
Rice Science ›› 2018, Vol. 25 ›› Issue (2): 61-72.DOI: 10.1016/j.rsci.2017.08.005
• Orginal Article • Next Articles
Radhesh Krishnan Subramanian1,2, Muthuramalingam Pandiyan1, Pandian Subramani1, Banupriya Ramachandradoss1, Chithra Gunasekar1, Ramesh Manikandan1( )
)
Received:2017-04-10
Accepted:2017-08-14
Online:2018-03-28
Published:2017-12-22
Radhesh Krishnan Subramanian, Muthuramalingam Pandiyan, Pandian Subramani, Banupriya Ramachandradoss, Chithra Gunasekar, Ramesh Manikandan. Sprouted Sorghum Extract Elicits Coleoptile Emergence, Enhances Shoot and Root Acclimatization, and Maintains Genetic Fidelity in indica Rice[J]. Rice Science, 2018, 25(2): 61-72.
Add to citation manager EndNote|Ris|BibTeX
| Germination medium | Basal medium | Additive | Hormone |
|---|---|---|---|
| MS | MS | - | - |
| HMS | ½ MS | - | - |
| MIBA-1 | MS | - | 0.5 mol/L IBA |
| MIBA-2 | MS | - | 1.5 mol/L IBA |
| MSSE-10 | MS | 10% SE | - |
| MSSE-20 | MS | 20% SE | - |
| MSSE-30 | MS | 30% SE | - |
| MSSE-40 | MS | 40% SE | - |
Table 1 Different types of media for germination and elongation.
| Germination medium | Basal medium | Additive | Hormone |
|---|---|---|---|
| MS | MS | - | - |
| HMS | ½ MS | - | - |
| MIBA-1 | MS | - | 0.5 mol/L IBA |
| MIBA-2 | MS | - | 1.5 mol/L IBA |
| MSSE-10 | MS | 10% SE | - |
| MSSE-20 | MS | 20% SE | - |
| MSSE-30 | MS | 30% SE | - |
| MSSE-40 | MS | 40% SE | - |
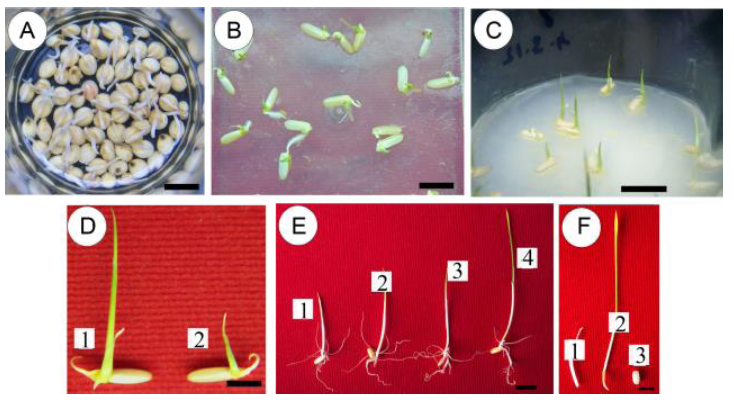
Fig. 1. Coleoptile emergence on different percentages of sorghum extract in medium.A, Germinated sorghum seeds in distilled water; B, Germinated ADT36 rice seeds on HMS medium; C, Germinated ADT36 rice seeds on MS medium; D, Germinated ADT36 rice seeds on MIBA-1 (1) and MIBA-2 (2) media; E, Coleoptile formation on MSSE-10 (1), MSSE-20 (2), MSSE-30 (3) and MSSE-40 (4) media; F, Dissected germinated coleoptile (1), leaf base (2) and seed (3) media. MS, Murashige and Skoog; HMS, Half of MS; MIBA, MS with indole-3-butyric acid; MSSE, MS with sorghum extract. The lengths of bars for A to F are 1 cm.
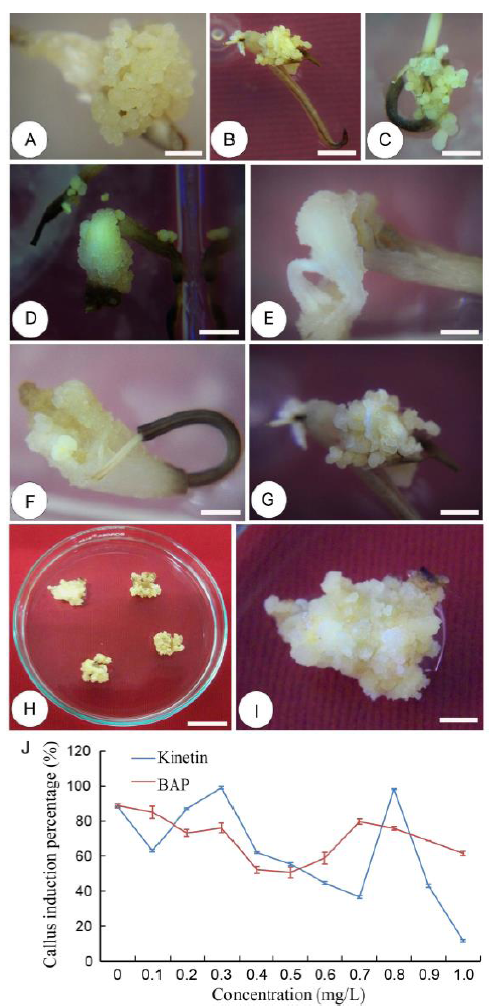
Fig. 3. Callus induction.A, Calli from mature seed; B, Calli from leaf base; C, Calli from coleoptile with 0.5 mg/L 2,4-D (2,4-dichlorophenoxyacetic acid); D, Calli from coleoptile with 1.0 mg/L 2,4-D; E, Calli from coleoptile with 1.5 mg/L 2,4-D; F, Calli from coleoptile with 2.0 mg/L 2,4-D; G, Calli from coleoptile with 2.5 mg/L 2,4-D; H, Proliferated calli; I, The closer view of proliferated calli; J, Effect of synthetic kinetin and 6-beniylamino purine (BAP) on callus proliferation. The lengths of bars for A to I are 1 cm. Bars for J represent the standard deviations.
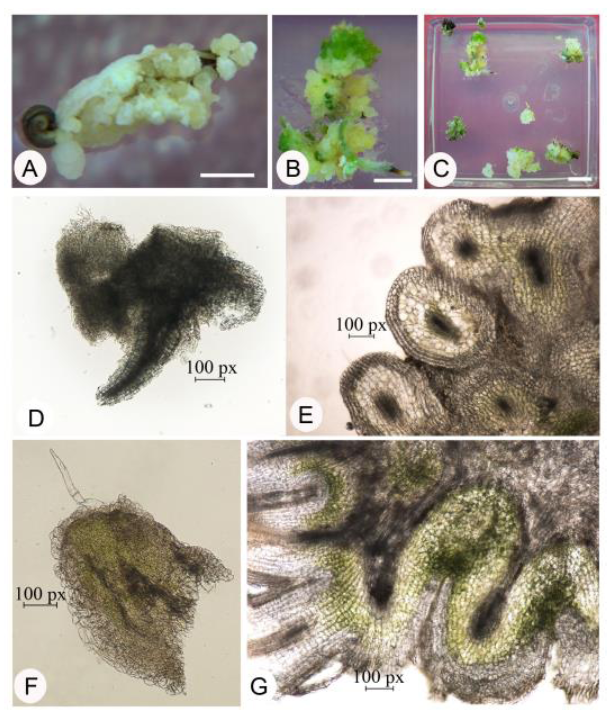
Fig. 4. Callus regeneration.A, Three-day-old regenerated callus (incubated at dark); B, Regenerated callus with SAMs (shoot apical meristems); C, Upper view of regenerated callus with shoot primordium; D, Bright field microscopic-cross sectional view of torpedo shaped callus; E, Globular callus; F, Regenerating globular callus; G, Regenerated callus with SAM. The lengths of scare bars for A to C are 1 cm.
| BAP | NAA (mg/L) | Average regeneration (%) | Shoot height | |
|---|---|---|---|---|
| (mg/L) | (cm) | |||
| Initiation of shoot primordium | ||||
| 0 | - | 5.67 ± 0.58 | - | |
| 0.1 | - | 3.33 ± 1.53 | - | |
| 0.2 | - | 11.33 ± 0.58 | - | |
| 0.3 | - | 12.00 ± 3.61 | - | |
| 0.4 | - | 15.33 ± 1.53 | - | |
| 0.5 | - | 13.00 ± 1.00* | - | |
| 0.6 | - | 14.00 ± 1.73 | - | |
| 0.7 | - | 14.33 ± 1.53 | - | |
| 0.8 | - | 16.00 ± 2.00 | - | |
| 0.9 | - | 20.00 ± 1.00* | - | |
| 1 | - | 21.67 ± 1.53 | - | |
| 1.5 | - | 25.00 ± 1.00* | - | |
| 2 | - | 26.67 ± 1.53 | - | |
| 2.5 | - | 35.67 ± 1.53 | - | |
| 3 | - | 33.67 ± 2.52 | - | |
| 3.5 | - | 38.67 ± 1.53 | - | |
| 4 | - | 35.67 ± 4.58 | - | |
| Development of SAMs | ||||
| 2.5 | 0.1 | 37.00 ± 1.00* | 8.20 ± 0.09* | |
| 2.5 | 0.2 | 36.67 ± 1.15* | 8.77 ± 0.66* | |
| 2.5 | 0.3 | 37.67 ± 1.53 | 10.07 ± 0.47* | |
| 2.5 | 0.4 | 38.67 ± 1.53 | 10.87 ± 0.36* | |
| 2.5 | 0.5 | 36.67 ± 2.08 | 10.30 ± 1.25 | |
| 2.5 | 0.6 | 43.67 ± 1.53 | 13.32 ± 0.36* | |
| 2.5 | 0.7 | 43.33 ± 1.53 | 13.43 ± 0.43* | |
| 2.5 | 0.8 | 47.00 ± 2.65 | 13.39 ± 1.02 | |
| 2.5 | 0.9 | 50.00 ± 1.00* | 14.50 ± 0.42* | |
| 2.5 | 1 | 49.67 ± 4.16 | 13.45 ± 1.84 | |
| 2.5 | 1.5 | 56.00 ± 2.65 | 15.58 ± 2.06 | |
| 2.5 | 2 | 56.00 ± 3.60 | 15.70 ± 0.48* | |
| 2.5 | 2.5 | 62.33 ± 1.53 | 15.72 ± 0.46* | |
| 2.5 | 3 | 62.00 ± 1.00 | 16.24 ± 0.64* | |
| 2.5 | 3.5 | 57.67 ± 1.53 | 14.92 ± 0.70* | |
| 2.5 | 4 | 53.00 ± 2.65 | 12.55 ± 0.67 | |
Table 2 Effect of plant growth hormones on callus regeneration initiation of shoot primordium.
| BAP | NAA (mg/L) | Average regeneration (%) | Shoot height | |
|---|---|---|---|---|
| (mg/L) | (cm) | |||
| Initiation of shoot primordium | ||||
| 0 | - | 5.67 ± 0.58 | - | |
| 0.1 | - | 3.33 ± 1.53 | - | |
| 0.2 | - | 11.33 ± 0.58 | - | |
| 0.3 | - | 12.00 ± 3.61 | - | |
| 0.4 | - | 15.33 ± 1.53 | - | |
| 0.5 | - | 13.00 ± 1.00* | - | |
| 0.6 | - | 14.00 ± 1.73 | - | |
| 0.7 | - | 14.33 ± 1.53 | - | |
| 0.8 | - | 16.00 ± 2.00 | - | |
| 0.9 | - | 20.00 ± 1.00* | - | |
| 1 | - | 21.67 ± 1.53 | - | |
| 1.5 | - | 25.00 ± 1.00* | - | |
| 2 | - | 26.67 ± 1.53 | - | |
| 2.5 | - | 35.67 ± 1.53 | - | |
| 3 | - | 33.67 ± 2.52 | - | |
| 3.5 | - | 38.67 ± 1.53 | - | |
| 4 | - | 35.67 ± 4.58 | - | |
| Development of SAMs | ||||
| 2.5 | 0.1 | 37.00 ± 1.00* | 8.20 ± 0.09* | |
| 2.5 | 0.2 | 36.67 ± 1.15* | 8.77 ± 0.66* | |
| 2.5 | 0.3 | 37.67 ± 1.53 | 10.07 ± 0.47* | |
| 2.5 | 0.4 | 38.67 ± 1.53 | 10.87 ± 0.36* | |
| 2.5 | 0.5 | 36.67 ± 2.08 | 10.30 ± 1.25 | |
| 2.5 | 0.6 | 43.67 ± 1.53 | 13.32 ± 0.36* | |
| 2.5 | 0.7 | 43.33 ± 1.53 | 13.43 ± 0.43* | |
| 2.5 | 0.8 | 47.00 ± 2.65 | 13.39 ± 1.02 | |
| 2.5 | 0.9 | 50.00 ± 1.00* | 14.50 ± 0.42* | |
| 2.5 | 1 | 49.67 ± 4.16 | 13.45 ± 1.84 | |
| 2.5 | 1.5 | 56.00 ± 2.65 | 15.58 ± 2.06 | |
| 2.5 | 2 | 56.00 ± 3.60 | 15.70 ± 0.48* | |
| 2.5 | 2.5 | 62.33 ± 1.53 | 15.72 ± 0.46* | |
| 2.5 | 3 | 62.00 ± 1.00 | 16.24 ± 0.64* | |
| 2.5 | 3.5 | 57.67 ± 1.53 | 14.92 ± 0.70* | |
| 2.5 | 4 | 53.00 ± 2.65 | 12.55 ± 0.67 | |
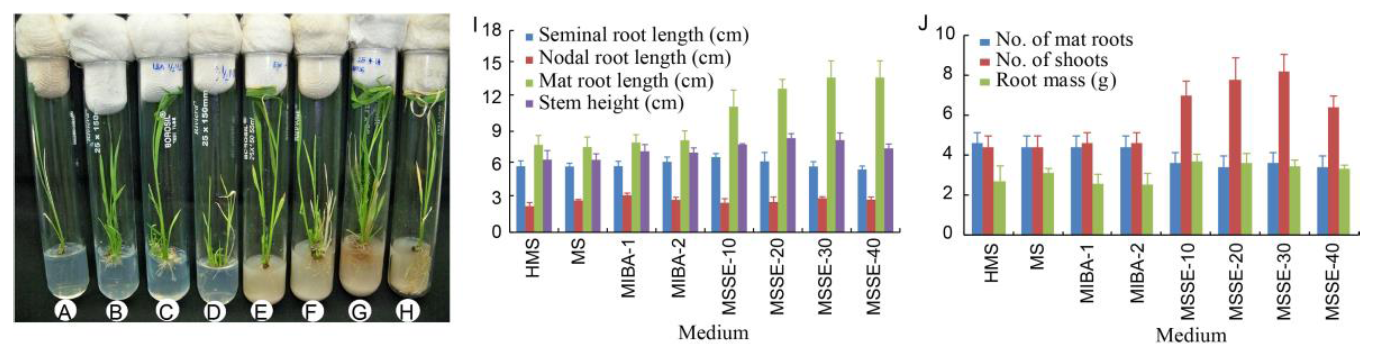
Fig. 5. Plantlet acclimatization and root and shoot establishment on medium with different plant growth hormones (PGHs) or sprouted sorghum extracts (SSEs).A, HMS; B, MS; C, MIBA-1; D, MIBA-2; E, MSSE-10; F, MSSE-20; G, MSSE-30; H, MSSE-40; I and J, Effect of PGHs or SSEs on seed sprouting and coleoptile induction.MS, Murashige and Skoog; HMS, Half of MS; MIBA, MS with indole-3-butyric acid; MSSE, MS with sorghum extract. Bar for I and J represents standard deviation.
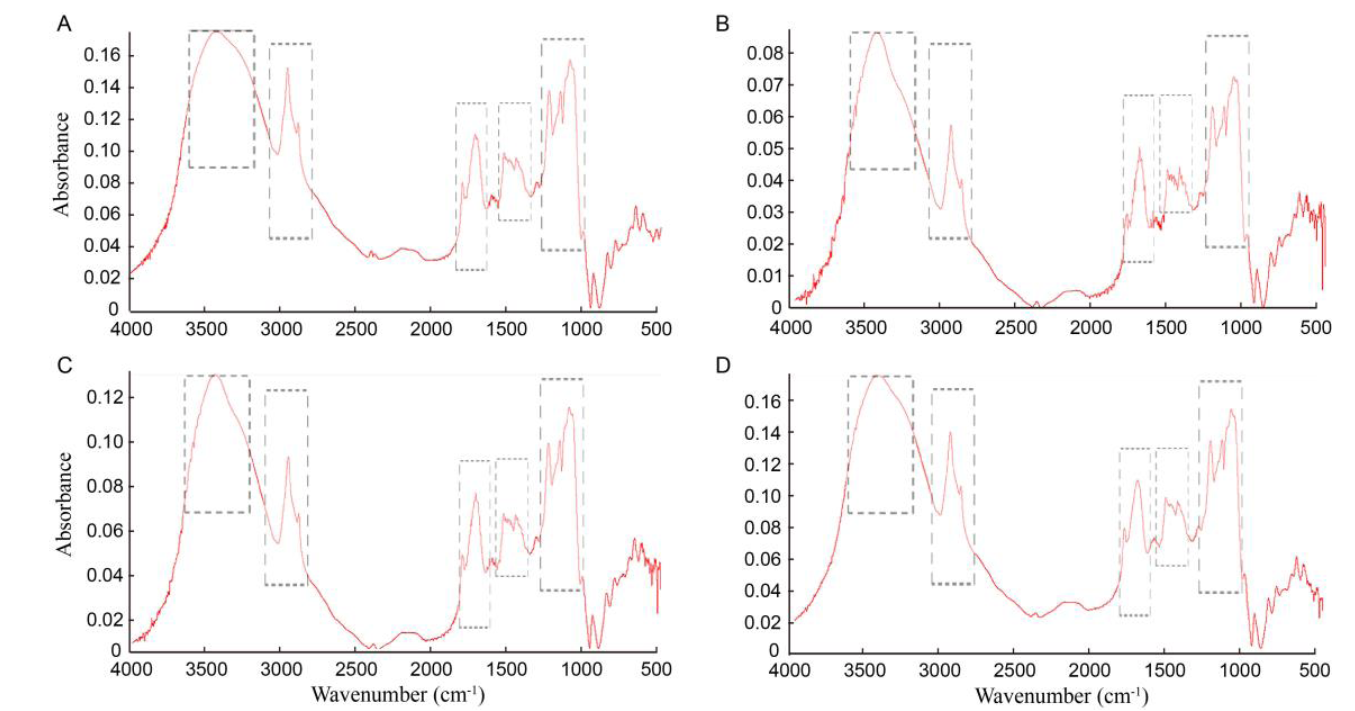
Fig. 6. Fourier-transform infrared spectroscopy spectrum from seeds of direct germinated (A), in vitro germinated (B), regenerated (C) and hardened plants (D).Dashed box represents the cellular component.
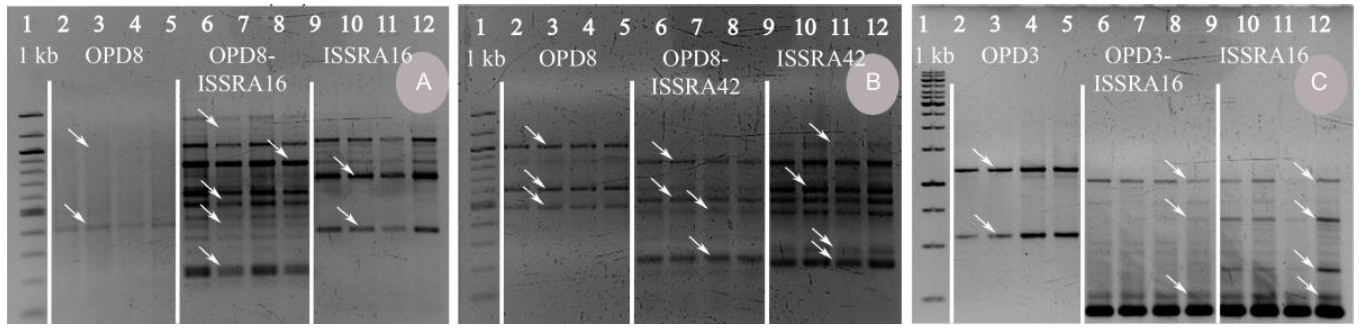
Fig. 7. Representative gel picture depicting genetic profile of random amplified polymorphic DNA (RAPD), inter-simple sequence repeat (ISSR), and R-ISSR (combination of RAPD and ISSR) primers of direct germinated (lanes 2, 6 and 10), in vitro germinated (lanes 3, 7 and 11), regenerated (lanes 4, 8 and 12), and hardened plants (lanes 5, 9 and 13).The arrow represents monomorphic band.
| No. | Primer | Annealing temperature | No. | Primer | Annealing temperature | No. | Primer | Annealing temperature | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 37 ºC | 55 ºC | 37 ºC | 55 ºC | 37 ºC | 55 ºC | ||||||
| 1 | OPD3 | + | + | 10 | ISSR46 | - | + | 18 | OPD3-ISSR35 | - | + |
| 2 | OPD8 | + | + | 11 | ISSR67 | + | + | 19 | OPD3-ISSR42 | + | - |
| 3 | OPD18 | + | - | 12 | ISSRA8 | - | + | 20 | OPD3-ISSR67 | - | - |
| 4 | OPF4 | + | - | 13 | ISSRA10 | - | + | 21 | OPD3-ISSRA16 | - | - |
| 5 | OPG5 | + | - | 14 | ISSRA16 | + | + | 22 | OPD8-ISSR35 | + | - |
| 6 | OPH3 | + | - | 15 | ISSRA33 | - | + | 23 | OPD8-ISSR42 | + | + |
| 7 | ISSR15 | - | + | 16 | ISSRY5 | - | + | 24 | OPD8-ISSR67 | - | + |
| 8 | ISSR35 | + | + | 17 | ISSRY11 | - | + | 25 | OPD8-ISSRA16 | + | - |
| 9 | ISSR42 | + | + | ||||||||
Table 3 Random amplified polymorphic DNA (RAPD) and inter-simple sequence repeat (ISSR) primers used to check genetic profile.
| No. | Primer | Annealing temperature | No. | Primer | Annealing temperature | No. | Primer | Annealing temperature | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 37 ºC | 55 ºC | 37 ºC | 55 ºC | 37 ºC | 55 ºC | ||||||
| 1 | OPD3 | + | + | 10 | ISSR46 | - | + | 18 | OPD3-ISSR35 | - | + |
| 2 | OPD8 | + | + | 11 | ISSR67 | + | + | 19 | OPD3-ISSR42 | + | - |
| 3 | OPD18 | + | - | 12 | ISSRA8 | - | + | 20 | OPD3-ISSR67 | - | - |
| 4 | OPF4 | + | - | 13 | ISSRA10 | - | + | 21 | OPD3-ISSRA16 | - | - |
| 5 | OPG5 | + | - | 14 | ISSRA16 | + | + | 22 | OPD8-ISSR35 | + | - |
| 6 | OPH3 | + | - | 15 | ISSRA33 | - | + | 23 | OPD8-ISSR42 | + | + |
| 7 | ISSR15 | - | + | 16 | ISSRY5 | - | + | 24 | OPD8-ISSR67 | - | + |
| 8 | ISSR35 | + | + | 17 | ISSRY11 | - | + | 25 | OPD8-ISSRA16 | + | - |
| 9 | ISSR42 | + | + | ||||||||
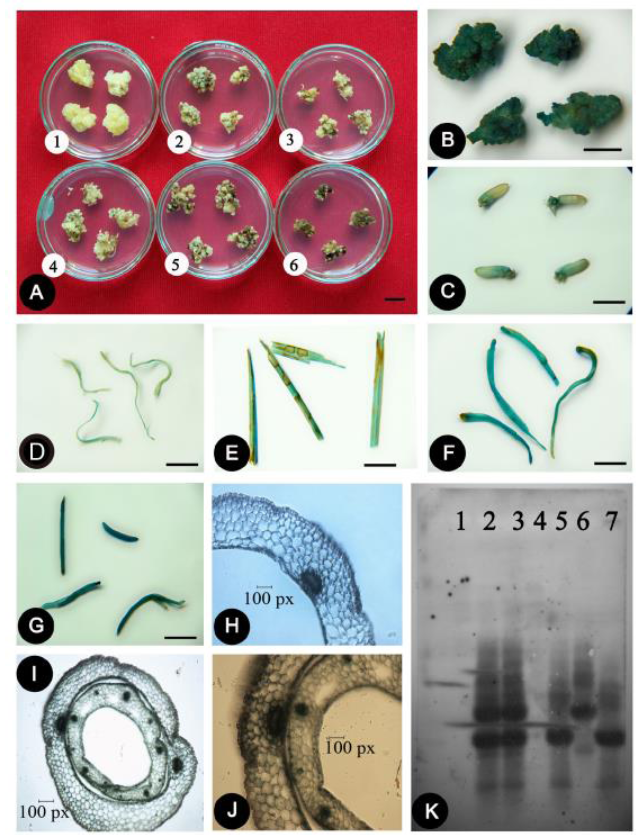
Fig. 9. T-DNA gene transfer and validation in different explants.A, Checking hygromycin resistance by coleoptile derived callus. 1 to 6 means control, 10, 20, 30, 40 and 50 μg/L hygromycin; B, GUS expression in callus; C, GUS expression in mature seed; D, GUS expression in root; E, GUS expression in leaf blade; F, GUS expression in leaf base; G, GUS expression in coleoptile; H, Cross-section of GUS expressed in leaf base; I-J, Cross-section of GUS expressed in coleoptile; K, DNA blot hybridization of hpt gene integration in different explants (1, Control-wild type leaf; 2, Callus; 3, Mature seed; 4, Root; 5, Leaf blade; 6, Leaf base; 7, Coleoptile).The lengths of bars for A to G are 1 cm.
| [1] | Abe T, Futsuhara Y.1985. Efficient plant regeneration by somatic embryogenesis from root callus tissues of rice (Oryza sativa L.). J Plant Physiol, 121(2): 111-118. |
| [2] | Abe T, Futsuhara Y.1986. Genotypic variability for callus formation and plant regeneration in rice (Oryza sativa L.). Theor Appl Genet, 72(1): 3-10. |
| [3] | Azria D, Bhalla P L.2000. Plant regeneration from mature embryo- derived callus of Australian rice (Oryza sativa L.) varieties. Aust J Agric Res, 51(2): 305-312. |
| [4] | Chaban C, Waller F, Furuya M, Nick P.2003. Auxin responsiveness of a novel cytochrome P450 in rice coleoptiles.Plant Physiol, 133(4): 2000-2009. |
| [5] | Chu Q R.1988. Genetics of callus formation and plant regeneration in rice (Oryza sativa L.). LSU Hist Diss Thes: 4491. |
| [6] | Dabul A N G, Belefant-Miller H, RoyChowdhury M, Hubstenberger J F, Lorence A, Phillips G C.2008. Screening of a broad range of rice (Oryza sativa L.) germplasm for in vitro rapid plant regeneration and development of an early prediction system. In Vitro Cell Dev-Pl, 45(4): 414-420. |
| [7] | Ellepola S W, Choi S M, Ma C Y.2005. Conformational study of globulin from rice (Oryza sativa) seeds by fourier-transform infrared spectroscopy. Int J Biol Macromol, 37: 12-20. |
| [8] | Gamborg O L, Miller R A, Ojima K.1968. Nutrient requirements of suspension cultures of soybean root cells.Exp Cell Res, 50(1): 151-158. |
| [9] | Geethalakshmi V, Lakshmanan A, Rajalakshmi D, Jagannathan R, Sridhar G, Ramaraj A P, Bhuvaneswari K, Gurusamy L, Anbhazhagan R.2011. Climate change impact assessment and adaptation strategies to sustain rice production in Cauvery basin of Tamil Nadu.Curr Sci, 101(3): 342-347. |
| [10] | Genkawa T, Ahamed T, Noguchi R, Takigawa T, Ozaki Y.2015. Simple and rapid determination of free fatty acids in brown rice by FTIR spectroscopy in conjunction with a second-derivative treatment.Food Chem, 191: 7-11. |
| [11] | George E F, Hall M A, Klerk G J D.2008. Plant Propagation by Tissue Culture. 3rd edn. Dordrecht, the Netherlands: Springer: 501. |
| [12] | Ghodrat V, Moradshahi A, Rousta M J, Karampour A.2013. Improving yield and yield components of rice (Oryza sativa L.) by indolebutyric acid (IBA), gibberellic acid (GA3) and salicylic acid (SA) pre- sowing seed treatments. Am J Agric Environ Sci, 13: 872-876. |
| [13] | Giancarla V, Emilian M, Radu S, Sorin C, Sorina P, Cerasela P.2012. The use of RAPD and ISSR markers for genetic diversity among some barley cultivars.Rom Biotechnol Lett, 17(4): 7493-7503. |
| [14] | Grant J N, Burris J N, Stewart C N, Lenaghan S C.2017. Improved tissue culture conditions for the emerging C4 model Panicum hallii. BMC Biotechnol, 17: 39. |
| [15] | Imani J, Kumar A, Neumann K H.2009. Plant Cell and Tissue Culture: A Tool in Biotechnology. Berlin, Heidelberg: Springer Berlin Heidelberg. |
| [16] | Jain M, Nijhawan A, Tyagi A K, Khurana J P.2006. Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR.Biochem Biophysiol Res Commun, 345(2): 646-651. |
| [17] | Jefferson R A, Kavanagh T A, Bevan M W.1987. GUS fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants.EMBO J, 6(13): 3901-3907. |
| [18] | Kant T, Kothari S L, Kononowicz-Hodges H, Hodges T K.2001. Agrobacterium tumefaciens-mediated transformation of rice using coleoptile and mature seed-derived callus. J Plant Biochem Biotechnol, 10(2): 121-126. |
| [19] | Karthikeyan A, Pandian S K, Ramesh M.2009. High frequency plant regeneration from embryogenic callus of a popular indica rice(Oryza sativa L.). Physiol Mol Biol Plants, 15: 371-375. |
| [20] | Karthikeyan A, Pandian S K, Ramesh M.2011a.Agrobacterium- mediated transformation of leaf base derived callus tissues of popular indica rice(Oryza sativa L. subsp. indica cv. ADT43). Plant Sci, 181(3): 258-268. |
| [21] | Karthikeyan A, Shilpha J, Pandian S K, Ramesh M.2011b.Agrobacterium-mediated transformation of indica rice cv. ADT43. Plant Cell, 109(1): 153-165. |
| [22] | Kefford N P.1961. Auxin-gibberellin interaction in rice coleoptile elongation.Plant Physiol, 380-386. |
| [23] | Koetje D S, Grimes H D, Wang Y C, Hodges T K.1989. Regeneration of indica rice(Oryza sativa L.) from primary callus derived from immature embryos. J Plant Physiol, 135(2): 184-190. |
| [24] | Krishnan S R, Priya A M, Ramesh M.2013. Rapid regeneration and ploidy stability of ‘cv IR36’ indica rice(Oryza sativa L.) confers efficient protocol for in vitro callus organogenesis and Agrobacterium tumefaciens mediated transformation. Bot Stud, 54: 47. |
| [25] | Li X M, Bai H, Wang X Y, Li L Y, Cao Y H, Wei J, Liu Y M, Liu L J, Gong X D, Wu L, Liu S Q, Liu G Z.2011. Identification and validation of rice reference proteins for western blotting.J Exp Bot, 62: 4763-4772. |
| [26] | Linsmaier E M, Skoog F.1965. Organic growth factor requirements of tobacco tissue cultures.Physiol Plant, 18(1): 100-127. |
| [27] | Magneschi L, Perata P.2009. Rice germination and seedling growth in the absence of oxygen.Ann Bot, 103(2): 181-196. |
| [28] | Mandal A B, Maiti A, Biswas A.2003. Somatic embryogenesis in root derived callus of indica rice. Plant Tiss Cult, 13(2): 125-133. |
| [29] | Manickavelu A, Nadarajan N, Ganesh S K, Ramalingam R, Raguraman S, Gnanamalar R P.2006. Organogenesis induction in rice callus by cyanobacterial extracellular product.Afr J Biot, 5(5): 437-439. |
| [30] | Miransari M, Smith D L.2014. Plant hormones and seed germination.Environ Exp Bot, 99: 110-121. |
| [31] | Mishra R, Rao G J N.2016. In-vitro and rogenesis in rice: Advantages, constraints and future prospects. Rice Sci, 23(2): 57-68. |
| [32] | Molnár Z, Virág E, Ördög V.2011. Natural substances in tissue culture media of higher plants.Acta Biol Szeged, 55(1): 123-127. |
| [33] | Murashige T, Skoog F.1962. A revised medium for rapid growth and bio-agsays with tohaoco tissue cultures.Physiol Plant, 15(3): 473-497. |
| [34] | Oinam G S, Kothari S L.1995. Totipotency of coleoptile tissue in indica rice(Oryza sativa L. cv. ch 1039). Plant Cell Rep, 14(4): 245-248. |
| [35] | Pengkumsri N, Chaiyasut C, Saenjum C, Sirilun S, Peerajan S, Suwannalert P, Sirisattha S, Sivamaruthi B S.2015a. Physicochemical and antioxidative properties of black, brown and red rice varieties of northern Thailand.Food Sci Technol, 35(2): 331-338. |
| [36] | Pengkumsri N, Chaiyasut C, Sivamaruthi B S, Saenjum C, Sirilun S, Peerajan S, Suwannalert P, Sirisattha S, Chaiyasut K, Kesika P.2015b. The influence of extraction methods on composition and antioxidant properties of rice bran oil.Food Sci Technol, 35(3): 493-501. |
| [37] | Priya A M, Pandian S K, Manikandan R.2012. The effect of different antibiotics on the elimination of Agrobacterium and high frequency Agrobacterium-mediated transformation of indica rice(Oryza sativa L.). Czech J Genet Plant Breeding, 48(3): 120-130. |
| [38] | Raemakers C J J M, Sofiari E, Jacobsen E, Visser R G F.1997. Regeneration and transformation of cassava.Euphytica, 96(1): 153-161. |
| [39] | Ramesh M, Murugiah V, Gupta A K.2009. Efficient in vitro plant regeneration via leaf base segments of indica rice(Oryza sativa L.). Ind J Exp Biol, 47(1): 68-74. |
| [40] | Rani W B, Amutha R, Muthulakshmi S, Indira K, Mareeswari P.2007. Diversity of rice leaf folders and their natural enemies.Res J Agric Biol Sci, 3(5): 394-397. |
| [41] | Ransom-Hodgkins W D.2009. The application of expression analysis in elucidating the eukaryotic elongation factor one alpha gene family in Arabidopsis thaliana. Mol Genet Genom, 281(4): 391-405. |
| [42] | Robbins W J, Bartley M A.1937. Vitamin B1 and the growth of excised tomato roots.Science, 85: 246-247. |
| [43] | Rout G R, Mohapatra A, Jain S M.2006. Tissue culture of ornamental pot plant: A critical review on present scenario and future prospects.Biotechnol Adv, 24(6): 531-560. |
| [44] | Sahoo K K, Tripathi A K, Pareek A, Sopory S K, Singla-Pareek S L.2011. An improved protocol for efficient transformation and regeneration of diverse indica rice cultivars. Plant Methods, 7: 49. |
| [45] | Sahrawat A K, Chand S.2004. High frequency plant regeneration from coleoptile tissue of barley (Hordeum vulgare L.). Plant Sci, 167(1): 27-34. |
| [46] | Sambrook J, Russel D W.2000. Molecular cloning : A laboratory manual. Cold Spring Harboc Lab Press, 3: 999. |
| [47] | Saleh B.2011. R-ISSR marker as a useful tool for detection of new genomic loci in Arthrocnemum macrostachyum. Biol Plant, 55(2): 327-330. |
| [48] | Sanyal I, Singh A K, Kaushik M, Amla D V.2005. Agrobacterium- mediated transformation of chickpea(Cicer arietinum L.) with Bacillus thuringiensis cry1Ac gene for resistance against pod borer insect Helicoverpa armigera. Plant Sci, 168(4): 1135-1146. |
| [49] | Sekar N, Veetil S K, Neerathilingam M.2013. Tender coconut water, an economical growth medium for the production of recombinant proteins in Escherichia coli. BMC Biotechnol, 13(1): 70. |
| [50] | Shilpha J, Silambarasan T, Largia M J V, Ramesh M.2014. Improved in vitro propagation, solasodine accumulation and assessment of clonal fidelity in regenerants of Solanum trilobatum L. by flow cytometry and SPAR methods. Plant Cell Tiss Organ, 117(1): 125-129. |
| [51] | Smolik M.2012. R-ISSR: Tool for generation of a new type of products, applied for the identification of putative molecular marker linked to QTL determined tolerance to nutrient deprivation stress in rye (Secale cereale L.). Not Bot Hort Agrob, 40(2): 238-246. |
| [52] | Storey M.2007. The harvested crop. In: Vreugdenhil D, Bradshaw J, Gebhardt C, Govers F, Taylor M, MacKerron D. Potato Biology and Biotechnology: Advances and Perspectives. Amsterdam: Elsevier: 441-470. |
| [53] | Thorpe T A.2007. History of plant tissue culture.Mol Biotechnol, 37: 169-180. |
| [54] | van Staden J, Menary R C.1976. Identification of cytokinins in the xylem sap of tomato.Z Pflanzenphys, 78(3): 262-265. |
| [55] | Wang M S, Zapata F J, Castro D C D.1987. Plant regeneration through somatic embryogenesis from mature seed and young inflorescence of wild rice (Oryza perennis Moench). Plant Cell Rep, 6(4): 294-296. |
| [56] | Ye C J, Yu Z W, Kong F N, Wu S W, Wang B.2005. R-ISSR as a new tool for genomic fingerprinting, mapping, and gene tagging.Plant Mol Biol Rep, 23(2): 167-177. |
| [57] | Zhang D, Wang Z H, Wang N N, Gao Y, Liu Y, Wu Y, Bai Y, Zhang Z B, Lin X Y, Dong Y Z, Ou X F, Xu C M, Liu B.2014. Tissue culture-induced heritable genomic variation in rice, and their phenotypic implications.PLoS One, 9(5): 5-7. |
| [1] | Md. Dhin Islam, Adam H. Price, Paul D. Hallett. Effects of Root Growth of Deep and Shallow Rooting Rice Cultivars in Compacted Paddy Soils on Subsequent Rice Growth [J]. Rice Science, 2023, 30(5): 459-472. |
| [2] | Sheikh Faruk Ahmed, Hayat Ullah, May Zun Aung, Rujira Tisarum, Suriyan Cha-Um, Avishek Datta. Iron Toxicity Tolerance of Rice Genotypes in Relation to Growth, Yield and Physiochemical Characters [J]. Rice Science, 2023, 30(4): 321-334. |
| [3] | Du Shuanglin, Wang Zhongwei, Chen Yun, Tan Yao, Li Xiang, Zhu Wenping, He Guanghua, Lei Kairong, Guo Longbiao, Zhang Yi. Coleoptile Purple Line Regulated by A-P Gene System Is a Valuable Marker Trait for Seed Purity Identification in Hybrid Rice [J]. Rice Science, 2022, 29(5): 451-461. |
| [4] | Yousef Alhaj Hamoud, Hiba Shaghaleh, Wang Ruke, Willy Franz Gouertoumbo, Amar Ali Adam hamad, Mohamed Salah Sheteiwy, Wang Zhenchang, Guo Xiangping. Wheat Straw Burial Improves Physiological Traits, Yield and Grain Quality of Rice by Regulating Antioxidant System and Nitrogen Assimilation Enzymes under Alternate Wetting and Drying Irrigation [J]. Rice Science, 2022, 29(5): 473-488. |
| [5] | Chen Wei, Cai Yicong, Shakeel Ahmad, Wang Yakun, An Ruihu, Tang Shengjia, Guo Naihui, Wei Xiangjin, Tang Shaoqing, Shao Gaoneng, Jiao Guiai, Xie Lihong, Hu Shikai, Sheng Zhonghua, Hu Peisong. NRL3 Interacts with OsK4 to Regulate Heading Date in Rice [J]. Rice Science, 2022, 29(3): 237-246. |
| [6] | Shuting Yuan, Chunjue Xu, Wei Yan, Zhenyi Chang, Xingwang Deng, Zhufeng Chen, Jianxin Wu, Xiaoyan Tang. Alternative Splicing of OsRAD1 Defines C-Terminal Domain Essential for Protein Function in Meiosis [J]. Rice Science, 2020, 27(4): 289-301. |
| [7] | Vijayaraghavareddy Preethi, Xinyou Yin, C. Struik Paul, Makarla Udayakumar, Sreeman Sheshshayee. Responses of Lowland, Upland and Aerobic Rice Genotypes to Water Limitation During Different Phases [J]. Rice Science, 2020, 27(4): 345-354. |
| [8] | Hussain Kashif, Yingxing Zhang, Anley Workie, Riaz Aamir, Abbas Adil, Hasanuzzaman Rani Md., Hong Wang, Xihong Shen, Liyong Cao, Shihua Cheng. Association Mapping of Quantitative Trait Loci for Grain Size in Introgression Line Derived from Oryza rufipogon [J]. Rice Science, 2020, 27(3): 246-254. |
| [9] | Mohammed Sulaiman, Abd Samad Azman, Rahmat Zaidah. Agrobacterium-Mediated Transformation of Rice: Constraints and Possible Solutions [J]. Rice Science, 2019, 26(3): 133-146. |
| [10] | Nurdiani Dini, Widyajayantie Dwi, Nugroho Satya. OsSCE1 Encoding SUMO E2-Conjugating Enzyme Involves in Drought Stress Response of Oryza sativa [J]. Rice Science, 2018, 25(2): 73-81. |
| [11] | Fernando Polesi Luís, Bruder Silveira Sarmento Silene, Guidolin Canniatti-Brazaca Solange. Starch Digestibility and Functional Properties of Rice Starch Subjected to Gamma Radiation [J]. Rice Science, 2018, 25(1): 42-51. |
| [12] | Singh Bhupinder, Raja Reddy Kambham, Diaz Redoña Edilberto, Walker Timothy. Screening of Rice Cultivars for Morpho-Physiological Responses to Early-Season Soil Moisture Stress [J]. Rice Science, 2017, 24(6): 322-335. |
| [13] | Jini D., Joseph B.. Physiological Mechanism of Salicylic Acid for Alleviation of Salt Stress in Rice [J]. Rice Science, 2017, 24(2): 97-108. |
| [14] | Fernando Polesi Luís, Divino da Matta Junior Manoel, Bruder Silveira Sarmento Silene, Guidolin Canniatti-Brazaca Solange. Starch Digestibility and Physicochemical and Cooking Properties of Irradiated Rice Grains [J]. Rice Science, 2017, 24(1): 48-55. |
| [15] | S. Abe S., Yamasaki Y., Wakatsuki T.. Assessing Silicon Availability in Soils of Rice-Growing Lowlands and Neighboring Uplands in Benin and Nigeria [J]. Rice Science, 2016, 23(4): 196-202. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||