem Raja, Fatima Rasool, Mahboob Ali Sial, Zhao Yan. Recent Insights into Signaling Responses to Cope Drought Stress in Rice [J]. Rice Science, 2022, 29(2): 105-117. |
| [7] | Balija Vishalakshi, Bangale Umakanth, Ponnuvel Senguttuvel, Makarand Barbadikar Kalyani, Prasad Madamshetty Srinivas, Rao Durbha Sanjeeva, Yadla Hari, Madhav Maganti Sheshu. Improvement of Upland Rice Variety by Pyramiding Drought Tolerance QTL with Two Major Blast Resistance Genes for Sustainable Rice Production [J]. Rice Science, 2021, 28(5): 493-500. |
| [8] | Baoxiang Wang, Yan Liu, Yifeng Wang, Jingfang Li, Zhiguang Sun, Ming Chi, Yungao Xing, Bo Xu, Bo Yang, Jian Li, Jinbo Liu, Tingmu Chen, Zhaowei Fang, Baiguan Lu, Dayong Xu, Kazeem Bello Babatunde. OsbZIP72 Is Involved in Transcriptional Gene-Regulation Pathway of Abscisic Acid Signal Transduction by Activating Rice High-Affinity Potassium Transporter OsHKT1;1 [J]. Rice Science, 2021, 28(3): 257-267. |
| [9] | Vera Jesus Da Costa Maria, Ramegowda Yamunarani, Ramegowda Venkategowda, N. Karaba Nataraja, M. Sreeman Sheshshayee, Udayakumar Makarla. Combined Drought and Heat Stress in Rice: Responses, Phenotyping and Strategies to Improve Tolerance [J]. Rice Science, 2021, 28(3): 233-242. |
| [10] | Panda Debabrata, Sakambari Mishra Swati, Kumar Behera Prafulla. Drought Tolerance in Rice: Focus on Recent Mechanisms and Approaches [J]. Rice Science, 2021, 28(2): 119-132. |
| [11] | Weidong Qi, Hongping Chen, Zuozhen Yang, Biaolin Hu, Xiangdong Luo, Bing Ai, Yuan Luo, Yu Huang, Jiankun Xie, Fantao Zhang. Systematic Characterization of Long Non-Coding RNAs and Their Responses to Drought Stress in Dongxiang Wild Rice [J]. Rice Science, 2020, 27(1): 21-31. |
| [12] | Donde Ravindra, Kumar Jitendra, Gouda Gayatri, Kumar Gupta Manoj, Mukherjee Mitadru, Yasin Baksh Sk, Mahadani Pradosh, Kumar Sahoo Khirod, Behera Lambodar, Kumar Dash Sushanta. Assessment of Genetic Diversity of Drought Tolerant and Susceptible Rice Genotypes Using Microsatellite Markers [J]. Rice Science, 2019, 26(4): 239-247. |
| [13] | Nahar Shamsun, R. Vemireddy Lakshminarayana, Sahoo Lingaraj, Tanti Bhaben. Antioxidant Protection Mechanisms Reveal Significant Response in Drought-Induced Oxidative Stress in Some Traditional Rice of Assam, India [J]. Rice Science, 2018, 25(4): 185-196. |
| [14] | Fantao Zhang, Yuan Luo, Meng Zhang, Yi Zhou, Hongping Chen, Biaolin Hu, Jiankun Xie. Identification and Characterization of Drought Stress- Responsive Novel microRNAs in Dongxiang Wild Rice [J]. Rice Science, 2018, 25(4): 175-184. |
| [15] | Nurdiani Dini, Widyajayantie Dwi, Nugroho Satya. OsSCE1 Encoding SUMO E2-Conjugating Enzyme Involves in Drought Stress Response of Oryza sativa [J]. Rice Science, 2018, 25(2): 73-81. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||